Publisher’s version / Version de l'éditeur:
Materiaux et constructions. Materials and Structures, 8, 47, pp. 373-376, 1975-09
READ THESE TERMS AND CONDITIONS CAREFULLY BEFORE USING THIS WEBSITE.
https://nrc-publications.canada.ca/eng/copyright
Vous avez des questions? Nous pouvons vous aider. Pour communiquer directement avec un auteur, consultez la première page de la revue dans laquelle son article a été publié afin de trouver ses coordonnées. Si vous n’arrivez pas à les repérer, communiquez avec nous à PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca.
Questions? Contact the NRC Publications Archive team at
PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca. If you wish to email the authors directly, please see the first page of the publication for their contact information.
NRC Publications Archive
Archives des publications du CNRC
This publication could be one of several versions: author’s original, accepted manuscript or the publisher’s version. / La version de cette publication peut être l’une des suivantes : la version prépublication de l’auteur, la version acceptée du manuscrit ou la version de l’éditeur.
Access and use of this website and the material on it are subject to the Terms and Conditions set forth at
Hydration and strength characteristics of synthetic Al-, Mg- and Fe
alites
Mascolo, G.; Ramachandran, V. S.
https://publications-cnrc.canada.ca/fra/droits
L’accès à ce site Web et l’utilisation de son contenu sont assujettis aux conditions présentées dans le site LISEZ CES CONDITIONS ATTENTIVEMENT AVANT D’UTILISER CE SITE WEB.
NRC Publications Record / Notice d'Archives des publications de CNRC:
https://nrc-publications.canada.ca/eng/view/object/?id=e8de4d9d-40b4-4825-8c99-6d49b8350434 https://publications-cnrc.canada.ca/fra/voir/objet/?id=e8de4d9d-40b4-4825-8c99-6d49b8350434Ser
T M
N21d
no.
678
National Research
Conseil national
c. 2
I
*
Council Canada
de recherches Canada
BLDG
DBR Paper No. 678
Division of Building Research
Price
10
cents
HYDRATION AND STRENGTH CHARACTERISTICS
OF
SYNTHETIC Al-
,
Mg
-
AND
Fe ALITES
by
G.
$4ascolo and V.S. Ramachandran59604"
Reprinted fromMaterials and Construction,
Vol. 8, No. 47, September/October 1975,
1
l q c - q-J"C.
RFSEARCH
1
p. 373 376
I
-
LIBRARY
-
g
I
h
I
1i
r t f r .'"' .I RESEARCH COUNClLI
OTTAWA
NRCC 15308
Hydration and strength characteristics
of synthetic
Al-, M g - and Fe alites
( * ) .
G. MASCOLO ( I ) V. S. RAMACHANDRAN (')
Alites containing different amounts of Al, M g or Fe were hydrated up to 30 days and their kinetics of hydration and compressive strengths were determined. At the same degree of hydration Fe-alites showed greatest strength. There was evidence that the nature of C - S H formed in different alites is not the same.
INTRODUCTION
The silicate-based phases, tricalcium silicate
(3 CaO
.
SiO,, abbreviated to C,S) (3) and dicalciumsilicate (2CaO. SiO,, abbreviated to C,S) together
constituting about 70-80
%
of ,Portland cement muchinfluence many of the properties of concrete. In commercial Portland cements however, these silicates are solid solutions containing a minor amount of oxides. Solid solutions of tricalcium silicate and dicalcium silicate are known respectively as alite and belite.
The major phase in Portland cement being alite, much more attention has been directed to its study than to other clinker minerals. In recent years some synthetically prepared alites have been examined with a view to investigating the possibility of producing an alite with higher reactivity and strength than the (*) Work carried out in Naples. Supported by National Research Council of Italy (C.N.R.).
( I ) Istituto di Chimica Applicata, Facolta di Ingegneria,
Universita di Napoli, Italy.
(2) Division of Building Research, National Research Council
of Canada, Ottawa, Canada.
(3) In cement chemistry C=CaO, S =Si02, H = H 2 0 .
normal alite. The hydraulic properties of alite depend on the chemical nature of the addition, its amount and the heat treatment accorded during their prepa- ration ([I]-[7]). In spite of some work on alites controversy still exists on many aspects and no general agreement exists on the relationship between'the reacti- vity and mechanical properties of various types of alites. In this paper alites containing different amounts of Al, Mg and Fe were prepared of same particle size and hydrated to different periods. Attempts have been made to understand the mechanical behaviour in terms of the kinetics of hydration.
EXPERIMENTAL
Pure 3CaO.Si0, was prepared by heating a stoi- chiometric mixture of CaCO, and SiO, (Reagent grade C. ERBA). Tricalcium silicate solid solutions (alites) were prepared by calcining a mixture of A1 (OH),, (MgCO,),. Mg (OH),. 5 H 2 0 or Fe,O, with stoichiometric proportions of CaCO, and SiO,. Each sample was ground in a ball mill to a Blaine fineness
of 5,040
k
40 cm2/g. The chemical composition andcrystallographic forms of the samples are described in Table I.
