Publisher’s version / Version de l'éditeur:
Vous avez des questions? Nous pouvons vous aider. Pour communiquer directement avec un auteur, consultez la
première page de la revue dans laquelle son article a été publié afin de trouver ses coordonnées. Si vous n’arrivez
pas à les repérer, communiquez avec nous à PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca.
Questions? Contact the NRC Publications Archive team at
PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca. If you wish to email the authors directly, please see the
first page of the publication for their contact information.
https://publications-cnrc.canada.ca/fra/droits
L’accès à ce site Web et l’utilisation de son contenu sont assujettis aux conditions présentées dans le site
LISEZ CES CONDITIONS ATTENTIVEMENT AVANT D’UTILISER CE SITE WEB.
Proceedings: Permafrost International Conference, pp. 225-229, 1963-10-01
READ THESE TERMS AND CONDITIONS CAREFULLY BEFORE USING THIS WEBSITE.
https://nrc-publications.canada.ca/eng/copyright
NRC Publications Archive Record / Notice des Archives des publications du CNRC :
https://nrc-publications.canada.ca/eng/view/object/?id=36918fa4-66b1-493e-8b57-ff385bc44b5d
https://publications-cnrc.canada.ca/fra/voir/objet/?id=36918fa4-66b1-493e-8b57-ff385bc44b5d
NRC Publications Archive
Archives des publications du CNRC
This publication could be one of several versions: author’s original, accepted manuscript or the publisher’s version. /
La version de cette publication peut être l’une des suivantes : la version prépublication de l’auteur, la version
acceptée du manuscrit ou la version de l’éditeur.
Access and use of this website and the material on it are subject to the Terms and Conditions set forth at
Suction and its effects in unfrozen water of frozen soils
Reprinted from t h e PROCEEDINGS: PERMAFROST INTERNATIONAL CONFERENCE, N A S - N R C , P u b l i c a t i o n 1287
SUCTION AND ITS EFFECTS I N UNFROZEN WATER OF FROZEN SOILS
P. J. WILLIAMS, Norwegian G e o t e c h n i c a l I n s t i t u t e , O s l o
S u b s t a n t i a l q u a n t i t i e s of w a t e r remain unfrozen in s o i l s a t t e m p e r a t u r e s of s e v e r a l d e g r e e s b e l o w ODC [ l , 2 , 3 1 . The proportion of unfrozen w a t e r d e c r e a s e s a s t h e temperature i s l o w e r e d , b u t a s much a s half of the w a t e r may e x i s t unfrozen a t - l ° C . This unfrozen water h a s b e e n a t t r i b u t e d [ 4 ] to the s u c t i o n s or n e g a t i v e pore p r e s s u r e s t h a t d e v e l o p a s a r e s u l t of i c e - l e n s growth in the s o i l . A n e g a t i v e pore p r e s s u r e in a s a t u r a t e d s o i l , in t h e a b s e n c e of e x t e r n a l l o a d i n g , r e s u l t s in a p o s i t i v e e f f e c t i v e s t r e s s ( a s t r e s s a c t i n g a c r o s s the g r a i n - to-grain c o n t a c t s ) e q u a l t o t h e n e g a t i v e pore p r e s s u r e . An i n c r e a s e in e f f e c t i v e s t r e s s c a u s e s c o n s o l i d a t i o n in c o m p r e s s i b l e s o i l s . It i s therefore of i n t e r e s t to i n v e s t i g a t e the c o n s o l i d a t i o n t h a t s h o u l d o c c u r in a frozen s o i l a s a r e s u l t of n e g a t i v e pore p r e s s u r e s in t h e unfrozen part of t h e moisture c o n t e n t . Such c o n s o l i d a t i o n would a l s o confirm thz e x i s t e n c e a n d , to s o m e e x t e n t , t h e magnitude of t h e n e g a t i v e pore p r e s s u r e s - a n d their r e l a t i o n s h i p to t e m p e r a t u r e . FREEZING EXPERIMENTS Direct i n v e s t i g a t i o n of t h e s e e f f e c t s i s d i f f i c u l t b e c a u s e of t h e irregularly d i s t r i b u t e d i c e - l e n s e s in a frozen s o i l w h i c h , on t h e o n e h a n d , g r e a t l y r e d u c e p e r m e a b i l i t y , a n d on the other h a n d , i n c r e a s e t h e o v e r - a l l s o i l volume. It i s n e c e s s a r y to d e v i s e a n experiment in which l e n s formation i s prevented in a part of t h e s o i l l a r g e e n o u g h for s u b s e q u e n t s t u d y of d e g r e e of c o n s o l i d a t i o n a n d w a t e r c o n t e n t . P e r s p e x r i n g s 2.2 cm ID a n d 0 . 4 cm in h e i g h t w e r e m a d e . A ring w a s f i l l e d w i t h a s o i l s a m p l e prepared f l u s h to t h e s u r - f a c e . P i e c e s of a membrane, p r s v i o u s l y s c a k e d in w a t e r , w e r e t h e n placed a c r o s s t h e f a c e s of t h e ring a n d s o i l s a m p l e a n d p r e s s e d tightly a g a i n s t t h e p e r s p e x w i t h a c l a m p a r r a n g e - ment (Fig. 1 ) . A thin s m e a r of petroleum jelly w a s u s u a l l y p l a c e d o n t h e p e r s p e x ring where it c a m e in c o n t a c t w i t h the membrane. Two further p i e c e s of t h e s a m e , or s o m e t i m e s d i f f e r e n t , s o i l were t h e n p r e s s e d a g a i n s t the, e x p o s e d s i d e s of t h e m e m b r a n e s .
