READ THESE TERMS AND CONDITIONS CAREFULLY BEFORE USING THIS WEBSITE. https://nrc-publications.canada.ca/eng/copyright
Vous avez des questions? Nous pouvons vous aider. Pour communiquer directement avec un auteur, consultez la première page de la revue dans laquelle son article a été publié afin de trouver ses coordonnées. Si vous n’arrivez pas à les repérer, communiquez avec nous à [email protected].
Questions? Contact the NRC Publications Archive team at
[email protected]. If you wish to email the authors directly, please see the first page of the publication for their contact information.
NRC Publications Archive
Archives des publications du CNRC
This publication could be one of several versions: author’s original, accepted manuscript or the publisher’s version. / La version de cette publication peut être l’une des suivantes : la version prépublication de l’auteur, la version acceptée du manuscrit ou la version de l’éditeur.
Access and use of this website and the material on it are subject to the Terms and Conditions set forth at
A Reference heat source for solar collector thermal testing
Harrison, S. J.; Bernier, M. A.
https://publications-cnrc.canada.ca/fra/droits
L’accès à ce site Web et l’utilisation de son contenu sont assujettis aux conditions présentées dans le site
LISEZ CES CONDITIONS ATTENTIVEMENT AVANT D’UTILISER CE SITE WEB.
NRC Publications Record / Notice d'Archives des publications de CNRC:
https://nrc-publications.canada.ca/eng/view/object/?id=7777b4f8-1b31-4db0-bf99-da1801f49ae8
https://publications-cnrc.canada.ca/fra/voir/objet/?id=7777b4f8-1b31-4db0-bf99-da1801f49ae8
Ser
TH1
0 .
1258
National Research
Conseil national
c.
2
BIJ3G
I
#
Council Canada
de recherches Canada
A REFERENCE HEAT SOURCE FOR SOLAR COLLECTOR THERMAL TESTING
by S.J. Harrison and M.A. Bernier
ANALYZED
Presented
at
ASME Winter Annual Meeting
New Orleans, LA, December, 1984
Solar Energy Division
Pamphlet Paper No. 84-WAISol-2,
7p.
DBR Paper No. 1258
Division of Building Research
Des
c a p t e u r s s o l a i r e s 3 l i q u i d e o n t
G t G
soumis
3
d e s e s s a i s au
moyen d'une s o u r c e chaude de r 6 f 6 r e n c e (SCR)
3
comparaison
d i r e c t e .
Un
r a d i a t e u r G l e c t r i q u e e s t r e l i g e n
sGrie au
c a p t e u r .
TAe f l i l i d e c a l o p o r t e u r a b s o r b e d e 1 ' C n e r g i e 3 un t a u x
mesurG p a r
l a SCR,
e t
l e t a u x de c a p t a g e d ' g n e r g i e s o l a i r e e s t
d G f i n i p a r
l e p r o d u i t de l 1 6 n e r g i e absorbGe e t l e r a p p o r t e n t r e
l e s G c a r t s de t e m p g r a t u r e d a n s l e c a p t e u r e t l e r a d i a t e u r .
La
SCR
e t
l e s
s o n d e s d e t e m p g r a t u r e a s s o c i ' e e s peuvent Gtre v u e s
comme un i n d i c a t e u r t h e r m i q u e de d g b i t massique,
l e p r o d u i t du
clgbit massique
e t d e l a c h a l e u r s p z c i f i q u e d t a n t mesurg
d i r e c t e m e n t dans l a b o u c l e d ' e s s a i .
D e s
e s s a i s d ' g t a l o n n a g e o n t 6 t 6 men& s u r deux SCR
3
d i f f G r e n t s
d g b i t s
e t
aux t e m p g r a t u r e s normalement o b s e r v g e s dans un e s s a i
de c a p t e u r p l a n ( d e -lO°C
3
-95OC).
On a g t a b l i d a n s chaque
c a s l e s v a l e u r s de
r3 s a v o i r l e ra,pport d e 1 ' Q n e r g i e
thermique (mesurge) t r a n s f G r 6 e p a r l a SCR a u c a l o p o r t e u r 3
l ' g n e r g i e G l e c t r i q u e consonunGe p a r l a SCR.
Les
r g s u l t a t s s o n t
pour l a SCR n O 1 :
nl-=99,61% ( & a r t - t y p e
de 0 , 2 2 ) ,
e t
pour l a
SCR n02:
n2=99,59% ( e c a r t - t y p e de 0 , 2 8 ) .
THEAHE
.
MECHAN. .
.
. .
.
,t be responr rnga ot tho 5 ssion is p r l n . I r,,hl,..-.ll-n f ST.. N O W K W K , N . T . ed ih p a m s of Printed iipubllcr in ASME Johr
Refem.,, ,,. ,=,,,,,, ,,~,.,,,,,,,,.
,,,;.
,,~,=..u,,,vl, , .,, b,ml,,L J ,,,t.,t. 09 given t o A: nhmlcat Oivlnlon, and the amenrs or opt'
its Dlvtslons
re paper is p
,...*-. r..,,-..
u t h o w l , Pam trs are availat
nlons aovanc or Sections. nhlIshM rn i - A , + ..L^..,rl * M in tl l f E mal. M E . nths
A Reference Heat Source for Solar Collector
Thermal Testing
S.
J. HARRISON
and M.
A. BERNIER
Division of Building Research
National Research Council of Canada
Ottawa, Ontario,
K I A
OR6
ABSTRACT
Liquid-based s o l a r c o l l e c t o r s have been t e s t e d u s i n g a d i r e c t - c o m p a r i s o n r e f e r e n c e h e a t s o u r c e (RHS). An e l e c t r i c h e a t e r i s i n s t a l l e d i n s e r i e s w i t h t h e s o l a r c o l l e c t o r u n d e r t e s t . Energy i s added t o t h e h e a t t r a n s f e r f l u i d a t a measured r a t e by t h e RHS, a n d t h e r a t e of s o l a r e n e r g y c o l l e c t i o n i s d e t e r m i n e d a s t h e p r o d u c t of t h i s e n e r g y i n p u t and t h e r a t i o of t h e d i f f e r e n t i a l t e m p e r a t u r e s a c r o s s t h e c o l l e c t o r a n d t h e h e a t e r . The RHS and a s s o c i a t e d t e m p e r a t u r e s e n s o r s c a n b e t h o u g h t o f a s a t h e r m a l mass f l o w m e t e r where, i n e f f e c t , t h e p r o d u c t of mass f l o w and s p e c i f i c h e a t i s measured d i r e c t l y i n t h e t e s t l o o p .
