Considering the degradation effects of amino-functional plasma polymer coatings for biomedical application
6
0
0
Texte intégral
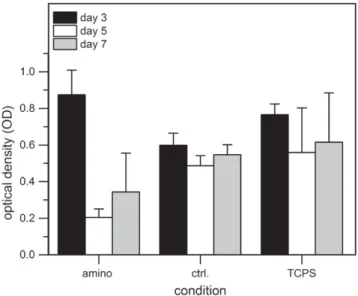
Figure


Documents relatifs