This a s s e m b l y w a s s l o w l y c o o l e d to a c h o s e n n e g a t i v e t e m p e r a t u r e . Freezing (with i c e - l e n s formation) o c c u r s in t h e e x p o s e d s o i l , b u t n o t in t h a t b e t w e e n t h e membranes b e c a u s e of t h e a b s e n c e of a n i c e n u c l e u s . S p o n t a n e o u s n u c l e a t i o n d o e s n o t occur-initially b e c a u s e t h e s a l r ~ p l e i s s m a l l a n d s u b s e q u e n t l y for r e a s o n s which w i l l become apparent-and i c e growth c a n n o t o c c u r through t h e membrane. The membrane i s of the water-permeable type u s e d in p r e s s u r e membrane t e s t s
i s ] .
The membrane p o r e s a r e s o s m a l l t h a t i c e growth o n l y o c c u r s within them a t t e m p e r a t u r e s of a t l e a s t s e v e r a l d e g r e e s b e l o w O°C.A modified d o m e s t i c - t y p e refrigerator w a s u s e d . A mercury c o n t a c t thermometer s w i t c h with a r e l a y s y s t e m maintained a fairly c o n s t a n t t e m p e r a t u r e . To a v o i d d e s i c c a t i o n and to e n s u r e uniform t e m p e r a t u r e , the s a m p l e a s s e m b l i e s w e r e p l a c e d in c l o s e d j a r s together w i t h p i e c e s of i c e t o s t a r t n u c l e a t i o n in the e x p o s e d s o i l . The s p e c i m e n s w e r e g e n e r a l l y maintained a t t h e c h o s e n temperature for three d a y s . T e s t s w i t h thermocouples i n d i c a t e d t h a t t h e s a m p l e a t t a i n e d t h e c h o s e n temperature from 5 to 1 0 h o u r s a f t e r refrigeration.
After r e f r i g e r a t i o n , t h e a s s e m b l y w a s d i s m a n t l e d ; t h e w a t e r c o n t e n t and volume of t h e inner s o i l layer ( l o c a t e d b e t w e e n t h e membranes) were d e t e r m i n e d . A s p e c i a l l y c o n s t r u c t e d g l a s s pycnometer (Fig. 2) c o n t a i n i n g paraffin w a s u s e d to d e t e r m i n e t h e volume. To o b t a i n s u f f i c i e n t a c c u r a c y , c o n - s i d e r a b l e a t t e n t i o n to temperature a n d other e f f e c t s w a s n e c e s s a r y .
M a n y further t e s t s involving determination of w a t e r c o n t e n t o n l y w e r e d o n e o v e r a r a n g e of O0 t o -3.3OC. In a l l c a s e s t h e
Bmss clamping rings,
/
\
/erspex ring ,Soil sample
\
F i g . 1 . Sample a s s e m b l y for f r e e z i n g e x p e r i m e n t s
F i g . 2 . Pycnometer u s e d for dry d e n s i t y d e t e r m i n a t i o n s
w a t e r c o n t e n t of the inner l a y e r w a s s u b s t a n t i a l l y l e s s t h a n t h e i n i t i a l w a t e r c o n t e n t . Preliminary t e s t s showed t h a t t h e w a t e r c o n t e n t of the inner l a y e r s b e c a m e p r a c t i c a l l y c o n s t a n t within t h r e e d a y s . In o n e t e s t s i m i l a r s o i l s a m p l e s p l a c e d in d i f f e r e n t p a r t s of t h e r e f r i g e r a t o r might g i v e r e s u l t s differing b y 1 to 2% of dry w e i g h t . T h i s w a s l a r g e l y d u e to s l i g h t t e m - p e r a t u r e d i f f e r e n c e s . S a m p l e s of t h e s a m e s o i l in o n e b o t t l e c o n s i s t e n t l y s h o w e d s i m i l a r w a t e r c o n t e n t s for t h e inner l a y e r within 0 . 1 t o 0.2% dry w e i g h t . When o n e s a m p l e w a s removed a n d the temperature t h e n lowered for a further p e r i o d , s a m p l e s removed l a t e r s h o w e d lower w a t e r c o n t e n t . In a l l c a s e s moisture c o n t e n t ( i c e a n d water) of t h e o u t e r s o i l l a y e r s (Fig. 1) w a s i n c r e a s e d .