C a l i b r a t i o n t e s t s were performed on two r e f e r e n c e h e a t s o u r c e s a t v a r i o u s f l o w r a t e s a n d a t t h e t e m p e r a t u r e s n o r m a l l y e n c o u n t e r e d i n f l a t p l a t e s o l a r c o l l e c t o r t e s t i n g (-10°C t o "95OC). V a l u e s o f
n,
t h e r a t i o of t h e measured RHS t h e r m a l power o u t p u t t o t h e f l u i d t o t h e RHS e l e c t r i c a l power i n p u t w e r e d e t e r m i n e d f o r e a c h c a s e . R e s u l t s i n d i c a t e t h a t f o r RHSI1,n1
= 99.61% ( s t a n d a r d d e v i a t i o n of 0.22) and f o r RHS#2,n 2
= 99.59% ( s t a n d a r d d e v i a t i o n of 0.28). NOMENCLATURE A = a p e r t u r e a r e a of c o l l e c t o r (m2) C = s p e c i f i c h e a t of h e a t t r a n s f e r f l u i d (J/ (kg .K) ) G' = t o t a l s o l a r i r r a d i a n c e o n t h e p l a n e o f t h e s o l a r c o l l e c t o r (w/m2) & = mass f l o w r a t e o f h e a t t r a n s f e r f l u i d ( k g / s ) PRHS = i n p u t power t o RHS (W) qc = r a t e of e n e r g y c o l l e c t i o n by t h e s o l a r c o l l e c t o r (W)GRHS
= o u t p u t power of t h e RHS (W) Ta = t e m p e r a t u r e o f a m b i e n t a i r s u r r o u n d i n g t h e RHS (OC) Ti = t e m p e r a t u r e of f l u i d a t i n l e t t o h e a t e r i n t h e RHS (OC) To = t e m p e r a t u r e of f l u i d a t o u t l e t o f h e a t e r i n t h e RHS (OC) Tm = mean f l u i d t e m p e r a t u r e i n t h e RHS (OC)n
= s o l a r c o l l e c t o r e f f i c i e n c ynRHS
= t h e r m a l e f f i c i e n c y of t h e RHS Ap = p r e s s u r e d r o p between t e m p e r a t u r e w e l l s i n t h e RHS ( P a ) ATc = f l u i d t e m p e r a t u r e r i s e t h r o u g h s o l a r c o l l e c t o r (K) ATRHS = f l u i d t e m p e r a t u r e r i s e t h r o u g h r e f e r e n c e h e a t s o u r c e (K) T = measured t i m e i n t e r v a l ( s ) M = mass of f l u i d c o l l e c t e d d u r i n g t i m e i n t e r v a l 7 ( k g ) V = volume f l o w r a t e of h e a t t r a n s f e r f l u i d (m3/s) P = d e n s i t y of t h e h e a t t r a n s f e r f l u i d (kg/m3) S u b s c r i p t s c = r e l a t e d t o t h e c o l l e c t o r RHS = r e l a t e d t o t h e r e f e r e n c e h e a t s o u r c e 1. INTRODUCTION The t h e r m a l e f f i c i e n c y ( n ) of a s o l a r c o l l e c t o r , a s d e t e r m i n e d by t e s t i n g , i s g i v e n a s , where, q c = r a t e of e n e r g y c o l l e c t i o n by s o l a r c o l l e c t o r , = (fi*Cp -AT)c ASHRAE S t a n d a r d 93-77[L]
o u t l i n e s a f o r m a l i z e d s o l a r c o l l e c t o r t e s t i n g p r o c e d u r e and s p e c i f i e s t h e a c c u r a c y r e q u i r e m e n t s of a l l t h e measured t e r m s . I n p a r t i c u l a r , f o r c o l l e c t o r s t h a t u s e l i q u i d a s t h e h e a t t r a n s f e r f l u i d , t h e a c c u r a c y of t h e mass f l o w r a t e measurement s h o u l d b e e q u a l t o o r b e t t e r t h a n f l % of t h e measured v a l u e . It i s normal p r o c e d u r e t o measure t h e v o l u m e t r i c f l o w r a t e and d e r i v e mass f l o w r a t e f r o m p u b l i s h e d v a l u e s of f l u i d d e n s i t y , ( i . e . ,6
= V *p). L i q u i d f l o w m e t e r s c a p a b l e of a c h i e v i n g t h e a c c u r a c y r e q u i r e m e n t o f t h e s t a n d a r d a r e a v a i l a b l e[2,2].
T u r b i n e f l o w m e t e r s a r e among t h e most p o p u l a r , b u t a s n o t e d by Reed[i],
t h e i r a c c u r a c y i s s e n s i t i v e t o d i f f e r e n c e s i n f l o w p a t t e r n , g a s e n t r a i n m e n t i n t h eh e a t t r a n s f e r l i q u i d and wear of t h e b e a r i n g s . The a c c u r a t e d e t e r m i n a t i o n of q c by t h i s method a l s o depends on a knowledge of t h e t h e r m a l and p h y s i c a l p r o p e r t i e s of t h e h e a t t r a n s f e r f l u i d , p r i m a r i l y t h e f l u i d d e n s i t y and s p e c i f i c h e a t , over a r a n g e of t e m p e r a t u r e s . The t e m p e r a t u r e d e p e n d e n c i e s o f t h e s e p r o p e r t i e s a r e o f t e n n o t w e l l known f o r t h e commonly u s e d h e a t t r a n s f e r f l u i d s s u c h a s t h e w a t e r g l y c o l m i x t u r e s . Recognizing t h e s e d i f f i c u l t i e s , i t was d e c i d e d t o u s e t h e r e f e r e n c e h e a t s o u r c e (RHS) method t o d e t e r m i n e t h e r a t e of e n e r g y c o l l e c t i o n of t h e t e s t s o l a r c o l l e c t o r i n t h e D i v i s i o n of B u i l d i n g Research S o l a r C o l l e c t o r T e s t F a c i l i t y
[L].
