annexe 1 Moveclim (format PDF / 5 Mo)
8
0
0
Texte intégral
Figure
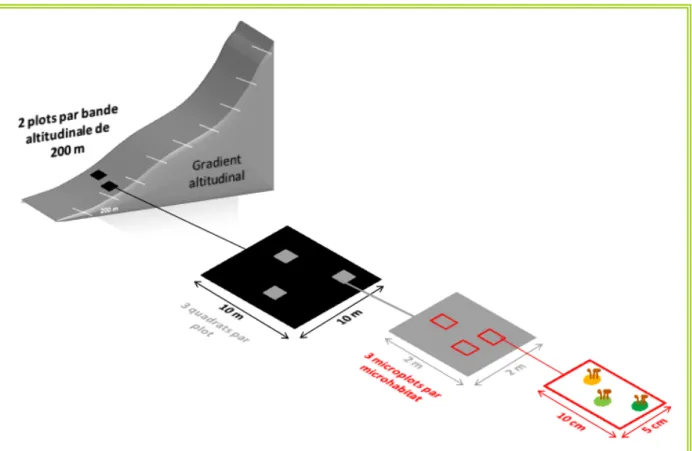
Documents relatifs
[r]
C’est inquiétant car beaucoup d’êtres vivants dépendent des insectes : les plantes pour se reproduire avec la pollinisation, ceux qui les mangent, nous, …..
En effet, dans chaque terme des inégalités à montrer, on peut échanger les rôles de x et y sans changer leur
[r]
[r]
[r]
Dire dans quel cas on peut lisser une série statistique et expliquer ce que
Consigne : Dans chaque cadre, dessine juste ce qu’il faut d’étoiles pour en avoir autant que sur le modèle.. Domaine d’activités : Construire les premiers outils pour structurer