VOLUME MEASUREMENTS
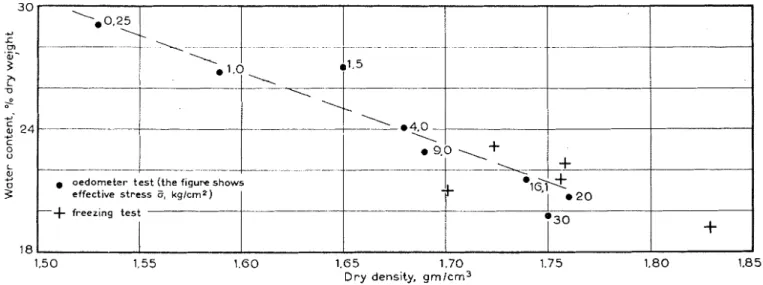
R e s u l t s of i n v e s t i g a t i o n s o n Leda c l a y KNB a r e s h o w n in Fig. 3. From volume d e t e r m i n a t i o n s , dry d e n s i t y ( w e i g h t dry m a t e r i a l p e r u n i t volume of s o i l i n m o i s t c o n d i t i o n in grams per c u b i c c e n t i m e t e r ) w a s c a l c u l a t e d a n d plotted ( F i g . 3 , c r o s s e s ) a s a function of w a t e r c o n t e n t . C o n s o l i d a t i o n of s a m p l e s from t h e inner l a y e r of t h e membrane a s s e m b l y occurred during freezing. T h i s i s c l e a r l y s h o w n b y c o m p a r i s o n with r e s u l t s of o e d o m e t e r t e s t s on s i m i l a r m a t e r i a l (Fig. 3 , c i r c l e s ) . If the m a t e r i a l i s s a t u r a t e d t h e r e i s a unique r e l a t i o n s h i p b e t w e e n d r y d e n s i t y , w a t e r c o n t e n t , a n d e f f e c t i v e s t r e s s , for t h e o b s e r v a t i o n s s h o w n in F i g . 3 . S p e c i f i c w e i g h t of the s o i l ? 2 5
1.50 1.55 1.60 1.65 1.70 1.75 1.80 1.8 5
Dry density, g m l c m 3
Fig. 3 . Dry density a s a function o f moisture content
particles was found t o be 2 . 7 8 g per cu c m , and from volu
-
to temperature. This direct procedure, while giving themetric consideration, dry density and water content relation- approximate magnitude o f the pore pressures, i s not entirely
ship for the saturated state was calculated (Fig. 3 , dashed s a t i s f a c t o r y . Reasons for this are apparent from earlier
l i n e ) . The relationship o f the observed points to this line experimental work [ 4 ] which can be used to obtain a more
shows that the soil was saturated over the range shown. accurate approximation o f the temperature-negative
According t o the e f f e c t i v e stress equation pore-pressure relationship.
6 = 0 - l l
INTERPRETATION OF OBSERVED WATER CONTENTS FROMwhere
5
i s e f f e c t i v e s t r e s s , i s total stress (equals CALORIMETRIC STUDIES OF FROZEN SOILSapplied load in oedometer), and p i s pore water pressure,
the load applied in the oedometer i s equal to the e f f e c t i v e In this earlier work, calorimetric methods were used t o deter-
stress (which i s responsible for consolidation) after equilib- mine the quantity o f water remaining unfrozen at various
rium i s reached, because pore water pressure i s then atmos- negative temperatures. Results o f this type can be expressed
pheric ( i . e . , z e r o ) . The relationship b ~ t w e e n dry density a s (and moisture content) and e f f e c t i v e stress i s thus given b y the oedometer t e s t s . The figure for e f f e c t i v e stress shown beside e a c h point in Fig. 3 , obtained from the oedomzter t e s t s , illustrates this relationship.
The magnitude o f e f f e c t i v e s t r e s s e s t o which samples from the membrane experiment have been subjected i s indicated b y those s t r e s s e s that in oedometer t e s t s produced a similar moisture content and dry d e n s i t y . In the absence o f external loading ( a s in the membrane freezing experiment), e f f e c t i v e s t r e s s i s e q u a l , but o f opposite s i g n , to pore water pressure. Positive e f f e c t i v e stress giving rise to consolidation arises from, and i s numerically equal t o , negative pore pressure developed by f r e e z i n g .