The r e f e r e n c e h e a t s o u r c e method, a l s o c a l l e d t h e c a l o r i f i c r a t i o t e c h n i q u e by Reed and A l l e n[kl,
u t i l i z e s a n e l e c t r i c a l h e a t e r i n s t a l l e d i n s e r i e s w i t h t h e t e s t s o l a r c o l l e c t o r i n t h e c a l o r i m e t r y l o o p , a s shown i n F i g u r e 1. With t h e same mass f l o w r a t e t h r o u g h b o t h t e s t c o l l e c t o r and RHS, e l e c t r i c a l e n e r g y i s i n p u t t o t h e RHS a t a r a t e PRH,
and t h e r e s u l t a n t f l u i d t e m p e r a t u r e r i s e (ATRHS) a n 8 t h e c o r r e s p o n d i n g f l u i d t e m p e r a t u r e r i s e t h r o u g h t h e t e s t c o l l e c t o r (ATc) a r e measured. The r a t e of e n e r g y c o l l e c t i o n by t h e s o l a r c o l l e c t o r ( q c ) i s g i v e n b y , ROOFI
CONTROL VALVE REFERENCE V) HEAT SOURCEI
19
1 I
TEMPERATURF Fig. 1. DBRINRCC s o l a r c o l l e c t o r t e s t l o o p u s i n g t h e r e f e r e n c e h e a t s o u r c e method The t h e r m a l t r a n s f e r e f f i c i e n c y of t h e r e f e r e n c e h e a t s o u r c e (nRHS) i s d e f i n e d : RHS t h e r m a l power o u t p u t - q ~ H s ( 3 )"'
= RHS e l e c t r i c a l power i n p u t PRHS where, qRHS = r a t e of t h e r m a l e n e r g y o u t p u t t o h e a t t r a n s f e r f l u i d i n RHS = ( 6 . ~ ~ - AT)RHs.
I n most s o l a r c o l l e c t o r t e s t s , t h e d i f f e r e n c e i n mean f l u i d t e m p e r a t u r e between t h e RHS and c o l l e c t o r can be k e p t s u f f i c i e n t l y s m a l l s o t h a t t h e d i f f e r e n c e i n f l u i d s p e c i f i c h e a t c a n b e n e g l e c t e d . Good d e s i g n of t h e RHS can a s s u r e t h a twS
a p p r o a c h e s 1. The r a t e of e n e r g y c o l l e c t i o n by t h e s o l a r c o l l e c t o r c a n , t h e r e f o r e , b e approximated by Thus, t h e RHS method p e r m i t s d e t e r m i n a t i o n of t h e e n e r g y c o l l e c t i o n by t h e t e s t c o l l e c t o r w i t h o u t d i r e c t l y measuring t h e f l u i d f l o w r a t e and w i t h o u t knowing t h e t h e r m o p h y s i c a l p r o p e r t i e s of t h e h e a t t r a n s f e r f l u i d . T h i s p a p e r d e s c r i b e s t h e p o t e n t i a l s o u r c e s of e r r o r w i t h t h i s method, and t h e s p e c i f i c d e s i g n and c a l i b r a t i o n of t h e RHS used i n t h e DBRINRCC S o l a r C o l l e c t o r T e s t F a c i l i t y . 2. SOURCES OF ERROR To e n s u r e t h a t a c c u r a t e r e s u l t s a r e o b t a i n e d when u s i n g a r e f e r e n c e h e a t s o u r c e , c o n s i d e r a t i o n must b e g i v e n t o b o t h i t s d e s i g n and i t s o p e r a t i o n . Heat l o s s e s from t h e r e f e r e n c e h e a t s o u r c e r e s u l t i n an o v e r p r e d i c t i o n of t h e RHS power i n p u t t o t h e f l u i d and, a s a r e s u l t , a n o v e r p r e d i c t i o n o f t h e c o l l e c t o r performance. These l o s s e s c a n b e minimized by i n s u l a t i n g t h e RHS a n d by u s i n g a t h e r m a l g u a r d ( d e s c r i b e d i n S e c t i o n 3 ) w i t h i n t h e RHS.Temperature measurement e r r o r s c a n r e s u l t f r o m l o c a l b o i l i n g of t h e h e a t t r a n s f e r f l u i d on t h e RHS h e a t e r element. T h i s problem i s most l i k e l y t o o c c u r a t low d e g r e e s of s u b c o o l i n g * and low f l u i d f l o w r a t e s . Experiments h a v e been c o n d u c t e d
[ I ]
o n s u r f a c e b o i l i n g i n an a n n u l u s f l o w p a s s a g e s i m i l a r t o t h a t i n t h e DBRfNRCC RHS. The r e s u l t s of t h i s s t u d y c a n b e u s e d t o e s t i m a t e t h e minimum f l o w r a t e s and d e g r e e s of s u b c o o l i n g r e q u i r e d t o a v o i d s u r f a c e b o i l i n g . The maximum a l l o w a b l e h e a t e r power d e n s i t y i n c r e a s e s w i t h i n c r e a s i n g d e g r e e s of s u b c o o l i n g and w i t h i n c r e a s i n g h e a t e r - t o - f l u i d h e a t t r a n s f e r c o e f f i c i e n t . Thus t e m p e r a t u r e measurement e r r o r s due t o l o c a l b o i l i n g c a n-
*
d e g r e e s of s u b c o o l i n g a r e r e f e r r e d t o h e r e a s t h e d i f f e r e n c e ( i n K) between t h e f l u i d a v e r a g e t e m p e r a t u r e i n t h e RHS and t h e s a t u r a t i o n t e m p e r a t u r e a t t h e o p e r a t i n g p r e s s u r e . For example i f t h e f l u i d i s w a t e r and t h e a v e r a g e t e m p e r a t u r e and p r e s s u r e i n t h e RHS a r e 90°C and 108.15 kPa r e s p e c t i v e l y (which c o r r e s p o n d s t o a s a t u r a t i o n temp. of 101.85OC) t h e n t h e f l u i d h a s 11.85 K of s u b c o o l i n g .be a v o i d e d by c a r e f u l c o n s i d e r a t i o n of h e a t e r power d e n s i t y (power i n p u t ) , d e g r e e s of s u b c o o l i n g ( o p e r a t i n g t e m p e r a t u r e ) and h e a t t r a n s f e r c o e f f i c i e n t (f l o w r a t e ) .
