Publisher’s version / Version de l'éditeur:
Cement and Concrete Research, 4, 2, pp. 177-192, 1974-03
READ THESE TERMS AND CONDITIONS CAREFULLY BEFORE USING THIS WEBSITE.
https://nrc-publications.canada.ca/eng/copyright
Vous avez des questions? Nous pouvons vous aider. Pour communiquer directement avec un auteur, consultez la
première page de la revue dans laquelle son article a été publié afin de trouver ses coordonnées. Si vous n’arrivez pas à les repérer, communiquez avec nous à PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca.
Questions? Contact the NRC Publications Archive team at
PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca. If you wish to email the authors directly, please see the first page of the publication for their contact information.
NRC Publications Archive
Archives des publications du CNRC
This publication could be one of several versions: author’s original, accepted manuscript or the publisher’s version. / La version de cette publication peut être l’une des suivantes : la version prépublication de l’auteur, la version acceptée du manuscrit ou la version de l’éditeur.
Access and use of this website and the material on it are subject to the Terms and Conditions set forth at
Exploratory studies of the utilization of sulfur in autoclaved products
Beaudoin, J. J.; Sereda, P. J.
https://publications-cnrc.canada.ca/fra/droits
L’accès à ce site Web et l’utilisation de son contenu sont assujettis aux conditions présentées dans le site LISEZ CES CONDITIONS ATTENTIVEMENT AVANT D’UTILISER CE SITE WEB.
NRC Publications Record / Notice d'Archives des publications de CNRC:
https://nrc-publications.canada.ca/eng/view/object/?id=d5d68fa7-bb2d-4fef-bfea-b2e414cb75d1 https://publications-cnrc.canada.ca/fra/voir/objet/?id=d5d68fa7-bb2d-4fef-bfea-b2e414cb75d1CEMENT and CONCRETE RESEARCH. Vol. 4 , pp. 177-192, 1974. Pergamon P r e s s , I n c . P r i n t e d i n t h e United S t a t e s .
EXPLORATORY STUDIES O F T H E UTILIZATION O F SULFUR IN AUTOCLAVED PRODUCTS
J. J. Beaudoin and P. J. S e r e d a
I/
M a t e r i a l s Section, Division of Building R e s e a r c h , National R e s e a r c h Council of Canada
OTTAWA (Communicated by G .
L .
Kalousek) (Received November 1 9 , 1973) ABSTRACT A significant i m p r o v e m e n t in t h e c o m p r e s s i v e s t r e n g t h of c e m e n t p a s t e autoclaved a t 2 1 6 0 ~ and 300 p s i s t e a m p r e s s u r eh a s been obtained with 1 t o 2 p e r c e n t s u l f u r additions. P h y s i c a l p r o p e r t i e s s u c h a s N, s u r f a c e a r e a , p o r e s i z e d i s t r i b u t i o n , f i r s t d r y i n g s h r i n k a g e , density, p o r o s i t y and m i c r o h a r d n e s s p r o v i d e evidence that t h e addition of s u l f u r p r o d u c e s p r o p e r t i e s a p p r o a c h i n g t h o s e of n o r m a l l y h y d r a t e d p a s t e . In autoclaved m o r t a r s s u l f u r additions a p p e a r to r e d u c e t h e bond between the autoclaved product and s i l i c e o u s a g g r e g a t e , although t h e a d - vantage of s u l f u r in t h e p a s t e i s s t i l l evident. Strengthening of t h e p a s t e (due t o s u l f u r addition) was fully r e a l i z e d when l i m e s t o n e fine a g g r e g a t e w a s u s e d .
On a obtenu, g r % c e 5 l'addition d e 1
5
2 pour cent d e s o u f r e , unea m e l i o r a t i o n i m p o r t a n t e d e l a r e s i s t a n c e
5
l a c o m p r e s s i o n d el a p%te d e ciment m i s e en autoclave
5
216°C e t 'a 300 lv/p2 d ep r e s s i o n d e vapeur. L e s p r o p r i e t e s physiques t e l l e s que l ' a i r e d e s u r f a c e 'a l'azote, l a r e p a r t i t i o n d e s p o r e s , l e r e t r a i t dlti au p r e m i e r s e c h a g e , l a d e n s i t e , l a p o r o s i t e et l a m i c r o d u r e t C i n - diquent que l'addition d e s o u f r e e n t r a r n e d e s p r o p r i e t e s s e r a p - p r o c h a n t d e c e l l e s d e l a p%te h y d r a t t e n o r m a l e m e n t . L'addition d e s o u f r e au m o r t i e r m i s en autoclave s e m b l e r C d u i r e l ' a d h e r - ence e n t r e l e produit d'autoclave et l e granulat silicieux, bien que l'avantage du s o u f r e dans l a p%te soit e n c o r e evident.
L'addition d e s o u f r e a u g m e n t e n e t t e m e n t l a r e s i s t a n c e d e l a p%te l o r s q u l o n u t i l i s e du granulat fin.
Vol.