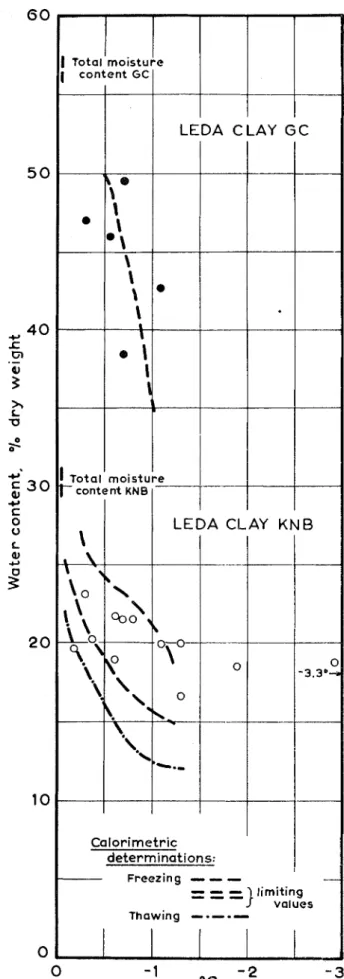
RELATIONSHIP OF TEMPERATURE TO NEGATIVE PORE PRESSURE As the temperature i s lowered and more pore water i s trans- ferred to ice m a s s e s , negative pore pressures (and hence the e f f e c t i v e stress) become greater. Negative pore pressure a s a function o f temperature cannot be determined from the f e w observations in Fig. 3 . However, i f water content o f the inner layer from the membrane experiment was known a s a function o f temperature with s u f f i c i e n t accuracy, it would be possible to combine this with information in Fig. 3 to obtain values o f negative pore pressure as a function o f temperature. Water content o f the inner ( i c e - f r e e ) layer, determined in many t e s t s i s shown in Fig. 4 (points and circles) a s a function o f temperature. In general, water content i s l e s s for lower tem- peratures. In addition t o the two soils illustrated, t e s t s were also made on an illitlc clay from Asrum, Norway, and a bentonite from W i n n i p e g , Canada. These t e s t s also showed decreased water content o f the inner layer for lower tempera-
ture. From information in Fig. 3 , it should be possible to find
negative pore pressure (which i s n u m e r i c a l l y equal to the e f f e c t i v e stress causing consolidation) corresponding to the d i f f e r e n t water contents o f the inner layer (Fig. 4) and thus
weight o f watsr remaining unfrozen
dry weight o f soil x 100
and then shown on Fig. 4 . At l e a s t to about - l ° C , water con- tent o f the inner ( i c e - f r e e ) layer in the membrane experiment i s , within limits o f experimental error, equal t o water content o f "normally" frozen soil ( i n which ice i s a l s o present). During the freezing process in the membrane experiment, water i s transferred from the inner layer t o the outer layers (where ice i s formed) because o f a pressure gradient in the water. W h e n water content o f the inner layer becomes constant, the water in both inner and outer layers would be expected t o have the same negative pressure. Thus the situation o f the inner soil l a y e r , isolated by membranes, does not d i f f e r fundamentally from parts o f the normally frozen soil in which i c e - l e n s e s may happen to be a b s e n t . The inner soil layer has the same ( u n f r o z e n ) water content under the same negative pressure.
In the calorimetric investigations it was a l s o found that the unfrozen water content a s a function o f temperature h a s ,
because o f h y s t e r e s i s , somewhat d i f f e r e n t v a l u e s depending = on whether the soil i s in process o f freezing or thawing
(Fig. 4 ) . In the membrane experiment, similar small d i f f e r - e n c e s in water content are expected depending on whether the measured temperature o f the refrigerator i s reached by cooling or slight warming. Temperature control o f the refrigerator was such that fluctuations o f * 0 . 1°C might occur and t h e s e are largely responsible for the scatter in the points shown. In addition, water content was t o some extent dependent on the degree o f disturbance o f the sample.
This relatively small scatter in the observed points i s s u f f i c i e n t to prevent an accurate calculation o f the relation- ship between temperature and e f f e c t i v e s t r e s s . Fig. 3 shows that small changes in water content are associated with very large changes in e f f e c t i v e s t r e s s , so that it i s necessary t o
know the water content-temperature relationship (Fig. 4) for
G O
5 0
Y4 0
r:
Gl.-
;
2'
TI 0 3 Y;
3 0
Yc
0 0 haJ
+IP
2 0
10
0
0
-
1
-2
-3
OC
Fig. 4 . Equilibrium water contents o f inner layers (where ice formation did not occur)
I t I
Calorimetric
determinations:
Freezing- - -
---
-
=
=)
limiting values Thawing---
DETERMINATION.OF TEMPERATURE AND NEGATIVE PORE PRESSURE REUTIONSHIP
Pressure e f f e c t s are generally accepted as mainly responsible for the presence o f unfrozen water in frozen s o i l s . Earlier work [ 4 ] provided a quantitative evaluation o f t h e s e e f f e c t s . Suction-water content relationships [ 6 ] were determined at room temperature for the soils investigated calorimetrically. A hypothesis was proposed that as freezing occurred and water w a s transferred into i c e - l e n s e s , the water remaining in the pores would b e under an increasing suction (negative pore pressure). This suction could b e predicted from suction-water content tests-and i s probably responsible for the presence o f the unfrozen water because o f i t s e f f e c t on the freezing point o f the latter. The suction predicted for each unfrozen water content was found to be related to the temperature b y an equation o f the type b y Schofield [ 7 ] :
where
H = suction expressed a s height o f a column o f water, cm (=g/sq 'cm); L = latent heat o f freezing o f water (3.336 x l o 9 e r g / g ) ; T = temperature, OK; AT = negative
temperature, OC; and gr = gravity.
This relationship could only be determined following a large number o f c a r e f u l l y controlled t e s t s on various soils-
reasoning a s a b o v e . Results are summarized in Fig. 5 , modi- fied from [ 4 ] . Except for a necessary minor correction t o allow for the e f f e c t o f dissolved salts on freezing point, the equation is apparently valid for all s o i l s . The equation gives the freezing-point depression o f water under negative pres- s u r e , which i s in contact with ice under atmospheric pressure
[ a ] .
