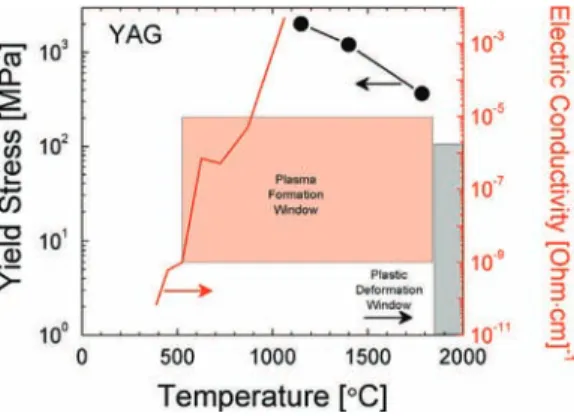
Spark and plasma in spark plasma sintering of rigid ceramic nanoparticles: A model system of YAG
Texte intégral
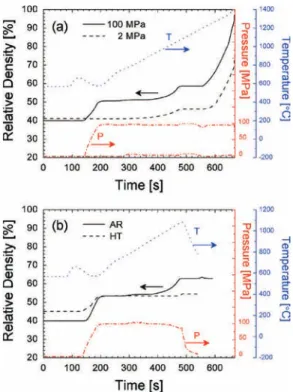
Figure



Documents relatifs
Flash Sintering, Spark Plasma Sintering (SPS)) that are well rec- ognized for advantageous enhanced kinetics [ 6 –8 ], the exploration of non-equilibrium sintering, through
The densification mechanism of Mo nanopowders during spark plasma sintering (SPS) is analyzed by the combination of the regression of the experimental data on regular SPS and on
Flash Sintering, Spark Plasma Sintering (SPS)) that are well recognized for advantageous enhanced kinetics [6-8], the exploration of non-equilibrium sintering, through
Keywords: Spark plasma sintering, Densification, Nanoparticles, Particle sliding, Plastic yield, Grain growth, Ceramics,
In this context, the present work aimed to produce NiCoCrAlY-Ta alloys with refined and homogenous microstructure by using the spark plasma sintering
Recently, we reported a new approach to develop bulk type all solid state Li ion batteries using the Spark Plasma Sintering (SPS) technique [5]. The SPS allows achieving cells with
Bonding of high purity polished copper was investigated using the Spark Plasma Sintering technique (SPS) showing the effect of SPS parameters (surface roughness,
Nanomaterials Processed by Spark Plasma Sintering Faming Zhang, Bikramjit Basu, Lianjun Wang, Izabel Fernanda Machado,..