4, No.
2
AUTOCLAVING
,
SULFUR, CEMENT PASTES, MORTARS
Introduction
As t h e supply of e l e m e n t a l sulfur in Canada i s expected t o r e a c h 50
million tons by 1980 (1), the challenge t o find new ways t o u s e t h i s m a t e r i a l
i s g r e a t . The s t u d i e s now r e p o r t e d w e r e fundamental in approach; t h e y w e r e undertaken in a n attempfto p r o v i d e a b a s i s for developing autoclaved c o n c r e t e
of s u p e r i o r p r o p e r t i e s with portland c e m e n t
-
sulfur b i n d e r s . One of the p r o -b l e m s with autoclaved c e m e n t p a s t e s i s t h a t t h e y a r e i n f e r i o r b i n d e r s due t o
t h e f o r m a t i o n of a l p h a dicalcium s i l i c a t e h y d r a t e . P o r t l a n d cement
-
sulfurp a s t e s , without r e a c t i v e s i l i c e o u s p o w d e r s , w e r e used a s a f i r s t b a s i c s t e p in the o v e r - a l l investigation. M o r t a r s u s i n g s i l i c e o u s and c a l c i t e s a n d s , without finely ground r e a c t i v e s i l i c e o u s m a t e r i a l s , w e r e studied a s well a s c e m e n t
-
f l y a s h and c e m e n t-
sand-
fly a s h m i x t u r e s .It was postulated that during t h e autoclaving p r o c e s s , sulfur would melt, flow into i n t i m a t e contact with newly f o r m e d hydration p r o d u c t s , and poly- m e r i z e . T h e m e r i t s of s u l f u r in t h i s s t a t e would then be i m p a r t e d t o t h e composite. It was hoped that t h e i n t i m a c y of s u r f a c e - t o - s u r f a c e contiguity between sulfur and m i c r o s t r u c t u r e would not only provide s t a b i l i t y but a l s o d e l a y t h e t r a n s f o r m a t i o n of p o l y m e r i c s u l f u r t o t h e S a o r c r y s t a l l i n e f o r m .
The p o s s i b i l i t y of c h e m i c a l o r topochemical r e a c t i o n a s well a s l a t t i c e substitution o r e n t r a n c e into solid solution m a y p r e s e n t other b a s e s on which t o a s s e s s e x p e r i m e n t a l r e s u l t s .
E x p e r i m e n t s with Cement P a s t e Autoclaving
To e n s u r e a r e l a t i v e l y high d e g r e e of p o l y m e r i z a t i o n of s u l f u r a s well a s rapid hydration of c e m e n t , t h e autoclaving r e g i m e followed t h a t of ASTM
C151. Essentially, t h e s a m p l e s w e r e heated f r o m r o o m t e m p e r a t u r e t o 216°C
and 300 p s i s t e a m p r e s s u r e w h e r e t h e y r e m a i n e d f o r 3 h o u r s . While t h e autoclave was being heated t h e s u l f u r had sufficient l a g t i m e in t h e low v i s - c o s i t y liquid s t a t e (120 t o 1 4 0 ° C ) t o p e r m e a t e t h e m i c r o s t r u c t u r e ; and a s i t
was finely divided and w e l l d i s p e r s e d throughout t h e m i x t h e effect of t o r
-
AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS V o l . 4, N o . 2
Mixes
Two-in. cubes were cast from the mixes outlined in Table 1 and moist cured for 24 hours p r i o r to autoclaving. At least t h r e e cubes were cast for every t e s t condition. TABLE 1 C e m e n t P a s t e M i x e s Mix 1 2 3 4 5 6 7 8 9 10 1 1 12 W / C Ratio 0. 30 0. 30 0. 30 0. 30 0. 30 0. 30 0. 35 0. 35 0. 35 0. 35 0. 35 0. 35 70 Sulfur ;" 0 1/2 1 1-1/2 2 10 0 1/2 1 1-1/2 2 10 -" Sulfur e x p r e s s e d a s 70 by weight of c e m e n t .