Although it appears unlikely at f i r s t , t h i s situation probably occurs in soils where the ice phase is in discrete l e n s e s or m a s s e s larger than pore s i z e , while the water l i e s within small pores under the influence o f capillary and other e f f e c t s .TEMPERATURE AND NEGATIVE PORE PRESSURE CONCLUSIONS Although earlier work [ 4 ] did not show directly the e x i s t e n c e o f negative pore pressures, present experiments clearly dem- onstrate these pressures and a l s o confirm that their magnitude i s similar to that predicted from ( 2 ) . A thermodynamic equa- tion o f the type o f ( 2 ) d o e s , t h e r e f o r e , correctly describe the temperature-pressure relationship for water freezing in s o i l s . On the b a s i s o f the detailed observations made in the earlier work it can then be concluded that ( 2 ) gives the b e s t available approximation o f the relationship between temperature and negative pore pressure (and thus e f f e c t i v e s t r e s s ) in unconfined samples.
APPLICABILITY OF EQUATION ( 2 )
There is an important limitation t o the present experiment in proving the range o f application o f ( 2 ) which remains t o be d i s c u s s e d . This i s illustrated b y Fig. 6 , where water content is shown a s a function o f e f f e c t i v e stress for samples in oedometer and suction t e s t s . So long a s the soil i s saturated, t h e s e t e s t s g i v e , with minor qualification, similar r e s u l t s . At 21% water c o n t e n t , the two s e t s o f r e s u l t s diverge. This i s the shrinkage l i m i t , and desaturation occurs in the suction t e s t s . In Fig. 4 t h i s i s reached in freezing to about - l ° C . O n freezing to lower temperatures the membrane experiments always gave water contents substantially higher than those determined calorimetrically. This i s probably because when desaturation o c c u r s , hydraulic flow is substantially reduced. Transference in the vapor phase may occur, but e v e n a f t e r three w e e k s at the low temperatures, water content remained higher i n the membrane experiment.
The information in Fig. 3 cannot be extrapolated to apply beyond the shrinkage limit (which i n the case o f Leda clay
S u c t i o n k g l c m 2 Fig. 5 . T h e o r e t i c a l a n d e x p e r i m e n t a l r e l a t i o n s h i p b e t w e e n temperature a n d s u c t i o n
40
*
+
observations
with ocdomctcr
oobservations in
suction tests
LEDA CLAY
KNB
PRESSURES O N THE ICE PHASE
0
F i g . 6 . Comparison of o b s e r v a t i o n s from s u c t i o n - w a t e r c o n t e n t t e s t s and from oedometer t e s t s
0 . 1
1
10
Agreement of e x p e r i m e n t a l o b s e r v a t i o n s w i t h (2) i m p l i e s t h a t the p r e s s u r e o n t h e i c e ( a t l e a s t where i t i s in c o n t a c t w i t h t h e water) i s a l w a y s a t m o s p h e r i c . M o s t of t h e i c e i s in b o d i e s c o n s i d e r a b l y larger t h a n pore s i z e , a n d might b e s u p - p o s e d to c a r r y the e f f e c t i v e s t r e s s , e s p e c i a l l y a t p o i n t s of c o n t a c t w i t h s o i l g r a i n s ( i c e and g r a i n s may b e s e p a r a t e d b y a bound film of w a t e r ) . The e x i s t e n c e in t h e i c e of a p o s i t i v e p r e s s u r e g r e a t e r t h a n a t m o s p h e r i c i s in c o n f l i c t with e v i d e n c e in F i g . 5 , e v e n w h e n a l l o w a n c e i s made for e x p e r i m e n t a l e r r o r s . C u r v a t u r e of very s m a l l r a d i i w i l l c a u s e i n c r e m e n t s o r d e c - r e m e n t s of p r e s s u r e l o c a l l y w i t h i n t h e i c e [ 9 , l o ] . I t i s s u g g e s t e d t h a t t h e i c e s u r f a c e w i l l b e c o n c a v e over g r a i n s a n d c o n v e x into p o r e s , with r a d i i s u c h t h a t l o c a l s t r e s s e s a r e r e l i e v e d a n d t h e i c e s u r f a c e in c o n t a c t with w a t e r i s uniformly a t a t m o s p h e r i c p r e s s u r e . Work now b e i n g c a r r i e d out i n v o l v e s f r e e z i n g t e s t s on simples under v a r i o u s e x t e r n a l l y a p p l i e d t o t a l - s t r e s s c o n d l ~ i o n s a n d may g i v e further information o n t h i s p o i n t .
