Knockout of the high-coupling cytochrome aa3 oxidase reduces TCA cycle fluxes in Bacillus subtilis
6
0
0
Texte intégral
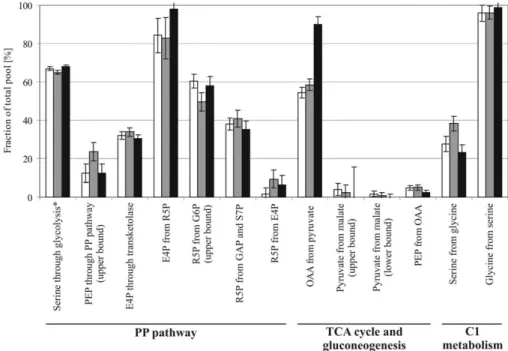
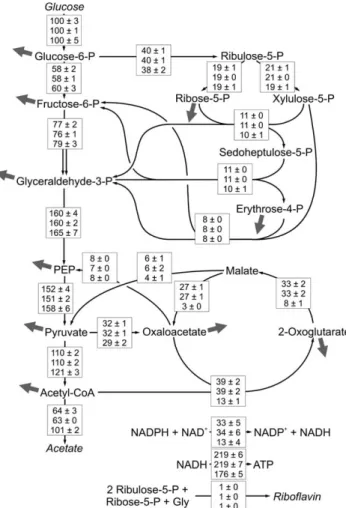
Figure

Documents relatifs