Results and Discussion
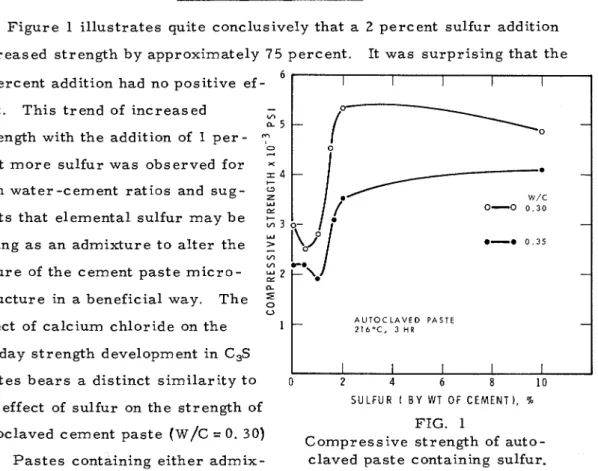
Figure 1 illustrates quite conclusively that a 2 percent sulfur addition
increased strength by approximately 7 5 percent. It was surprising that the 1 percent addition had no positive ef-
fect. This trend of increased CA
a 5 strength with the addition of 1 p e r - c e n t m o r e s u l f u r w a s observedfor
both water -cement ratios and sug-
g
W W/C
P: 0-0 0 . 3 0
gests that elemental sulfur may be
acting a s an admixture to alter the 0-0 0 . 3 5
nature of the cement paste m i c r o -
structure in a beneficial way. The
2
U
A U T O C L A V E D PASTE
effect of calcium chloride on the Z I ~ O C , 3 H R
28 -day strength development in C,S
s
pastes b e a r s a distinct similarity to o 2 4 6 8 1 0
S U L F U R ( B Y WT O F CEMENT), %
the effect of sulfur on the strength of -
FIG. 1
autoclaved cement paste (w/C = 0.30) Compressive strength of auto-
-
(2). P a s t e s containing either admix- claved paste containing sulfur.
t u r e show a decrease in strength for additions greater than 2 percent. It
appears, therefore, that calcium chloride i s not unique in providing optimum
AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS V o l . 4, N o . 2 p h y s i c a l m e a s u r e m e n t s was m a d e t o identify changes in p h y s i c a l p r o p e r t i e s . P h y s i c a l P r o p e r t i e s
N2 s u r f a c e a r e a . Nitrogen s u r f a c e a r e a s i n c r e a s e d l i n e a r l y with sulfur a d d i -
tion. They w e r e 9, 13 and 2 4 m2/g, r e s p e c t i v e l y , f o r 0, 2 and 10 p e r c e n t s u l -
fur additions.
P o r e s i z e distribution. F i g u r e 2 shows quite c l e a r l y t h a t t h e distribution of
p o r e s , a s d e t e r m i n e d by Hg p o r o s i m e t r y , i s s y s t e m a t i c a l l y changed f r o m a c o a r s e t o a fine d i s t r i b u t i o n a s sulfur content i n c r e a s e s .
P R E S S U R E . P S I
D l A M E T E R , fY
FIG. 2
P o r e s i z e distribution of autoclaved p a s t e s containing s u l f u r .
F i r s t drying s h r i n k a g e . S a m p l e s of autoclaved p a s t e (initially at 100 p e r c e n t
RH) w e r e equilibrated a t 11 p e r c e n t RH and simultaneous length change m e a -
s u r e m e n t s taken. F i g u r e 3 d e m o n s t r a t e s t h e proportionality of f i r s t drying s h r i n k a g e with s u l f u r content. It i s evident that the sulfur additive a l t e r s t h e m i c r o s t r u c t u r e significantly and t h a t length change f o r t h e p a s t e with 10
V o l . 4, No. 2
AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS
p e r c e n t s u l f u r additive a p p r o a c h e s t h a t of a n o r m a l l y h y d r a t e d p a s t e with W/C = 0. 25. Density. D e n s i t i e s d e t e r - mined b y h e l i u m d i s p l a c e - m e n t w e r e 2. 639 and 2. 490, r e s p e c t i v e l y , f o r p a s t e with 0 and 1 0 p e r c e n t s u l - f u r a d d i t i o n s . D e n s i t i e s of n o r m a l l y h y d r a t e d p a s t e u s u a l l y r a n g e f r o m 2. 00 t o 2. 20 (3). T h i s o f f e r s f u r - t h e r evidence t h a t t h e a d d i - tion of s u l f u r b r i n g s t o t h e autoclaved p a s t e p r o p e r
-
t i e s a p p r o a c h i n g t h o s e of n o r m a l l y h y d r a t e d c e m e n t p a s t e . P o r o s i t y . S a t u r a t e d p a s t e s a m p l e s w e r e weighed in d a m p - d r y condition in a i r , 0 1 1 1 1 1 1 1 1 I I 1 1 1 1 1 1 1 1 1 1 1 1 1 1 1 A U T O C L A V E D - 0'1 SULFUR w / c = 0 3 5 - A U T O C L A V E D 2 7 SULFUR X 1-
- A U T O C L A V E D 25 - 30 - S H R I N h A G E VS T I M E H Y D R A T E D l o o o / -I lo/ RH I I I 1 i l l 1 1 1 1 1 1 1 1 1 0 . 0 1 0 . 1 1 . 0 1 0 TIME, D A Y S FIG. 3 Fir s t d r y i n g s h r i n k a g e of autoclaved p a s t e s containing s u l f u r .weighed in w a t e r , t h e n D - d r i e d . T h e D-dried s a m p l e s w e r e vacuum s a t u - r a t e d in m e t h a n o l and r e - w e i g h e d . T a b l e 2 r e c o r d s t h e r e s u l t s T A B L E 2 Methanol Volumes Hg Sample:: V W a t e r V Methanol V W a t e r 15, 000
70
70
V Methanol70
:: E a c h value i s t h e a v e r a g e of five d e t e r m i n a t i o n s .182 V o l . 4, No. 2 AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS
It i s significant that Water i s approximately unity, even a f t e r D-drying,
V Methanol
f o r p a s t e with 0 p e r c e n t sulfur. This suggests that during autoclaving t h e
l a y e r s approach a s t a t e of complete collapse and that w a t e r removed on D- drying i s essentially adsorbed water (4). On r e -saturation with methanol t h e volume adsorbed i s equal t o that f o r water; methanol apparently occupies
s i t e s s i m i l a r t o those occupied by w a t e r on t h e external s u r f a c e s of t h e l a y e r s . With the addition of sulfur t h e l a y e r e d system i s modified t o the extent that the volume of w a t e r adsorbed i s 16 and 33 p e r c e n t g r e a t e r than that f o r methanol. This i s f u r t h e r evidence that modification of t h e m i c r o s t r u c t u r e
is such that i t approaches that of n o r m a l l y hydrated p a s t e . It i s s u r p r i s i n g
that strength i s not i n c r e a s e d although s u r f a c e a r e a continues t o change in t h e region 2 t o 10 p e r c e n t sulfur addition.