CONCLUSIONS
Effective stress
6,
kg/cm2
Experiments involving f r e e z i n g of s o i l s a m p l e s , with r e s t r i c t e d i c e - l e n s development s h o w t h a t water w h i c h r e m a i n s unfrozen w i t h i n a f r e e z i n g s o i l h a s a n e g a t i v e p r e s s u r e . T h i s n e g a t i v e pore p r e s s u r e g i v e s r i s e t o a n e f f e c t i v e s t r e s s w h i c h c a u s e s c o n s o l i d a t i o n . T h i s volume d e c r e a s e w i l l u s u a l l y b e o b s c u r e d by i c e - l e n s g r o w t h .
N e g a t i v e pore p r e s s u r e i s g r e a t e r a t lower t e m p e r a t u r e s . Unfrozen w a t e r in s a t u r a t e d s o i l moves f r e e l y a l o n g pore p r e s - s u r e g r a d i e n t s . The e x p e r i m e n t s confirm !hat, for tempera- t u r e s down t o t h e l e v e l a t which the s h r i n k a g e limit i s r e a c h e d , a n e q u a t i o n of t h e t y p e proposed b y Schofield g i v e s a p p r o x i m a t e l y the s u c t i o n ( n e g a t i v e pore p r e s s u r e ) , in unconfined s a m p l e s , a s a f u n c t i o n of t e m p e r a t u r e .
The e f f e c t i v e s t r e s s d e v e l o p e d , e v e n a t t e m p e r a t u r e s of -0
.a0
t o -1. O°C, c a u s e s c o n s i d e r a b l e c o n s o l i d a t i o n in com- p r e s s i b l e s o i l s . T h i s , together w i t h d i s c o n t i n u i t i e s l e f t by i c e - l e n s e s , r e s u l t s in a s p e c i a l s t r u c t u r e in s o i l s s u b j e c t e d t o a f r e e z e - t h a w c y c l e .ACKNOWLEDGMENTS
I am i n d e b t e d to my c o l l e a g u e s a t the Norwegian G c o t e c h n i c a l I n s t i t u t e , J
.
Moum a n d 0 . Ihlen S o p p , and t o t h e D i r e c t o r , L. Bjerrum, for a s s i s t a n c e and s t i m u l a t i n g d i s c u s s i o n .REFERENCES
[ l ] 2 . A. N e r s e s s o v a , N . A. T s y t o v i c h . " M c r z l y e g r u n t y kok s l o z h n y e mnogofaznye s i s t e m y , " M a t . po laboratornym i s s l c d . merzlykh q r u n t o v , Akad. Nauk SSSR, I n s t . M c r z l o t . S b . 2 ,
1 9 5 7 , p p . 10-22.
[ 2 ] C . W . Lovell. "Temperature Effects on P h a s e C o m p o s i - tion and Strength of Partially-Frozen S o i l , " Hiqhway
Res.
Bd. Bull. N o . 1 6 8 , 1 9 5 7 , p p . 74-95.
[ 3 ] P . J . W i l l i a m s . " S p e c i f i c H e a t and Unfrozcn W a t e r C o n - tent of Frozen S o i l s , " P r o c . 1 s t C a n a d i a n C o n f . Permafrost, 1 9 6 3 , pp. 109-126.
[ 4 ] P. J . ' W i l l i a m s . "Unfrozen W a t e r C o n t e n t of Frozen S o i l s and S o i l Moisture S u c t i o n , " G e o t e c h n i q u e , S e p t . 1964. [ 5 ] L. A. R i c h a r d s . " P r e s s u r e Membrane A p p a r a t u s , C o n - s t r u c t i o n and U s e , " Aqr. E n q r . , Vol. 2 8 , 1 9 4 7 , p p . 451-460. [ 6 ] D . C r o n e y , J . D . C o l e m a n , P . M . Bridge. "Suction of M o i s t u r e Held in S o i l a n d Other Porous M a t e r i a l s , " Road R e s . T e c h . Paper 2 4 , 1952 (D.S . I . R . , R.R.L., Harmondsworth, M i d d l e s e x . )
.
[ 7 ] R. K . S c h o f i e l d . "The pF of the W a t e r in t h e S o i l , "
3rd
I n t l , C o n g . S o i l S c i . T r a n s . , Vol. 2 , 1 9 3 5 , p p . 37-48. [ 8 ] N. E. E d l e f s e n , A. B. C.
Anderson. "Thermodynamics of S o i l M o i s t u r e , " Hi!gardia, Vol. 1 5 , 1943, No. 2, p p . 119-120. [ 9 ] D. H . Everett. "The Thermodynamics of F r o s t Damage t o Porous S o l i d s , " T r a n s . F a r a d a y S o c . , Vol. 5 7 , 1 9 6 1 , P t . 9 , p p . 1541-1551.[ l o ] L. W . G o l d . "A P o s s i b l e Forcc M e c h a n i s m A s s o c i a t e d w i t h the Freezing of W a t e r in Porous M a t e r i a l s , " S i g h w a y R e s . Bd. Bull. No. 1 6 8 , 1 9 5 7 , p p . 65-73.
