A risk forecasting process for nanostructured materials, and nanomanufacturing
Texte intégral
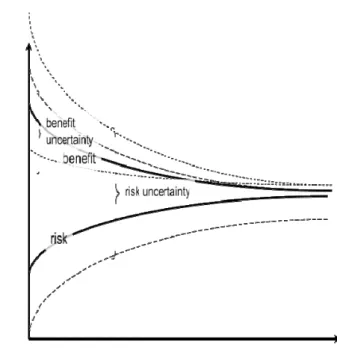
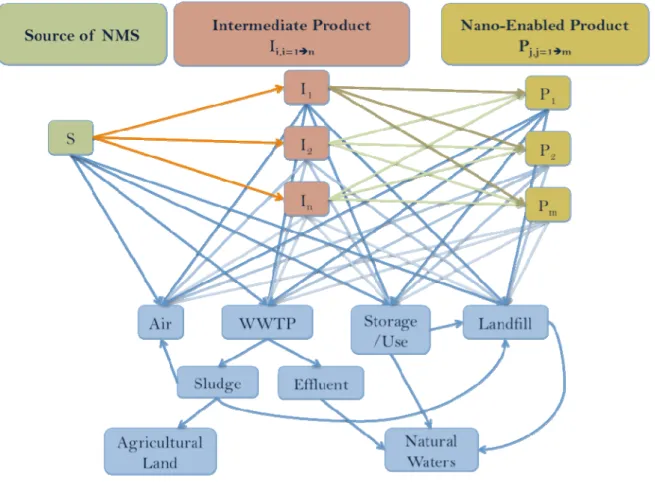
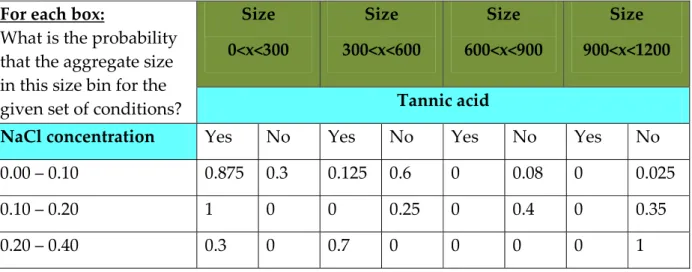
Figure

Documents relatifs
Attacks of a domestic origin were isolated, carried out by groups or individuals with disparate agendas, neither creating major economic disruption nor many casualties (Kunreuther
This paper proposes a formal language, Event-Condition-Action-Event (ECAE), for integrating Colored Petri Nets (CPN)-based business process with a set of business rules.. We
CHU, Professor of Pharmacology, National College of Medicine, Shanghai.. Secretary:
Building on these early findings, one can envision several clinical applications in the near term, including (i) stratifying patients for checkpoint blockade based on the number
Based on an international Delphi study, this article introduces 14 criteria that (potential) BPMM users must consider to obtain a fit for pur- pose. The findings will
The goal of the current paper is to describe a modern risk management framework for calculating possible time deviations from defined by business continuity or do-
At the same time, business wit- nesses also concerned themselves keenly with labor issues at Congress: in fact, the individual hearings that drew the greatest number of
Ideally, every business process management system (BPMS) would provide process-related risk management capabilities covering the three main aspects of risk management: risk