HAL Id: hal-02433885
https://hal.archives-ouvertes.fr/hal-02433885
Submitted on 9 Jan 2020HAL is a multi-disciplinary open access archive for the deposit and dissemination of sci-entific research documents, whether they are pub-lished or not. The documents may come from teaching and research institutions in France or abroad, or from public or private research centers.
L’archive ouverte pluridisciplinaire HAL, est destinée au dépôt et à la diffusion de documents scientifiques de niveau recherche, publiés ou non, émanant des établissements d’enseignement et de recherche français ou étrangers, des laboratoires publics ou privés.
Modelling of precipitation in a continuous fluidized bed
reactor with mechanical stirring
M. Bertrand, La. Mojica, H. Muhr, E. Plasari, F. Auger
To cite this version:
M. Bertrand, La. Mojica, H. Muhr, E. Plasari, F. Auger. Modelling of precipitation in a continuous fluidized bed reactor with mechanical stirring. ISIC 20, Sep 2017, Dublin, Ireland. �hal-02433885�
Modelling of precipitation in a continuous fluidized bed reactor with mechanical
stirring
M. Bertrand1, LA. Mojica1, H. Muhr2, E. Plasari2, F. Auger3
1CEA, Nuclear Energy Division, Research Department of Mining and Fuel Recycling Processes
B.P. 17171, 30207 Bagnols-sur-Cèze, France.
2National Highest School of Chemical Industries, Reactions and Process Engineering Laboratory, University of
Lorraine, CNRS, 1 rue Grandville, BP 20451, 54001 Nancy, France
3Areva Mines/SEPA, 87250 Bessines-sur-Gartempe, France
Keywords: Precipitation, Fluidized bed reactor, Modelling Introduction
Modelling of precipitation reactions requires the knowledge of flow pattern inside the reactor, the thermodynamic properties as well as the kinetic laws of the different mechanisms (nucleation, crystal growth, agglomeration…). Thanks to this information, population balance can be solved in support of design and operating of industrial process.
Precipitations performed in fluidized bed reactor are commonly used, as fluidized bed leads to solid particles of excellent properties, in terms of morphology, granulometry or density. In this study we propose a new simple model to describe precipitations carried out in continuous fluidized bed reactor equipped with a mechanical stirring: the Mixed Suspension Separated Product Removal (MSSPR). Inspired by the classical Mixed Suspension Mixed Product Removal (MSMPR), this model was applied to the precipitation of uranium peroxide. This latter is involved in the processing of uranium ores to recover uranium from purified leach solution as yellow cake [1].
Thermodynamic study
The uranium peroxide precipitation is achieved at pH ~ 3 by mixing hydrogen peroxide and uranyl sulfate solutions according to the following equilibrium:
UO2SO4 + H2O2 𝐻
↔
2𝑂 UO2(O2) .4 H2O + H2SO4 (1) As the ore solutions are concentrated and multicomponent with a high ionic strength, the deviation from the ideality has to be taken into account to calculate the driving force. The supersaturation ratio, S, is linked to the activities of the uranyl and peroxide ion. The uranyl ion activity is obtained as the product of the uranyl activity coefficient and the uranyl free concentration, which are calculated from the Specific Interaction Theory and the total concentration balance, respectively [2].Kinetic study
The kinetic laws for the primary nucleation were identified using a specific apparatus patented by the French Atomic Energy Commission and AREVA. As the primary nucleation occurs very rapidly, this apparatus allows the reagent mixing state at molecular scale to be controlled. It has been shown to achieve very efficient micromixing time, inferior to the millisecond, with a high degree of precision and reproducibility. The homogeneous and heterogeneous nucleation rates are in agreement with the classical theory proposed by Volmer and Weber.
The crystal growth rate was studied in a batch stirred tank reactor. It was found to be controlled by the surface integration into the crystal lattice, according to a screw dislocation mechanism [2]. The crystal growth rate is first order with respect to the supersaturation and independent from the impeller speed.
Dealing with the agglomeration kernel, it was determined from continuous experimental runs and the mathematical treatment of the crystal size distributions. It appears to be particle size independant, directly proportional to the crystal growth rate and inversely proportional to the shear rate [3].
Experimental runs performed at pilot scale in a continuous fluidized bed reactor showed that the breakage is negligible.