A. D i s c u s s i o n
R. 3. MILLER, C o r n e l l University-This i s a very i n t e r e s t i n g a n d s i g n i f i c a n t c o n t r i b u t i o n . W i l l i a m s adroitly a v o i d s t h e problems i n h e r e n t in the u s e of h i s s e c o n d e q u a t i o n , or s e e m s to ( c f . comments on paper by Lange and McKim)
,
b y k e e p i n g the i c e p h a s e o u t s i d e the s a m p l e . This m a n e u v e r , w h i c h a l l o w s the i c e - l e n s e s t o grow a t a t m o s p h e r i c p r e s s u r e , o s t e n - s i b l y f i x e s t h e p r e s s u r e o n the i c e p h a s e and e x c l u d e s i c e from the p o r e s where the p r e s s u r e in t h e i c e i s unknown when the w a t e r p r e s s u r e i s unknown. T h i s i s o n e of the f e w e x a m p l e s of u s e of the e q u a t i o n in a manner t h a t r e d u c e s the u n c e r t a i n t i e s of the boundary c o n d i t i o n s .I t i s i n t e r e s t i n g t o n o t e t h a t unfrozen water c o n t e n t v e r s u s temperature r e l a t i o n s h i p s o b s e r v e d under two e x p e r i m e n t a l c o n d i t i o n s d i v e r g e a t t h e s h r i n k a g e l i m i t , b u t a g r e e down t o t h a t p o i n t . T h i s s u g g e s t s t h a t w h e r e i t w a s p r e s e n t , the membrane prevented i c e e n t r y into t h e pore s y s t e m of t h e s a m p l e under c o n d i t i o n s where e n t r y o c c u r r e d in the a b s e n c e of the membrane. C o n s e q u e n t l y , s o m e w a t e r in t h e s a m p l e w i t h the membrane f a i l e d t o f r e e z e simply b e c a u s e n u c l e a t i o n of f r e e z i n g w a s b l o c k e d by t h e membrane and t h e unfrozen w a t e r c o n t e n t o b s e r v e d w a s g r e a t e r than t h a t o b s e r v e d w h e n t h e membrane w a s a b s e n t . T h i s e x p l a n a t i o n o b v i a t e s t h e n e e d for t h e e x p l a n a t i o n offered b y W i l l i a m s , b u t a t the s a m e time d o e s n o t i n v a l i d a t e h i s s u g g e s t i o n . For t h e a b o v e e x p l a n a t i o n t o b e v a l i d , i t i s n e c e s s a r y t o a s s u m e t h a t e i t h e r t h e membrane a l s o b l o c k e d a i r e n t r y or t h a t t h e s u r f a c e e n e r g y of t h e a i r - w a t e r i n t e r f a c e e x c e c d s t h a t of the i c e - w a t e r i n t e r - f a c e . O t h e r w i s e t h e w a t e r in q u e s t i o n would h a v e b e e n d i s - p l a c e d b y a i r i n s t e a d of i c e , where the l a t t e r w a s e x c l u d e d b y t h e membrane. Either or b o t h a r e p r o b a b l e .
B. CLOSURE-I am indebted t o Miller for drawing my a t t e n t i o n t o a n important p o i n t .
I s i t to b e e x p e c t e d t h a t the w a t e r c o n t e n t of t h e layer b e t w e e n t h e membranes w i l l b e e q u a l t o t h e (unfrozen) water
c o n t e n t of "normally f r o z e n " s o i l s ? The problem i s most e a s i l y u n d e r s t o o d b y c o n s i d e r i n g t h e e x t e n t t o w h i c h i c e in "normally frozen" s o i l i s l o c a l i z e d into a f e w r e l a t i v e l y large b o d i e s o r i s d i s p e r s e d through the p o r e s . As temperature i s
lowered i c e w i l l tend t o e n t e r s m a l l e r and s m a l l e r p o r e s . The s i z e of the p o r e s of the membrane i s s o s m a l l t h a t it i s q u i t e c e r t a i n t h a t i c e c a n n o t grow through them e x c e p t a t t e m p e r a - t u r e s far lower t h a n t h o s e of i n t e r e s t in t h e p r e s e n t c a s e . At some much h i g h e r temperature there w i l l b e a s i g n i f i c a n t number of p o r e s in t h e s o i l l a y e r b e t w e e n t h e membranes w h i c h would h a v e c o n t a i n e d i c e h a d the membranes n o t p r e v e n t e d t h i s . If t h e s e p a r t i c u l a r p o r e s a r e water-filled a t t h a t
t e m p e r a t u r e , then t h e w a t e r c o n t e n t of the l a y e r w i l l b e h i g h e r . Following Everett [ l ] the s i z e of pore's e n t e r e d by i c e a t a s u c t i o n , u i s g i v e n b y
