Introduction to PNC Science and Technology
Texte intégral
Figure

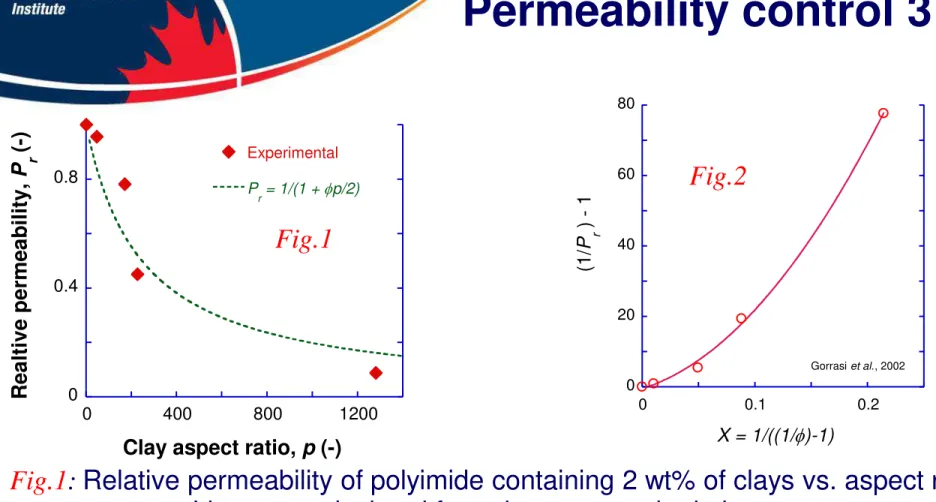
Documents relatifs
Ringel, The preprojective algebra of a tame quiver: the irreducible components of the module varieties, Trends in the representation theory of finite dimensional algebras (Seattle,
We investigate the transience/recurrence of a non-Markovian, one-dimensional diffusion process which consists of a Brownian motion with a non-anticipating drift that has two phases –
Unité de recherche INRIA Rennes : IRISA, Campus universitaire de Beaulieu - 35042 Rennes Cedex (France) Unité de recherche INRIA Rhône-Alpes : 655, avenue de l’Europe -
The aim of this study was to evaluate the global potential reduction of such emissions by the use of addi- tives (2:1 clay and/or biochar): during (vermi)composting processes and
The C++ language files contain the definition of the stochastic types on which the control of accuracy can be performed, CADNA C specific functions (for instance to enable or
Dynamic light scattering shows two populations of diffusion coefficients; one population originates from “free” microemulsion droplets and the other from the polymer (for
It also seemed relevant to examine the value attributed to this belief in itself, therefore we asked: is having to work with a colleague who believes (vs. who does not believe) in
Two leak-proof infectious waste bags - one for disposable material (destruction) - one for reusable materials (disinfection) Step 1c: Fill out patient documentation. Label