E f f e c t i v e mixing d e v i c e s l o c a t e d ahead of t h e t e m p e r a t u r e w e l l s a r e a l s o e s s e n t i a l f o r good t e m p e r a t u r e measurement. These m i x e r s must r e m a i n e f f e c t i v e o v e r a w i d e r a n g e of f l o w r a t e s . As w e l l , f l u i d f r i c t i o n c a n i n c r e a s e t h e f l u i d t e m p e r a t u r e and c a u s e a n o v e r p r e d i c t i o n o f ATRHS; c o n s e q u e n t l y , t h e r e f e r e n c e h e a t e r s h o u l d b e d e s i g n e d t o minimize t h e p r e s s u r e d r o p between t h e t e m p e r a t u r e s e n s o r s . A major s o u r c e of e r r o r i n s o l a r c o l l e c t o r t e s t i n g i s non-steady t e s t i n g c o n d i t i o n s . The r e f e r e n c e h e a t s o u r c e i s s i m i l a r t o a c o l l e c t o r i n t h a t i t h a s t h e r m a l mass; t h u s i t c a n s t o r e and r e l e a s e energy d u r i n g ' t r a n s i e n t o p e r a t i o n . T h i s can r e s u l t i n an over- o r
,
u n d e r p r e d i c t i o n of c o l l e c t o r performance. For t h i s r e a s o n , t h e f l o w r a t e , i n l e t t e m p e r a t u r e , h e a t e r power, and s u b s e q u e n t t e m p e r a t u r e r i s e must b e h e l d c o n s t a n t d u r i n g e a c h t e s t p e r i o d . R e f e r e n c e[L]
o u t l i n e s r e q u i r e m e n t s f o r s t e a d y s t a t e t e s t i n g of s o l a r c o l l e c t o r s : t h e s e same r e q u i r e m e n t s a p p l y t o t h e r e f e r e n c e h e a t s o u r c e . I To minimize t h e e r r o r r e s u l t i n g from t h eI
assumption t h a t t h e f l u i d s p e c i f i c h e a t (C ) i s t h eI
same i n t h e h e a t e r and t h e c o l l e c t o r , t h e g e a n f l u i d t e m p e r a t u r e i n t h e RHS must b e k e p t c l o s e t o t h a t i n t h e s o l a r c o l l e c t o r . With w a t e r a s t h e h e a t t r a n s f e r1
f l u i d , t h i s e r r o r c a n u s u a l l y b e k e p t t o l e s s t h a n 0.25% f o r t h e t e m p e r a t u r e s n o r m a l l y e n c o u n t e r e d i n t e s t i n g . With g l y c o l s and o i l s , however, t h e v a l u e o fC i s s t r o n g l y dependent on t e m p e r a t u r e . It i s ,
i
t g e r e f o r e , s u g g e s t e d t h a t w i t h t h e s e h e a t t r a n s f e r f l u i d s , t h e i n l e t t e m p e r a t u r e and t e m p e r a t u r e r i s e f o r b o t h t h e RHS and t h e t e s t c o l l e c t o r b e k e p ta p p r o x i m a t e l y t h e same. T h i s can be accomplished by c o o l i n g t h e f l u i d between t h e RHS and t h e c o l l e c t o r , and by a d j u s t i n g t h e h e a t e r i n p u t t o make t h e
t e m p e r a t u r e r i s e n e a r l y e q u a l a c r o s s b o t h t h e RHS and t h e c o l l e c t o r . As shown by t h e example i n Table 1 , t h e e r r o r r e s u l t i n g from a l a r g e t e m p e r a t u r e d i f f e r e n t i a l between t h e RHS and t h e c o l l e c t o r i s q u i t e s i g n i f i c a n t ( ~ 2 % ) w i t h a g l y c o l / w a t e r m i x t u r e , b u t i n s i g n i f i c a n t ("0.1%) w i t h water. Although t h e example i s f o r a "worse c a s e " s i t u a t i o n , i t d o e s i n d i c a t e t h e need f o r a v o i d i n g l a r g e d i f f e r e n c e s i n t e m p e r a t u r e c o n d i t i o n s between RHS and c o l l e c t o r .
TABLE 1 EXAMPLE OF ERROR I N ASSUMING Cp,, = Cp, RHS C o l l e c t o r type: e v a c u a t e d t u b e , h i g h f l u i d r e s i d e n c e
t i m e
F l u i d : ( 5 0 % by volume e t h y l e n e g l y c o l / w a t e r m i x t u r e )
Theref o r e ,
For w a t e r , t h e c o r r e s p o n d i n g r a t i o would be:
To summarize: t h e RHS s h o u l d b e d e s i g n e d t o a c h i e v e t h e f o l l o w i n g :
1 ) n e g l i g i b l e o r d e f i n a b l e h e a t l o s s ,
2 ) low power d e n s i t y h e a t i n g e l e m e n t , and a h i g h h e a t t r a n s f e r c o e f f i c i e n t between t h e h e a t e r and t h e f l u i d t o a v o i d b o i l i n g , 3 ) good f l u i d m i x i n g u p s t r e a m of e a c h t e m p e r a t u r e measurement p o i n t t o e n s u r e a c c u r a t e f l u i d t e m p e r a t u r e measurements, 4 ) s m a l l p r e s s u r e d r o p between t e m p e r a t u r e w e l l s t o a v o i d f r i c t i o n a l h e a t i n g of t h e f l u i d . The d e s i r e d o p e r a t i n g c o n d i t i o n s f o r t h e RHS a r e : 1 ) s t e a d y o p e r a t i o n , i . e . , a c o n s t a n t f l o w r a t e , i n l e t t e m p e r a t u r e , and h e a t e r power i n p u t , 2 ) s u f f i c i e n t f l o w r a t e , o r low o u t l e t t e m p e r a t u r e , s o t h a t t h e f l u i d i n t h e RHS d o e s n o t b o i l , 3) t e m p e r a t u r e r i s e l a r g e enough t o minimize AT e r r o r s , 4 ) mean f l u i d t e m p e r a t u r e i n t h e RHS c l o s e t o t h a t i n t h e c o l l e c t o r t o minimize C e r r o r s . P
3. DESIGN OF THE DBR/NRCC REFERENCE HEAT SOURCE Because t h e DBRINRCC S o l a r C o l l e c t o r T e s t F a c i l i t y h a s t h e c a p a b i l i t y of t e s t i n g two c o l l e c t o r s
s i m u l t a n e o u s l y , i t h a s two s e p a r a t e c a l o r i m e t e r l o o p s , e a c h w i t h i t s own r e f e r e n c e h e a t s o u r c e . The two ( r e f e r r e d t o a s RHSiIl and RHSiI2) a r e i d e n t i c a l e x c e p t f o r minor d i f f e r e n c e s i n t h e t h e r m o p i l e d e s i g n . The r e f e r e n c e h e a t s o u r c e d e s i g n e d a t NRCC i s shown i n F i g u r e 2 . It c o n s i s t s of a 7 5 0 q a t t e l e c t r i c immersion h e a t e r e l e m e n t e n c l o s e d i n a s e c t i o n of c o p p e r p i p e . T h i s r e p r e s e n t s a maximm power d e n s i t y of a p p r o x i m a t e l y 49 kw/m2.