w h e r e ,y. i s t h e i n t e r f a c i a l e n e r g y i c e / w a t e r . If u i s t h e s u c t i o n e ' 3 a b l i s h e d a t a g i v e n temperature ( i n a g r e e m e n t w i t h (2)) then p o r e s of larger r a d i u s than r , a r e i c e f i l l e d a t t h a t t e m p e r a t u r e . From t h e s u c t i o n - m o i s t u r e c o n t e n t curve (Fig. 6). it a p p e a r s t h a t a l a r g e number of p o r e s of Leda c l a y KNB a r e e m p t i e d a t a s u c t i o n of a b o u t 1 1 kg/ s q c m . The s i z e of p o r e s d r a i n e d a t a g i v e n s u c t i o n i s g i v e n b y a s i m i l a r e q u a t i o n
w h e r e (I,, i s t h e i n t e r f a c i a l e n e r g y a i r / w a t e r . Both ,yiw
and ,y a r e now q u i t e a c c u r a t e l y known [ Z ] . H e r e w e n e e d o n l y c g c s i d e r the r a t i o b e t w e e n them, t h a t i s , a p p r o x i m a t e l y 2 to 5 to s e e t h a t p o r e s e n t e r e d b y a i r a t a s u c t i o n o f 1 1 kg/sq cm might b e e n t e r e d b y i c e a t a l i t t l e over 4 kg/sq c m . From ( 2 ) , t h e l a t t e r s u c t i o n o c c u r s a t a b o u t -0.4'C. In the c a s e of the l a y e r b e t w e e n t h e membranes s u c h p o r e s w i l l b e w a t e r f i l l e d a t l e a s t u n t i l a s u c t i o n of
1 1 kg/sq cm i s r e a c h e d . The w a t e r c o n t e n t for t h i s layer might therefore b e e x p e c t e d t o d i v e r g e from c a l o r i m e t r i c a l l y d e t e r m i n e d w a t e r c o n t e n t s a t t h i s t e m p e r a t u r e . The r e l a t i v e l y i m p r e c i s e o b s e r v a t i o n s d o n o t r e v e a l t h i s ; i t i s a l s o o p e n to q u e s t i o n w h e t h e r t h e s e s i m p l e c o n s i d e r a t i o n s may b e a p p l i e d t o p o r e s of the s i z e found in c l a y s where much of t h e w a t e r i s bound t o the p a r t i c l e s and probably not s u s c e p t i b l e t o f r e e z i n g in t h e u s u a l s e n s e . In a n y c a s e , the p r e s s u r e in the w a t e r in t h e s o i l b e t w e e n t h e membranes a n d t h a t o u t s i d e them c a n b e a s s u m e d e q u a l a t t h e termination of e a c h t e s t . M e a s u r e m e n t s of w a t e r c o n t e n t of t h e inner l a y e r a l l o w determination of e f f e c t i v e s t r e s s d e v e l o p e d , a n d t h u s of s u c t i o n , a t l e a s t u n t i l the s h r i n k a g e limit i s r e a c h e d . The c o n s i d e r a t i o n s g i v e n a b o v e a r e r e l e v a n t t o e a r l i e r [ 3 ] e x p e r i m e n t a l e v i d e n c e ( F i g . 5 ) , t h a t (2) d e s c r i b e s t h e r e l a - t i o n s h i p b e t w e e n temperature and s u c t i o n in t h e w a t e r p h a s e of frozen s o i l s . T h e y s u g g e s t a n e x p l a n a t i o n for t h e t e n d e n c y of e x p e r i m e n t a l p o i n t s ( o b t a i n e d u s i n g c o n v e n t i o n a l s u c t i o n - m o i s t u r e c o n t e n t t e s t s ) t o l i e under t h e t h e o r e t i c a l c u r v e . The s i z e of t h e c o r r e c t i o n t h a t might b e made t o s u c h p o i n t s , o n a c c o u n t of t h e d i f f e r e n c e in i n t e r f a c i a l e n e r g i e s ,yiw and
-..
,yaw i s l a r g e l y beyond the s c o p e of the p r e s e n t d i s c u s s i o n . I t a p p e a r s t o b e s m a l l for t e m p e r a t u r e s a t which t h e r e i s l i t t l e e n t r y of i c e i n t o the p o r e s b u t might t h e o r e t i c a l l y involve a , , r e d u c t i o n b y t h r e e - f i f t h s of t h e s u c t i o n shown for c a s e s w h e r e i c e i s w i d e l y d i s p e r s e d through t h e p o r e s . I t i s m o s t r e l e v a n t w h e r e s o i l p o r e s a r e l a r g e , a n d where c o n s e q u e n t l y i c e e n t r y o c c u r s a t r e l a t i v e l y h i g h t e m p e r a t u r e s . Another p a p e r in t h i s v o l u m e , by M i l l e r , d i s c u s s e s t h e matter in further d e t a i l . REFERENCES
[ 1 1 D . H . E v e r e t t . "The Thermodynamics of F r o s t Damage t o Porous S o l i d s , " T r a n s . F a r a d . S o c . , Vol. 5 7 , 1 9 6 1 , Pt. 9 , pp. 1541-1551.
[ 2 1 E . H e s s t v e d t . The I n t e r f a c i a l Energy I c e / W a t e r , Norwe- g i a n G e o t e c h . I n s t . , P u b . No. 5 6 , 1 9 6 4 .
[ 3 ] P . J . W i l l i a m s . "Unfrozen W a t e r C o n t e n t of Frozen S o i l s a n d S o i l M o i s t u r e S u c t i o n , " GBotechnique, S e p t . 1 9 6 4 .