Microhardness. Table 3 gives values of m i c r o h a r d n e s s and strength f o r
W/C = 0. 30 paste. A Leitz m i c r o h a r d n e s s testing machine with a Vickers
indenter was used (5).
TABLE 3
Values of Microhardness and Strength f o r W/C
=
0. 30 P a s t eSample* Microhardness Strength
(kg/mm2) (PS i)
0% S
9.
4 2940*
Average of a t l e a s t t h r e e determinations.It i s c l e a r that with the addition of sulfur subsequent autoclaving of cement p a s t e increased t h e h a r d n e s s of the m a t e r i a l . This appears to have a direct relation t o the c o m p r e s s i v e strength of the m a t e r i a l .
SEM Micrographs
In the p a s t e the m a j o r f e a t u r e in the p r e s e n c e of sulfur (1 0 ~ e r c e n t ) appeared t o be i n t e r s p e r s e d , i r r e g u l a r , platy m a s s e s . Typical micrographs
Vol. 4, No.
2
1 8 3
AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS
a r e shown i n F i g s . 4 (A t o
c ) .
In c o n t r a s t , t h e morphology of t h e autoclavedp a s t e without sulfur ( F i g s . 4 (D and E) ) w a s e s s e n t i a l l y f r e e of p l a t y m a t t e r .
FIG. 4
Scanning e l e c t r o n m i c r o g r a p h s of
autoclaved p a s t e ; A, B, C with 10
p e r c e n t s u l f u r addition; D, E, with n o s u l f u r addition.
1 8 4
AUTOCLAVING
,
SULFUR, CEMENT PASTES, MORTARS V o l . 4 , N o . 2Differential T h e r m a l A n a l y s i s
Differential t h e r m a l a n a l y s i s w a s c a r r i e d out u s i n g t h e Du Pont 900 T h e r m a l A n a l y s e r . F i g u r e 5 gives t h e DTA t r a c e s of s u l f u r and p a s t e c o n - taining 0, 2 and 10 p e r c e n t s u l f u r , r e s p e c t i v e l y . Distinguishing f e a t u r e s include: I l l I I 1 I I I I
I
-
A U T O C L A V E D C E M E N T PASTE 2 1 6 " C , 3 HR I I I I I I I I I II
I
200 4 0 0 6 0 0 8 0 0 1 0 0 0 T E M P E R A T U R E . 'C FIG. 5 Differential t h e r m a l a n a l y s i s of autoclaved c e m e n t p a s t e con-
taining sulfur. (1) t h e l i m e p e a k d i s a p p e a r s in t h e p r e s e n c e of 10 p e r c e n t s u l f u r ,'
(2) t h e r e i s a s m a l l e x o t h e r m a l p e a k a t a p p r o x i m a t e l y 820 t o 830°C in t h e p r e s e n c e of 10 p e r c e n t s u l - f u r . It i s p o s s i b l e t h a t t h e a b s e n c e of a l i m e peak a t 10 p e r c e n t sulfur content indicates t h a t t h e l i m e h a s gone into t h e f o r m a t i o n of a high l i m e p r o d u c t . R e s u l t s f r o m m e - c h a n i c a l m i x t u r e s of l i m e and s u l -f u r indicate that l i m e does not i n t e r -
a c t with s u l f u r during t h e DTA run. F i g u r e 6, h o w e v e r , shows a DTA r u n on a 1:l m i x t u r e of l i m e and s u l f u r autoclaved p r i o r t o DTA. In c o n t r a s t , t h e r e i s a n e x o t h e r m a l p e a k at a p p r o x i m a t e l y 795 t o 800°C and a s m a l l endothermic p e a k a t about 465°C. F o r t h i s p a r t i c u l a r 1: 1 m i x t u r e t h i s might s u g g e s t t h e p r e s e n c e of calcium sulfide. It i s of i n t e r e s t that t h e e x o t h e r m a l p e a k probably r e s u l t s f r o m t h e autoclaving p r o c e s s and not f r o m t h e enthalpic effect of t h e DTA r u n i t s e l f . T h e r m o p r a v i m e t r i c Analysis
T a b l e 4 t a b u l a t e s d a t a e s t i m a t e d f r o m c u r v e s of t h e r m o g r a v i m e t r i c
V o l . 4, N o . 2 1 8 5 AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS
FIG. 6 Differential t h e r m a l a n a l y s i s of m i x t u r e s of l i m e and sulfur. I I I I I I
I
M I X T U R E S O F C a ( O H ) : , A N D S U L F U R A - A U T O C L A V E D S U L F U R 0 - M E C H A N I C A L M I X T U R E O F L l M E A N D S U L F U R C - A U T O C L A V E D M I X T U R E O F L l M E A N D S U L F U R.