The RHS was d e s i g n e d t o b e compact t o f a c i l i t a t e t h e i n c o r p o r a t i o n of a t h e r m a l guard r i n g . The t h e r m a l g u a r d i s i n t e n d e d t o l i m i t t h e h e a t l o s s e s ( o r g a i n s ) between t h e i n t e r n a l components and t h e s u r r o u n d i n g s . G l a s s f i b r e i n s u l a t i o n i s u s e d t o i n s u l a t e t h e i n t e r n a l components from e a c h o t h e r and from t h e g u a r d r i n g . The t h e r m a l g u a r d c o n s i s t s of a c o i l e d c o p p e r t u b e s o l d e r e d t o a copper s h e e t . The f l u i d i s c i r c u l a t e d t h r o u g h t h e g u a r d b e f o r e e n t e r i n g t h e h e a t e r s e c t i o n o f t h e RHS. T h i s c o n f i g u r a t i o n s i g n i f i c a n t l y r e d u c e s t h e t e m p e r a t u r e d i f f e r e n c e and r e s u l t a n t h e a t l o s s between t h e h e a t e r ' s i n t e r n a l components and i t s immediate s u r r o u n d i n g s . The g u a r d r i n g t e m p e r a t u r e f o l l o w s t h e i n l e t f l u i d t e m p e r a t u r e w i t h o u t t h e need f o r a c o n t r o l l e r e v e n when t h e h e a t t r a n s f e r f l u i d i s c o o l e r t h a n t h e a i r a d j a c e n t t o t h e RHS. The e x t e r i o r of t h e t h e r m a l g u a r d r i n g i s i n s u l a t e d t o minimize h e a t l o s s e s and l i m i t t h e t e m p e r a t u r e g r a d i e n t a l o n g i t s f l o w l e n g t h . An a d v a n t a g e of t h e f l u i d g u a r d r i n g o v e r a n e l e c t r i c guard r i n g , i n a d d i t i o n t o i t s c a p a b i l i t y t o o p e r a t e below t h e a m b i e n t t e m p e r a t u r e , i s t h a t i t d o e s n o t r e q u i r e a c o n t r o l c i r c u i t which c o u l d i n t r o d u c e " n o i s e " i n t o t h e t e m p e r a t u r e measurement.
A mixing d e v i c e and a t e m p e r a t u r e measurement w e l l a r e l o c a t e d a t b o t h t h e i n l e t and t h e o u t l e t of t h e h e a t e r . To a c h i e v e h i g h r e s o l u t i o n and r e l i a b i l i t y , t y p e "T" ( c o p p e r c o n s t a n t a n ) d i f f e r e n t i a l t h e r m o p i l e s a r e u s e d t o measure t h e f l u i d t e m p e r a t u r e r i s e a c r o s s t h e h e a t e r e l e m e n t s . (RHSdk1 h a s 1 0 j u n c t i o n s and RHSdI2 h a s 8.) The t h e r m o p i l e s were c a l i b r a t e d by t h e D i v i s i o n of P h y s i c s o f t h e NRCC, i n t e r m s o f t h e I n t e r n a t i o n a l P r a c t i c a l Temperature S c a l e of 1968 (IPTS-68). The e l e c t r i c a l power i n p u t (PRHS) i s
measured w i t h a c a l i b r a t e d e l e c t r o n i c power t r a n s d u c e r (20.25% of f u l l s c a l e ) .
The f l u i d p i p e s l e a d i n g t o and f r o m t h e RHS a r e i n s u l a t e d t o l i m i t a x i a l t h e r m a l c o n d u c t i o n , a s a r e t h e w i r e s g o i n g t o and f r o m t h e t h e r m o p i l e and h e a t e r . The
T O P
V I E W
-.
, ,\ ,THERMAL GUARD RING
(COILED COPPER TUBE SOLDERED TO COPPER SHEET)
BLOWN GLASS FIBRE
-<=-'
1 . INSULATIONd
FLOWEXIT
+
t
FLOW ENTRANCETEMPERATURE WELL
THERMAL GUARD RING
HEATER
16 mrn DIA. x 300 mm LG 750 WATT
FLOW FROM GUARD
4.1 T e s t P r o c e d u r e
A s e r i e s of c a l i b r a t i o n t e s t s was ~ e r f o r m e d t o d e p t h of immersion of t h e t e m p e r a t u r e w e l l s i s
a p p r o x i m a t e l y 30 d i a m e t e r s , which e x c e e d s t h e recommendations of t h e ASHRAE S t a n d a r d 41.1-74: S e c t i o n on Temperature Measurements
[i].