The MSSPR model
While the feed flow rate entering the column in our conditions was not sufficient to create fluidization, the use of a high recirculation rate meant a fluidized-bed was obtained in which a good reagent mixing took place. The fluidized bed precipitator operating in a different way from the perfect mixed reactor, the classical
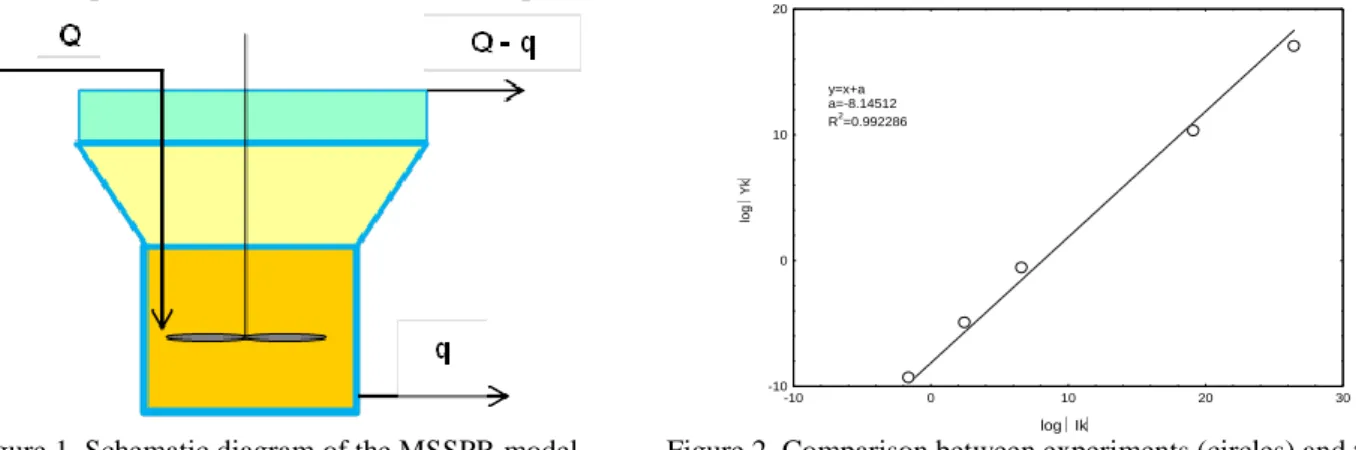
equations of population balance have to be adapted. That is why, we propose a new model which takes into account different outlets for the solid and liquid phases as shown in Figure 1 where Q is the total feed flow rate and q is the outlet flow rate of the suspension.
Figure 1. Schematic diagram of the MSSPR model. Figure 2. Comparison between experiments (circles) and the MSSPR model (line)
The MSSPR model is based on a separation coefficient between the liquid phase and the suspension: α=Q/q. For a MSSPR precipitator in presence of nucleation, crystal growth and agglomeration, the population balance in terms of moment can be expressed as follows:
𝑚𝑘 𝛼𝜏 = 𝑅𝑁∙ 𝐿 ∗𝑘+ 𝑘 ∙ 𝐺𝑚 𝑘−1+ 𝛽𝐼𝑘
(2) with 𝐼𝑘=∫ ∫ [ 1 2(𝐿 3+ 𝜆3)𝑘/3− 𝐿𝑘]∙(𝐿) ∙(𝜆) ∙ 𝑑𝐿 ∙ 𝑑𝜆 ∞ 0 ∞ 0 (3)
where is the residence time (s-1), L and are the particle sizes, (L) is the number particle size density, mk is the k-order moment, G is the crystal growth rate, RN is the nucleation rate, is the agglomeration kernel. The ability of the MSSPR model to describe the precipitation in continuous stirred fluidised bed with recirculation was assessed from several experiments at pilot scale. Equation (2) can be written in logarithmic scale as follows:
log(Yk) = log() + log(Ik) (4)
with
𝑌
𝑘=
𝑚𝛼𝜏𝑘− 𝑅
𝑁∙ 𝐿
∗𝑘− 𝑘 ∙ 𝐺 𝑚
𝑘−1 (5)Figure 2 shows an example of the comparison between the experimental results and the equation (4) and points out a good agreement.
Conclusion
We present in this study a new model adapted to any precipitation reaction performed in a continuous fluidized bed reactor with a recirculation and a mechanical stirring. The successful application of this model reveals its potential for sizing fluidized-bed reactors at the industrial scale.
Acknowledgments: The authors gratefully acknowledge the help of AREVA in sponsoring this study. References:
[1] L.A. Mojica-Rodriguez, M. Bertrand, J.P. Gaillard, F. Auger, H. Muhr, E. Plasari, Experimental study on the uranium peroxide precipitation in a fluidized-bed reactor, Nuclear Engineering and Design, Vol. 293, pp 150-158 (2015).
[2] S. Planteur, M. Bertrand, E. Plasari, B. Courtaud, JP. Gaillard, Thermodynamic and crystal growth kinetic study of the uranium peroxide, CrystEngComm - The Royal Society of Chemistry, Vol. 15 (12), pp 2305-2313 (2013).
[3] M. Bertrand, L. Mojica, E. Plasari, F. Auger, H. Muhr, Study on Agglomeration Kinetics of Uranium Peroxide, Procedia Chemistry, pp. 263-270 (2016).
-10 0 10 20 -10 0 10 20 30 y=x+a a=-8.14512 R2 =0.992286 log Ik lo g Y k