1 1 1 1 I I I I 200 400 6 0 0 800 1000 T E M P E R A T U R E . " C TABLE 4 Calculations f r o m TGA T r a c e s f o r Autoclaved P a s t e sSample Ca(OH)a Total Weight W720
-
W l ~ ~ ~L o s s
70
IgnitedOJo
Sulfur Weight70
D - d r y70
70
of TotalX - r a y Diffraction
X - r a y diffraction a n a l y s e s w e r e p e r f o r m e d using a P h i l i p s powder c a m - e r a (PW 1024). T h e m i n e r a l s identified in autoclaved p a s t e containing 0, 2
V o l . 4, N o . 2 AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS
and 10 p e r c e n t sulfur w e r e t o b e r m o r i t e , aC,SH and Ca(OH),. T h e r e was
evidence a l s o of xontolite i n p a s t e s containing 0 and 2 p e r c e n t s u l f u r additions.
Only i n t h e autoclaved p a s t e containing 10 p e r c e n t s u l f u r was t h e r e evidence of CSH (11).
X - r a y diffraction indicated t h e p r e s e n c e of negligible amounts of c r y s
-
t a l l i n e sulfur in autoclaved p a s t e a t 30 d a y s , suggesting t h a t c o n v e r s i o n of s u l f u r f r o m t h e p o l y m e r i c f o r m t o t h e o r t h o r h o m b i c f o r m i s negligible at room t e m p e r a t u r e . T h i s i s i n a g r e e m e n t with equal s t r e n g t h s f o r autoclaved p a s t e with sulfur a t 30 d a y s .
M o r t a r with Sulfur Additions
T h e f i r s t p h a s e of t h i s investigation showed t h a t s u l f u r h a s a m a j o r effect
on t h e s t r e n g t h and p r o p e r t i e s of autoclaved p a s t e . It i s known t h a t auto
-
claving of m o r t a r containing s i l i c e o u s sand r e s u l t s in a l a r g e i n c r e a s e in s t r e n g t h when c o m p a r e d with t h e s a m e m o r t a r hydrated a t n o r m a l conditions b e c a u s e of t h e pozzolanic effect (owing t o t h e i n c r e a s e i n t h e bond between
A U T O C L A V E D M O R T A R C U B E S
I
A / C = 2 . 7 5 : 1 , W / C = 0 . 5 0 0 -R O - OI
0 - O O T T A W A S A N D A N N E A L E D i l O ° C / 2 HRI
0-0 N A T U R A L S A N D - .-0 O T T A W A S A N D 1 5 0 ° C / 6 HR A-A L I M E S T O N E F I N E A G G S U L F U R ( B Y W T O F CEMENT). % FIG. 7 sand and c e m e n t p a s t e ) . T h e second p h a s e w a s designed t o show whether both t h e beneficial effect of s u l f u r on c e m e n t p a s t e and t h e pozzolanic r e a c t i o n with sand could b e r e a l i z e d t o g e t h e r . F i g u r e 7 c l e a r l y i l l u s t r a t e s that f o r m i x e s 1, 4 and 5 ( T a b l e 5) t h e r e i s up t o 50 p e r c e n t d e c r e a s e i n s t r e n g t h f o r a wide r a n g e of a g g r e g a t e - c e m e n t r a t i o s when sulfur i s p r e s e n t . F i g u r e 8 plots c o m p r e s s i v e s t r e n g t h v e r s u s sulfur content f o r m i x e s 1, 2and 3. Mixes with both n a t u r a l sand
and Ottawa sand exhibit up t o 50 p e r
-
C o m p r e s s i v e s t r e n g t h of autoclaved cent d e c r e a s e i n s t r e n g t h in t h e p r e -
V o l . 4, No. 2 1 8 7
AUTOCLAVING
,
SULFUR, CEMENT PASTES, MORTARSf o r 2 h o u r s a f t e r autoclaving a t 2 1 6 0 ~ had l i t t l e effect on t h i s t r e n d ; and low- p r e s s u r e s t e a m c u r i n g (150°C f o r 6 h o u r s ) g a v e no significant change in
s t r e n g t h f o r m o r t a r containing s u l f u r . In a l l t h e e x p e r i m e n t s , m o r t a r m a d e with s i l i c e o u s a g g r e g a t e exhibited l a r g e s t r e n g t h d e c r e a s e s when autoclaved i n t h e p r e s e n c e of s u l f u r . With n o s u l f u r it showed 100 p e r c e n t i n c r e a s e when t h e t e m p e r a t u r e of r e a c t i o n was i n c r e a s e d f r o m 150 t o 21 6 ° C . When c r u s h e d l i m e s t o n e w a s u s e d a s a fine a g g r e g a t e , s u l f u r i n c r e a s e d t h e s t r e n g t h of t h e autoclaved p r o d u c t b y a p p r o x i m a t e l y 50 p e r c e n t , showing t h a t t h e b e n e f i c i a l effect of s u l f u r on p a s t e i s r e t a i n e d . T h i s f a c t a l l o w s one t o p o s t u l a t e t h a t t h e r e i s a s u r f a c e r e a c t i o n a n d / o r modification between t h e s i l i c a and t h e p a s t e in t h e p r e s e n c e of s u l f u r . F i g u r e 9 i l l u s t r a t e s t h a t s u l f u r c a n b e b e n e f i c i a l i f sand i s p a r t i a l l y r e -
