Validation of a model to predict adverse outcomes in patients with pulmonary embolism
6
0
0
Texte intégral
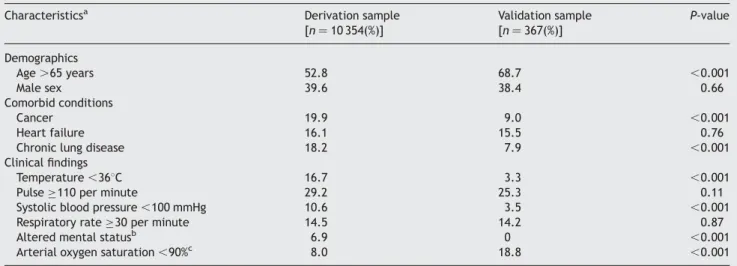
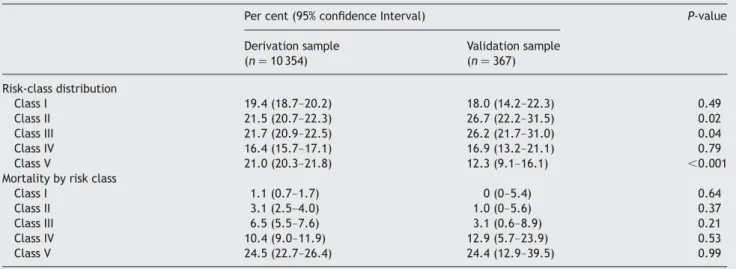
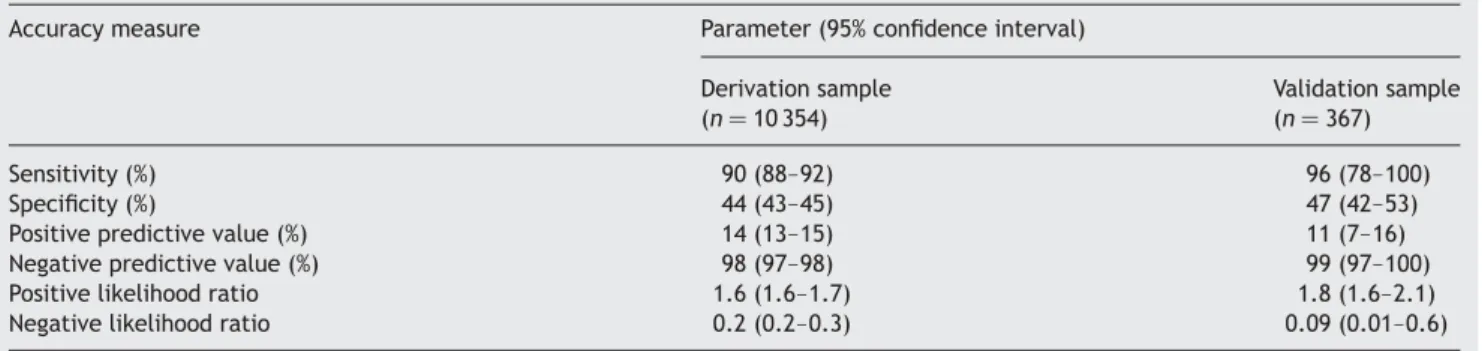
Figure
Documents relatifs