A s mentioned e a r l i e r , a s t a b l e i n l e t t e m p e r a t u r e , good f l o w r a t e c o n t r o l and r e g u l a t i o n o f t h e power s u p p l i e d t o t h e RHS a r e a l l e s s e n t i a l t o s t e a d y - s t a t e o p e r a t i o n of t h e RHS. The DBRINRCC s o l a r c o l l e c t o r t e s t l o o p u s e s a p r e s s u r e - s e n s i t i v e c o n t r o l v a l v e t o m a i n t a i n t h e f l o w r a t e t o f0.1% of t h e s e t p o i n t . The s t a b i l i t y of t h e i n l e t t e m p e r a t u r e , u s i n g a w e l l - m i x e d t e m p e r a t u r e b a t h u p s t r e a m of t h e RHS, was 3.1 K d u r i n g a t e s t p e r i o d . H e a t e r power f l u c t u a t i o n s a r e reduced t o -f0.1% w i t h t h e u s e of a v o l t a g e r e g u l a t o r . d e t e r m i n e t h e e f f e c t on
nRHS,
o f ;-
t e m p e r a t u r e r i s e t h r o u g h h e a t e r ,-
t e m p e r a t u r e d i f f e r e n c e between h e a t e r f l u i d a n d s u r r o u n d i n g a i r ,-
f l u i d f l o w r a t e . I n o r d e r t o p e r f o r m t h e c a l i b r a t i o n t e s t s , t h e upper p o r t i o n of t h e c o l l e c t o r t e s t f a c i l i t y was m o d i f i e d a s shown i n F i g u r e 3, w i t h a dump t a n k and a measuring s c a l e i n p l a c e of t h e s o l a r c o l l e c t o r . T h i s p e r m i t t e d a n i n d e p e n d e n t d e t e r m i n a t i o n of a v e r a g e mass f l o w r a t e , by measuring t h e t o t a l mass of f l u i d f l o w i n g t h r o u g h t h e r e f e r e n c e h e a t e r o v e r a p r e c i s e l y measured time i n t e r v a l . For t h e c a l i b r a t i o n t e s t s , t h e v a l u e of f l u i d s p e c i f i c h e a t (C ) was d e t e r m i n e d a t t h e mean PTABLE 2 SAMPLE DATA FROM CALIBRATION TEST
RHS 11 1
Run Number 5 7
Average Temperature Measurements i n RHS
I n l e t f l u i d t e m p e r a t u r e (Ti) 44.70°C
O u t l e t f l u i d t e m p e r a t u r e (To) 53.08OC
-
1Tm --• (Ti
+
To) 48.89OC2
AT,, 8.39 K
Ambient t e m p e r a t u r e (Ta) 27.0°C
Tm- Ta 21.9 K
WELL-MIXED
TEMPERATURE BATH G r a v i m e t r i c Flow R a t e CONTROL
I VALVE
-
-
- - Net weight 9.26 kg-
-
-
- -
-
Time p e r i o d 599.9 s Mass f l o w r a t e - Volume f l o w r a t e 0.016 k g / s 0.98 Llmin VI PUMP REFERENCE 3 aI
1r PowerHEAT SOURCE
&
-
r l h d TEMPERATURE P~~~ 568.1 w a t t s CONTROL LOOP qRHS 566.6 w a t t s a T ~ ~ ~ nRHs 99.7% Fig. 3. C a l i b r a t i o n set-up A t h i g h f l u i d t e m p e r a t u r e s , e v a p o r a t i o n from t h e f l u i d t e m p e r a t u r e i n t h e h e a t e r , u s i n g v a l u e s t a k e n weighing t a n k i n t r o d u c e s a measurement e r r o r ,
from r e f e r e n c e
[?I.
e s p e c i a l l y a t low f l o w r a t e s when t h e weighing p e r i o dA c a l i b r a t i o n t e s t was performed a s f o l l o w s . The i s long. To r e d u c e t h i s e r r o r due t o e v a p o r a t i o n
h e a t t r a n s f e r f l u i d was drawn from a w e l l - m i x e d l o s s e s , t h e f l u i d was cooled b e f o r e e n t e r i n g t h e
t e m p e r a t u r e c o n t r o l l e d b a t h , pumped through t h e h e a t e r , weighing tank and a 'Over was used O n t h e
cooled and t h e n r e t u r n e d t o t h e bath. Flowrate was tank.
m a i n t a i n e d c o n s t a n t w i t h a p r e s s u r e s e n s i t i v e c o n t r o l A s e r i e s of t e s t s were performed t o d e t e r m i n e t h e
v a l v e . The f l u i d t e m p e r a t u r e r i s e through t h e h e a t e r p r e s s u r e drop c h a r a c t e r i s t i c of t h e r e f e r e n c e h e a t
and t h e i n l e t f l u i d t e m p e r a t u r e were c o n t i n o u s l y s o u r c e , a s s i g n i f i c a n t f l u i d f r i c t i o n h e a t i n g c o u l d
monitored on a s t r i p c h a r t r e c o r d e r t o d e t e c t any i n t r o d u c e e r r o r s . The p r e s s u r e d r o p between t h e r m o p i l e
t r a n s i e n t o r abnormal c o n d i t i o n s , and t o i n d i c a t e when w e l l s was measured a t s e v e r a l f l o w r a t e s from 0.008 t o
s t e a d y c o n d i t i o n s were a c h i e v e d ( s e e F i g u r e 4 f o r an 0.125 k g / s .
example of t h e t e m p e r a t u r e c o n d i t i o n s d u r i n g a t e s t To d e t e r m i n e t h e t h e r m a l r e s p o n s e of t h e RHS t o
r u n ) . When s t e a d y c o n d i t i o n s were achieved, i.e. when variations in power the tests were
t h e i n l e t t e m p e r a t u r e d i d n o t v a r y by more t h a n 3 . 1 K performed a t v a r i o u s f l u i d f l o w r a t e s . For e a c h
and t h e t e m p e r a t u r e r i s e a c r o s s t h e RHS, by more t h a n f l o w r a t e t h e RHS was allowed t o s t a b i l i z e and t h e v a l u e
k0.05 K, f o r a p e r i o d g r e a t e r t h a n t h e t e s t t i m e of ATRHS measured. The h e a t e r power i n p u t t o t h e RHS
i n t e r v a l (approx. 3 t o 1 5 min.), t h e f l o w was d i v e r t e d was t h e n s w i t c h e d o f f , w h i l e t h e i n l e t f l u i d
t o a weighing s c a l e and an a v e r a g e mass flow t e m p e r a t u r e and mass f l o w r a t e were m a i n t a i n e d c o n s t a n t .
measurement o b t a i n e d . T a b l e 2 g i v e s t h e r e s u l t s of a The s u b s e q u e n t decay of t h e v a l u e of ATRHS was r e c o r d e d
t y p i c a l t e s t run. u n t i l i t was e f f e c t i v e l y z e r o .
4.2 T e s t R e s u l t s
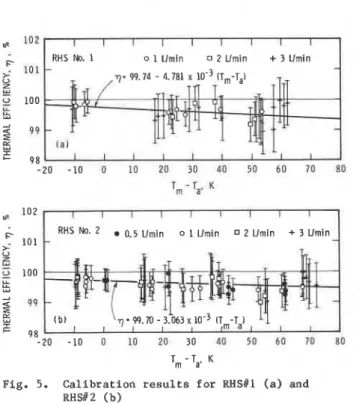
F i g u r e s 5a and 5b show c a l i b r a t i o n r e s u l t s f o r two
END i d e n t i c a l RHS's w i t h w a t e r a s t h e h e a t t r a n s f e r f l u i d .