placed (Mixes
6
and 7) b y f l y a s h . F o r a 50/50 r e p l a c e m e n t t h e r e i s about a26 p e r c e n t i n c r e a s e in s t r e n g t h a t 2 p e r c e n t s u l f u r content. F o r a 25 p e r c e n t r e p l a c e m e n t t h e r e i s a p p r o x i m a t e l y a 15 p e r c e n t i n c r e a s e i n s t r e n g t h a t 2 p e r c e n t s u l f u r content. T A B L E 5 Cement/A g g r e g a t e Mixes No. A g g r e g a t e P r o p o r t i o n s
w
/c
1 Ottawa sand 1:2. 75 C / A 2. Ottawa sand 1: 1 . 00 C / A 3 Ottawa sand 3 : l . 00 C / A 4 N a t u r a l sand l : 2 . 75 C / A 5 L i m e s t o n e f i n e a g g r e g a t e 1:2. 75 C / AOttawa s a n d and f l y a s h C:S:fly a s h
1:2. 06:O. 68
W
- -
- 0. 382 C t a s hOttawa sand and f l y a s h C:S:fly a s h
1 : l . 37: 1. 37
W
--
- 0 . 2 7 1188 V o l . 4, N o . 2 AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS
Effect of Sulfur in the P r e s e n c e
of Siliceous Aggregate
-
CT)n 5
F u r t h e r comment on the r e l a t i o n s
p r e s e n t e d in F i g u r e s 1 and 7 m a y s e r v e
0
t o c l a r i f y t h e effect of sulfur on the Z w
E engineering s t r e n g t h of autoclaved
r,
3 W > m o r t a r containing siliceous a g - - CT) CT) w 2 g r e g a t e . E a 5 0T h e r o l e sulfur plays in enhancing
t
A U T O C L A V E D M O R T A R C U B E S 00,- S U L F U R - oIc----020b S U L F U R O-OIOoo S U L F U R q I - the c o m p r e s s i v e s t r e n g t h of autoclavedI
p a s t e i s c l e a r l y i l l u s t r a t e d in F i g u r e 1.,,
1
I I II
F i g u r e 7 d e m o n s t r a t e s t h e attainment 0. 3 3 1. 0 2. 0 2. 7 5 A G G R E G A T E I C E M E N Tof added s t r e n g t h owing to pozzolanic
FIG. 8
S U L F U R ( B Y W T O F C E M E N T ) , %
C o m p r e s s i v e s t r e n g t h vs a g g r e g a t e -
cement r a t i o of autoclaved m o r t a r cubes containing sulfur.
activity in t h e p r e s e n c e of a siliceous
p h a s e . In the p r e s e n c e of both sulfur
and a siliceous phase, however, the
s t r e n g t h of the composite r e v e r t s t o
that of cement p a s t e alone. In effect,
sulfur with s i l i c a negates the advan
-
t a g e offered by pozzolanic activity.
That this i s due to t h e fact that sulfur
coats t h e a g g r e g a t e and in itself im
-
p a r t s poor bond c h a r a c t e r i s t i c s to t h ea g g r e g a t e - m a t r i x i n t e r f a c e i s im -
probable; examination of thin sections
revealed l i t t l e or no sulfur a t t h e
FIG. 9 a g g r e g a t e m a t r i x i n t e r f a c e . T h e
C o m p r e s s i v e s t r e n g t h of autoclaved autoclaved product, modified in t h e
m o r t a r containing sulfur and
flv a s h , p r e s e n c e of s u l f u r , m a y simply have
V o l . 4, N o . 2
AUTOCLAVING, SULFUR, CEMENT P A S T E S , MORTARS
T h e n a t u r e and p r e s e n c e of t h e nullifying m e c h a n i s m a p p e a r s t o be d e - pendent on t h e inclusion of a s i l i c e o u s a g g r e g a t e in t h e m a t r i x . T h e negative
effect of s u l f u r i s a b s e n t in t h e p r e s e n c e of l i m e s t o n e fine a g g r e g a t e . T h e m a j o r d i f f e r e n c e between m o r t a r m a d e with l i m e s t o n e f i n e a g g r e g a t e and m o r t a r m a d e with s i l i c e o u s a g g r e g a t e i s t h e a b s e n c e in t h e f o r m e r of a n y pozzolanic r e a c t i o n d u r i n g autoclaving.
