Nonlinear microbubble behaviour for enhanced drug uptake
Texte intégral
Figure
Documents relatifs
Expert opinion: Some pesticides belonging to several chemical classes, such as organochlorine, pyrethroid and organophosphorus pesticides, have been demonstrated to interact
The predictions of the model are reasonably consistent with available data on lateralization scaling of 500-Hz pure tones, and the model predicts the form of
This chapter will explore the history context of Polypterus Senegalus and other fish with natural predator resistance skin and armor, understand the fundamental
The aim of this work was to develop new methods to achieve a more robust, in vivo evaluation of the functional flow within the tumor vascular network with DCE-US. Three specific
Séquence de Nombres Linéaires (A) Réponses Remplissez les nombres qui manquent de chaque
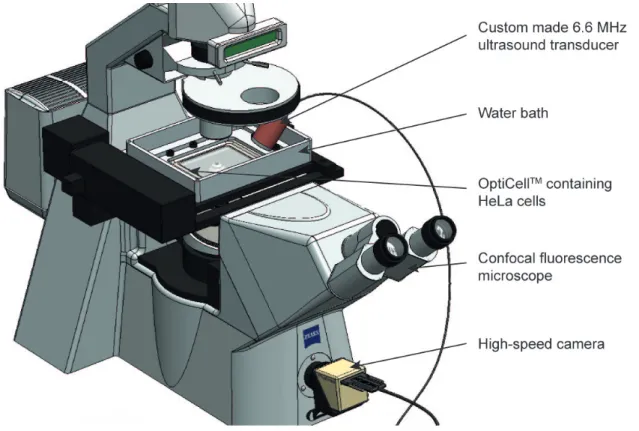
Sonoporation is the transient permeabilisation and resealing of a cell membrane with the help of ultrasound and/or an ultrasound contrast agent, allowing for the
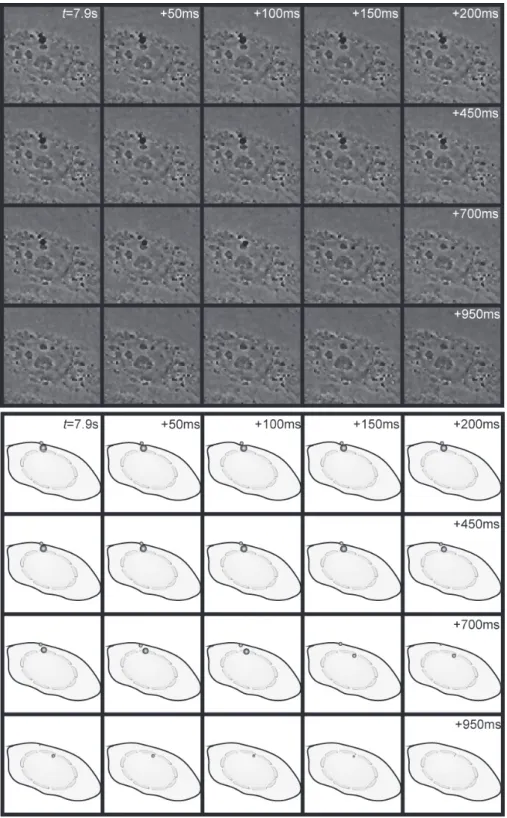
Sonoporation enhances SYTOX-Green uptake through the formation of permeant structures To directly monitor membrane modifications induced by sonoporation, U-87 MG glioblastoma cells
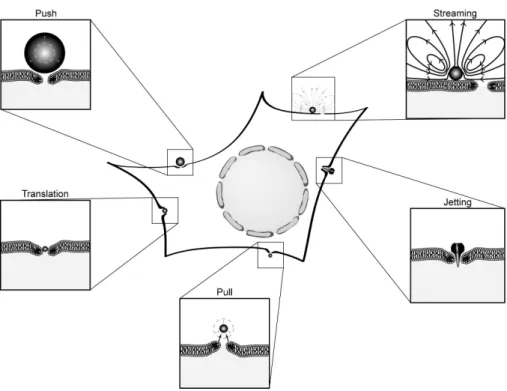
1XPHULFDOVLPXODWLRQVKDYHEHHQPDGHIRUDEXEEOHZLWK VKHOO SURSHUWLHV VLPLODU WR WKRVH XVHG LQ WKH 0DUPRWWDQW VKHOOPRGHO7KHVLPXODWLRQVKDYHVKRZQWKDWFRQWDFWZLWK WKH ULJLG ZDOO GHFUHDVHV