The t h e r m a l e f f i c i e n c y (%HS) i s p l o t t e d a g a i n s t t h e
d i f f e r e n c e between t h e mean f l u i d t e m p e r a t u r e i n t h e RHS and t h e s u r r o u n d i n g ambient a i r t e m p e r a t u r e
(Tm
-
Ta) f o r d i f f e r e n t f l o w r a t e s . The magnitude of It h e v e r t i c a l b a r s r e p r e s e n t s t h e u n c e r t a i n t y a s s o c i a t e d
w i t h each d a t a p o i n t (Appendix A). For a f l o w r a t e
between 0.008 k g / s and 0.050 k g / s (0.5 t o 3 L/min.), t h e v a l u e s of measured RHS e f f i c i e n c y a r e s l i g h t l y l e s s t h a n 100% f o r t h e normal r a n g e of t e s t t e m p e r a t u r e s . I f a " l e a s t s q u a r e s " c u r v e f i t i s a p p l i e d t o t h e d a t a , t h e v a l u e of e f f i c i e n c y i s s e e n t o d e c r e a s e v e r y s l i g h t l y w i t h i n c r e a s i n g v a l u e s of (T,
-
T ). START I f t h e dependence of qRHS on (T,-
T,B
i s n e g l e c t e d , t h e v a l u e of t h e r m a l e f f i c i e n c y (nRHS) o b t a i n e d f o r RHStl i s 99.61% w i t h a s t a n d a r d d e v i a t i o nPig. 4. V a r i a t i o n of t h e t e m p e r a t u r e c o n d i t i o n s d u r i n g Of 0'22 and f o r RHS#Zs %HS = 99'59% with a standard
a t y p i c a l t e s t r u n d e v i a t i o n of 0.28. Thus t h e requirement f o r 1%
,
102 I I I I I 1 1 1 I.-..
RHS No. 1 o 1 Umin o 2 Umin+
3 Umin1
Fig. 5. C a l i b r a t i o n r e s u l t s f o r RHSill ( a ) and RHSl2 ( b )
accuracy on t h e measurement of mass f l o w r a t e o v e r t h e t e s t r a n g e a s r e q u i r e d by ASHRAE
[L]
i s surpassed. F i g u r e 6 p r e s e n t s t h e same d a t a ( f o rRHS82
o n l y ) p l o t t e d a g a i n s t t h e t e m p e r a t u r e r i s e through t h e h e a t e r (ATRHs). A c u r v e f i t t o t h e d a t a shows t h a t t h e e f f i c i e n c y i s e s s e n t i a l l y independent of t h e t e m p e r a t u r e r i s e through t h e RHS. Although t h e l o s s e s from t h e RHS s h o u l d b e z e r o when Tm = Ta ( i . e . , qRH = l o o % ) , t h e r e s u l t s i n F i g u r e 5a and 5b show t g a t t h i s i s n o t t h e c a s e . The r e s i d u a l e r r o r shown c o u l d be t h e r e s u l t of a s y s t e m a t i c e r r o r i n h e r e n t i n t h e d e s i g n of t h e RHS. It could a l s o be due t o t h e t a b u l a t e d v a l u e s of C u s e d i n P t h i s c a l i b r a t i o n , which may n o t a d e q u a t e l y a c c o u n t f o r any d i s s o l v e d a i r o r m i n e r a l s p r e s e n t i n t h e t e s t f l u i d . F i g u r e 7 shows t h e p r e s s u r e d r o p c h a r a c t e r i s t i c of t h e RHS w i t h w a t e r a s t h e h e a t t r a n s f e r f l u i d . With a flow of 0.10 k g / s , a f l o w r a t e h i g h e r t h a n t h a t n o r m a l l y encountered i n s o l a r c o l l e c t o r t e s t i n g , t h e p r e s s u r e d r o p between w e l l s was a p p r o x i m a t e l y 350 Pa, which c o r r e s p o n d s t o a p p r o x i m a t e l y 0.035 w a t t s of f r i c t i o n a l h e a t i n g . T h i s amount i s two o r d e r s of magnitude"
100.5 RHS No. 2 7) = 99.62-
7.466 x ( a T R H S ) Z Y U 9 9 . 5 LL Y w.
H E A T E R T E M P E R A T U R E R I S E .AT^^^,
K Fig. 6. Thermal e f f i c i e n c y v s t e m p e r a t u r e r i s e (RHS#Z) F L O W R A T E , k g l s Fig. 7. P r e s s u r e d r o p between t h e r m o p i l e w e l l s i n RHS TIME I N T E R V A L ,r ,
sI
Fig. 8. Thermal r e s p o n s e of RHS t o a s t e p change i n h e a t e r power l e v e l s m a l l e r t h a n t h e a p p a r e n t h e a t l o s s from t h e RHS and i s , t h e r e f o r e , n e g l i g i b l e . The t r a n s i e n t r e s p o n s e o f t h e r e f e r e n c e h e a t s o u r c e was determined by s h u t t i n g t h e h e a t e r o f f d u r i n g s t e a d y - s t a t e o p e r a t i o n and r e c o r d i n g t h e t i m e d e c a y of t h e d i f f e r e n t i a l t e m p e r a t u r e measurement. R e s u l t s a r e shown i n F i g u r e 8 a s v a l u e s of t e m p e r a t u r e r i s e ATRHS a t time i n t e r v a l 7, (normalized t o ATms a t 7 = 0) v s 7. The r e s u l t s i n d i c a t e t h a t t h e r e s p o n s e t i m e of t h e RHS i s dependent on t h e f l u i d f l o w r a t e . The r e s p o n s e of ATRHS t o a s t e p v a r i a t i o n i n h e a t e r power
i s e f f e c t i v e l y e x p o n e n t i a l , preceded by a s m a l l t i m e l a g which i n c r e a s e s a t lower f l o w r a t e s . To avoid e r r o r s i n t r o d u c e d by t h e t h e r m a l i n e r t i a of t h e RHS, t h e d e v i c e s h o u l d be o p e r a t e d under s t e a d y s t a t e c o n d i t i o n s .