TABLE I
Sample Chemical analysis Crystallographic
No. (weight per cent) form
SiO, CaO Free CaO MgO Fez03
1 26.07 73.47 0.33
-
- - 2 26.08 73.17 0.27 0.45 --
1, 3 25.02 71.90 0.12 1 .OO - - T, - - T,, 4 25.64 72.25 0.19 2.01 5 25.98 73.57 0.15 - 0.32 - MI 6 25.68 73.00 0.11 - 0.90 - T,-
- TI, 7 25.91 73.51 0.50 0.80 TI 8 26.35 72.50 0.24 --
1.08 T11G. MASCOLO
-
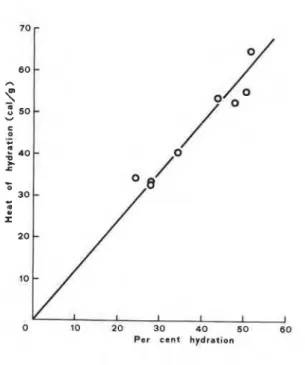
V. S. RAMACHANDRANFig. 1. - Heats of hydration of alites compared to the degree of hydration at 1 day.
1 2 3 5 7 14 30
Hydration times . days
2 0 0
Mg
-
alites 100Hydration
.
per centFig. 2. - Kinetics of hydration of alites..
Fig. 3. - Compressive strengths of alites in terms of the degree of hydration. I 1 I I I I . 2 3 5 7 14 30 H y d r a t i o n t i m e s
.
daysFig. 4. - Temperatures of the exothermal peaks due to j-wollastonite formation in hydrated alites.
VOL. 8 - No 47
-
MATERIAUX ET CONSTRUCTIONSACKNOWLEDGEMENTS
T h a n k s a r e due to G. M. Polomark of Division of Building Research, National Research Council, Ottawa, for experimental assistance and to P. J. Sereda for his keen interest in the work.
L'hydratation et les caractkristiques de rbistance
des alites synthktiques d'Al, de Mg et de Fe. - Des
alites contenant dgdrentes quantitb d'Al, de Mg ou de Fe sont hydratdes pendant une pdriode allant jusqu'a 30 jours et on en ddtermine la cindtique d'hydra-
tation et la rbistance a la compression. Les courbes calorimdtriques de conduction indiquent que, malgrd I'hydratation initiale rapide de quelques dchantillons, celle-ci ralentit ensuite en moins d'une journde. L'dtude montre dgalement que la vitesse de la rdaction ne se corrile pas directement avec la forme cristallographique de l'alite ou avec le genre d'ion dans le rdseau. I1 y a quelques corrdlation entre la rbistance
2
la compression et le genre d'dldment dans la solution solide. Parmi les alites mises a I'dtude, les alites de Fe deviennent le plus du C,S et leur rdsistance est plus dlevde que celle des autres alites quel que soit le degrd d'hydra- tation. La tempdrature d laquelle se produit la cristalli-sation en B-wollastonite varie d'une alite a l'autre, ce qui indique que le genre de C - S H formd n'est pas le mgme dans toutes les alites.
[l] SERSALE R. - Hydraulic behaviour of Al, Fe, Mg bearing alites. VI International Congress on Chemistry of Cement, Moscow, Supplementary Paper, Section 11,
1974.
[2] KURDOWSKI W. - Influence of minor components on hydraulic activity of Portland Cement clinker. VI Inter- national Congress on Chemistry of Cement, Moscow, Supplementary Paper, Section I, 1974.
[3] KONDO, R., UEDA, S. - Kinetics and Mechanisms of the hydration of cements. Principal Paper, Proc. V, International Symposium on Chemistry of Cements, Tokyo, Vol. 2, pp. 203-248, 1968.
[4] ONO, Y., UNO, T., AND SODA,^. - Effect of crystal- lographic properties of alite and belite on the strength in cement. Semento Gijitsu. Nempo, 19, pp. 93-103, 1965.
[5] LOCHER, F., RICHARTZ, W. - Study of the hydration mechanism of cement. VI International Congress on Chemistry of Cement, Moscow, Principal Paper, 1974. [6] REGOURD, M., GUINIER, A. - The crystal chemistry of the constituents of Portland cement clinker. VI Inter- national Congress on Chemistry of Cement, Moscow, Principal Paper, 1974.
[7] DESCAMPS, J., FIERENS, P., AND VERHAEGEN, J. P. - Chemical defects and hydration of doped tricalcium silicates. VI International Congress on Chemistry of Cement, Moscow, Supplementary Paper, Section 11, 1974.
[8] RAMACHANDRAN, V. S. - Estimation of tricalcium silicate through polymorphic transformation. J . Thermal
Analysis, 3, pp. 181-190, 1971.
[9] MASCOLO, G . - Evaluation of the degree of hydration of synthetic alites by quantitative dzffrential thermal analysis, Thermochimica Acta, 10, pp. 245, 251, 1974. [lo] MASCOLO, G., MARCHESE, B., FRIGIONE, G., AND
SERSALE, R. -Influence ofpolymorphism and stabilizing
ions on the strength of alite. Journal Americ. Ceram. Society, 4, 39, 1973.
[ l l ] YAMAGUCHI, G . , SHIRASUKA, K., AND OTA T. - Comparison of hydration properties between monoclinic and inverted triclinic alite. Symposium on Structure of Portland Cement Paste and Concrete, Highway Research Board Special Report No. 90, Washington, 1966.
[12] RAMACHANDRAN, V. S. - Applications of dzfferential thermal analysis in cement chemistry, Chemical Publishing Co., New York, pp. 81-87, 1969.