Without s u l f u r , t h e r e f o r e , autoclaved m o r t a r m a d e with s i l i c e o u s a g g r e - gate h a s a m u c h g r e a t e r s t r e n g t h than h a s l i m e s t o n e m o r t a r . As mentioned p r e v i o u s l y , however, s u l f u r enhances t h e s t r e n g t h p r o p e r t y of l i m e s t o n e m o r -
t a r by about 50 p e r c e n t , c o r r e s p o n d i n g roughly to t h e i n c r e a s e in p a s t e s t r e n g t h f r o m s u l f u r .
F l y Ash Additions
T h e p o s s i b i l i t y was explored that t h e f i n e n e s s of t h e s i l i c a might be a
controlling f a c t o r in s t r e n g t h reduction r e s u l t i n g f r o m s u l f u r addition in m o r
-
t a r with s i l i c e o u s a g g r e g a t e . It w a s postulated that a finely divided s i l i c a in the f o r m of fly a s h might p r o v i d e a s o u r c e of pozzolanic a c t i v i t y t h a t would o v e r r i d e any i l l effects f r o m s u l f u r and t a k e advantage of t h e beneficial effects it gives to the p a s t e a l o n e .
T o b e optimally useful s u l f u r should p r o v i d e s t r e n g t h i n c r e a s e a t t h e optimum s i l i c a content, p r e f e r a b l y o v e r t h e c o m p l e t e r a n g e of the w e l l -
established Menzel r e l a t i o n ( 6 ) . F i g u r e 10 plots s t r e n g t h a s a function of fly
a s h content f o r m i x e s containing 0, 2 and 10 p e r c e n t s u l f u r . As noted, both 2 and 10 p e r c e n t s u l f u r additions give l a r g e s t r e n g t h i n c r e a s e s in p a s t e when no a s h i s p r e s e n t . At about 10 p e r c e n t a s h the 2 p e r c e n t sulfur gives no advantage, and a t t h e optimum s i l i c a content it gives about 15 p e r c e n t a d v a n - t a g e . With 10 p e r c e n t s u l f u r t h e r e i s no advantage u n t i l t h e optimum, w h e r e equality i s r e a c h e d . At about 75 p e r c e n t a s h , h o w e v e r , t h e r e i s a p p r o x i
-
m a t e l y 75 p e r c e n t i n c r e a s e in s t r e n g t h f o r 10 p e r c e n t s u l f u r and about 30 p e r c e n t i n c r e a s e f o r 2 p e r c e n t s u l f u r .SEM M i c r o g r a p h s
1 9 0 V o l . 4, N o . 2 AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS
akin t o that of a high w a t e r -cement r a t i o l 6 I I I I
A U T O C L A V E D C U B E S
p a s t e ( F i g u r e 11). T h e s t r u c t u r e a p - C E M E N T A N D
F L Y A S H 2 1 b D C / 3 H R
-
p e a r s t o be highly p o r o u s and open and 14-
h a s a t l c a c t u s - l i k e t ' composition, with Z a 12 n e e d l e s emanating f r o m d i s c r e t e c l u s - 0 t e r s of hydration product.
h
c o n t r a s t , ;I 1 10p a s t e without sulfur ( F i g u r e 4(e)) h a s a
5
W
distinctly different morphology. I-
* 8 W Differential T h e r m a l Analysis 5 * * F i g u r e 12 shows DTA t r a c e s f o r t h e r a n g e of a g g r e g a t e - c e m e n t r a t i o s w 0 0 % S U L F U R - 2% S U L F U R t e s t e d . In a l l c a s e s t h e m a x i m u m C. 1 0 % S U L F U R amount of l i m e i s p r e s e n t at 2 p e r c e n t W A T E R / S O L I D S = 0 . 2 8 6 -
sulfur content; only s m a l l o r negligible
I
I
amounts of l i m e a r e p r e s e n t at 0 and
n
n
10 p e r c e n t s u l f u r content. In c o m - "0 20 4 0 60 80 100
F L Y A S H ( B Y W T O F S O L I D ) , %
p a r i s o n , f o r p a s t e t h e r e i s a m a x i - FIG. 10
mum amount of l i m e without sulfur C o m p r e s s i v e s t r e n g t h of autoclaved
c e m e n t p a s t e containing s u l f u r and fly a s h . and a s i m i l a r a b s e n c e of l i m e a t 10 p e r - c e n t s u l f u r . In t h e m o r t a r (A/C = 2. 75) t h e r e i s an e x o t h e r m i c peak a t 810°C for 10 p e r c e n t s u l f u r , suggesting t h e p r e - s e n c e of t o b e r m o r i t e ; t h i s p e a k i s not p r e s e n t in p a s t e with 0 p e r c e n t s u l f u r . Conclusions 1. T h e s t r e n g t h of autoclaved p a s t e con- taining 2 p e r c e n t s u l f u r i n c r e a s e s by a s FIG
.