5. CONCLUSIONS I
A major advantage of t h e RHS i s i t s c a p a b i l i t y t o measure t h e p r o d u c t of mass f l o w and s p e c i f i c h e a t d i r e c t l y i n t h e t e s t l o o p d u r i n g c o l l e c t o r t e s t i n g . T h i s a v o i d s e r r o r s t h a t c o u l d b e i n t r o d u c e d t h r o u g h t h e u s e of mechanical f l o w m e t e r s , which r e q u i r e an a c c u r a t e knowledge of t h e t e m p e r a t u r e dependence o f t h e t h e r m a l and p h y s i c a l p r o p e r t i e s of t h e h e a t t r a n s f e r f l u i d b e i n g used. To a c h i e v e s a t i s f a c t o r y r e s u l t s w i t h a n RHS, however, c a r e must be e x e r c i s e d i n i t s d e s i g n and s t e a d y - s t a t e o p e r a t i o n i s e s s e n t i a l ; a s t a b l e i n l e t t e m p e r a t u r e , good f l o w r a t e c o n t r o l and r e g u l a t i o n o f t h e power s u p p l i e d t o t h e h e a t e r , a r e r e q u i r e d .
C a l i b r a t i o n t e s t s w e r e performed on t h e NRCC r e f e r e n c e h e a t s o u r c e o v e r a r a n g e of f l o w r a t e s and i n l e t t e m p e r a t u r e s n o r m a l l y e n c o u n t e r e d d u r i n g t h e t e s t i n g of l i q u i d c o o l e d s o l a r c o l l e c t o r s , ( i . e . ,
&
from 0.008 k g / s t o 0.05 k g / s and Ti from t o 95OC). When u s i n g t h e RHS o u t s i d e t h e s e o p e r a t i n g c o n d i t i o n s , c a u t i o n must b e e x e r c i s e d . At low f l o w r a t e s (c0.008 k g / s ) , l o c a l i z e d b o i l i n g may i n t r o d u c e e r r o r s i f t h e h e a t e r power d e n s i t y i s n o t reduced a s w e l l . O p e r a t i o n a t f l o w r a t e s g r e a t e r t h a n 0.05 k g / s r e d u c e s t h e t e m p e r a t u r e r i s e a c r o s s t h e RHS, i n c r e a s i n g t e m p e r a t u r e measurement u n c e r t a i n t y . R e s u l t s i n d i c a t e t h a t t h e p r e s e n t RHS e x c e e d s t h e ASHRAE t e s t method r e q u i r e m e n t[L]
of 1% a c c u r a c y i n t h e d e t e r m i n a t i o n of mass f l o w r a t e . ACKNOWLEDGEMENT The a u t h o r s wish t o e x p r e s s t h e i r a p p r e c i a t i o n t o L.P. Chabot f o r h i s a s s i s t a n c e . T h i s p a p e r i s a c o n t r i b u t i o n from t h e D i v i s i o n of B u i l d i n g R e s e a r c h , N a t i o n a l R e s e a r c h C o u n c i l of Canada, and i s p u b l i s h e d w i t h t h e a p p r o v a l of t h e D i r e c t o r of t h e D i v i s i o n . REFERENCESMethods of T e s t i n g t o Determine t h e Thermal
P e r f o r m a n c e of S o l a r C o l l e c t o r s . American S o c i e t y f o r H e a t i n g , R e f r i g e r a t i n g and Air-Conditioning E n g i n e e r s , ASHRAE S t a n d a r d 93-77, 1977.
H a l l , J . , Choosing a Flow Monitoring Device, I n s t r u m e n t s and C o n t r o l Systems, J u n e 1981, p. 51-59.
P l a c h e , K.O., C o r i o L i s / G y r o s c o p i c Flow Meter, Mechanical E n g i n e e r i n g , March 1979, p. 36-41. Reed, K.A., I n s t r u m e n t a t i o n f o r t h e r m a l performance measurements: s t r i v i n g f o r measurement a s s u r a n c e i n s o l a r c o l l e c t o r t e s t i n g , ASME S o l a r Energy Conference, Albuquerque 1982, p. 337-340. H a r r i s o n , S.J. and S a s a k i , J.R., S t u d i e s on S o l a r C o l l e c t o r Performance a t NRC, Renewable A l t e r n a t i v e s , P r o c e e d i n g s of t h e F o u r t h Annual Conference of t h e S o l a r Energy S o c i e t y of Canada, August 1978, London, O n t a r i o , Canada.
Reed, K.A. and A l l e n , J.W., S o l a r Thermal C o l l e c t o r T e s t i n g : C a l o r i f i c R a t i o Technique, S o l a r
D i v e r s i f i c a t i o n , P r o c e e d i n g s of t h e I n t e r n a t i o n a l Energy S o c i e t y , I n c . , August 1978, Denver, CO, USA.
McAdams, W.H., e t a l . , H e a t T r a n s f e r a t High R a t e s t o Water With S u r f a c e B o i l i n g , I n d u s t r i a l and E n g i n e e r i n g C h e m i s t r y , September 1949, p. 1945. S t a n d a r d Measurement Guide: S e c t i o n on t e m p e r a t u r e measurements, American S o c i e t y f o r Heating,
R e f r i g e r a t i n g and Air-Conditioning E n g i n e e r s , ASHRAE S t a n d a r d 41.1-74, 1974.
Weast, R.C., ed.. Handbook of Chemistry and P h y s i c s , Forty-Ninth E d i t i o n , The Chemical Rubber Co., C l e v e l a n d , OH, 1968.
K l i n e , S.J. and McClintock, F.A., D e s c r i b i n g U n c e r t a i n t i e s i n Single-Sample Experiments, Mechanical E n g i n e e r i n g , J a n u a r y 1953.
APPENDIX A A n a l y s i s o f t h e Random Measurement U n c e r t a i n t y on nRHS
The u n c e r t a i n t y a s s o c i a t e d w i t h nRHS c a n b e e v a l u a t e d u s i n g t h e p r o p a g a t i o n of random u n c e r t a i n t y t e c h n i q u e s
[El.
U s i n g e q u a t i o n ( 3 ) we have:
The mass f l o w r a t e (ni) i s d e t e r m i n e d by w e i g h i n g a mass of f l u i d (M) f l o w i n g t h r o u g h t h e RHS o v e r a p r e c i s e l y measured time i n t e r v a l ( T ) s o Eq. ( 5 ) becomes,
Performing an a n a l y s i s a s d e s c r i b e d i n Ref. 1 0 t h e u n c e r t a i n t y i n t h e d e p e n d e n t v a r i a b l e (W llauc ~ k a n b e e x p r e s s e d i n t e r m s of t h e u n c e r t a i n t i e s in""e"ach of t h e i n d e p e n d e n t v a r i a b l e s (WM, W T , Wcp, VAT, WPRHS). where, WM = i O . O l k g W~ P +0.1% of C P