In-situ observations and modeling of nitric acid-containing particles in a cirrus cloud formation region
Texte intégral
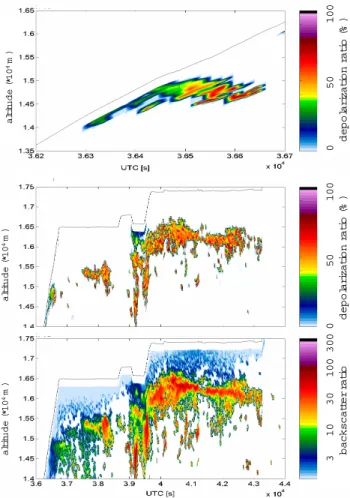
Figure



Documents relatifs
Title Page Abstract Introduction Conclusions References Tables Figures ◭ ◮ ◭ ◮ Back Close. Full Screen
Title Page Abstract Introduction Conclusions References Tables Figures J I J I Back Close.. Full Screen
Title Page Abstract Introduction Conclusions References Tables Figures ◭ ◮ ◭ ◮ Back Close.. Full Screen
Title Page Abstract Introduction Conclusions References Tables Figures ◭ ◮ ◭ ◮ Back Close.. Full Screen
It is followed by a slowly-developing, descending layer (the delayed.. Orsolini et al. Title Page Abstract Introduction Conclusions References Tables Figures ◭ ◮ ◭ ◮ Back Close
Title Page Abstract Introduction Conclusions References Tables Figures ◭ ◮ ◭ ◮ Back Close.. Full Screen
Kaminski Title Page Abstract Introduction Conclusions References Tables Figures ◭ ◮ ◭ ◮ Back Close.. Full Screen
Christopher Title Page Abstract Introduction Conclusions References Tables Figures ◭ ◮ ◭ ◮ Back Close.. Full Screen