11 much a s 75 p e r c e n t . T h i s i n i t s e l f c o n - Scanning f i r m s t h a t s u l f u r h a s a m a j o r influence i n autoclaved m o r t a r containingV o l . 4, N o . 2 1 9 1 AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS
t h e final p r o d u c t . F u r t h e r confirmation
of t h e effect of s u l f u r on the h y d r a t i o n A / C o 3 3
product i s offered by t h e following: (1) s u r f a c e a r e a i n c r e a s e , (2) p o r e s i z e t r a n s i t i o n f r o m c o a r s e to fine, A / C - 1 0 0 (3) i n c r e a s e d d r y i n g s h r i n k a g e , (4) VH,o/ V ~OH ~i n c r e a s e , , (5) d i s a p p e a r a n c e of l i m e and a p - I-
a
p e a r a n c e of CSH (11) f o r p a s t e containing 10 p e r c e n t s u l f u r . 2. T h e fact t h a t a r e l a t i v e l y s m a l l amount of s u l f u r p r o d u c e s s u c h m a j o r changes s u g g e s t s t h a t t h e effect i s t h a t of a n a d m i x t u r e . 3. Sulfur c a n be u s e d to p r o d u c e a u t o - claved p a s t e with p r o p e r t i e s a p p r o a c h - 0 2 0 0 4 0 0 6 0 0 8 0 0 1 0 0 0 ing t h o s e of n o r m a l l y h y d r a t e d p a s t e . T E M P E R A T U R E . " C ( N o r m a l l y h y d r a t e d p a s t e i s s t r o n g e r FIG. 12 than autoclaved p a s t e f o r s i m i l a r Differential t h e r m a l a n a l y s i s of a u t o -m i x e s . ) claved m o r t a r containing sulfur.
4. T h e r e i s a n a p p a r e n t d e c r e a s e in s t r e n g t h of autoclaved m o r t a r i n c o r - p o r a t i n g s u l f u r a s a n additive when i t i s m a d e with a s i l i c e o u s a g g r e g a t e , c o m p a r a b l e t o t h e gain due t o pozzolanic a c t i o n between s i l i c a and p a s t e . 5. T h e r e i s a n a p p a r e n t i n c r e a s e i n s t r e n g t h of autoclaved m o r t a r i n c o r - p o r a t i n g s u l f u r a s a n additive when t h e m o r t a r i s m a d e with a n o n - s i l i c e o u s a g g r e g a t e s u c h a s l i m e s t o n e fine a g g r e g a t e , c o m p a r a b l e t o t h e gain in s t r e n g t h of p a s t e contributed by s u l f u r .
6. T h e ill effects of s i l i c e o u s a g g r e g a t e s on s t r e n g t h of autoclaved m o r t a r
i n t h e p r e s e n c e of s u l f u r a r e p a r t i a l l y o v e r c o m e when s i l i c a ( i n finely divided f o r m ) r e p l a c e s c e m e n t i n such quantity a s w i l l equal o r exceed t h e optimum
1 9 2
AUTOCLAVING, SULFUR, CEMENT PASTES, MORTARS V o l . 4, No. 2
of t h e M e n z e l s t r e n g t h r e p l a c e m e n t function.
T h e f u n d a m e n t a l a p p r o a c h t a k e n h a s d e m o n s t r a t e d t h e c a r d i n a l f e a t u r e s of t h e c e m e n t - s u l f u r s y s t e m d u r i n g autoclaving and indicated t h e p o t e n t i a l f o r f u r t h e r w o r k d i r e c t e d t o w a r d p r a c t i c a l application.
Acknowledgements
The a u t h o r s w i s h t o t h a n k t h e i r c o l l e a g u e s R. F. F e l d m a n and V. S. R a m a c h a n d r a n f o r m a n y helpful d i s c u s s i o n s , and t o acknowledge t h e c o n t r i -
bution of S. Dods, R. M e y e r s , G. P o l o m a r k and E. Quinn f o r a s s i s t i n g with
the e x p e r i m e n t a l work.
T h i s p a p e r i s a c o n t r i b u t i o n f r o m the Division of Building R e s e a r c h , National R e s e a r c h Council of Canada, and i s published with t h e a p p r o v a l of t h e D i r e c t o r of the Division.
R e f e r e n c e s
1. A. H. Vroom. "Sulphur Utilization
-
A Challenge and an Opportunity".National R e s e a r c h Council of Canada. NRC 12241, 80p. (1971).
2. V. S. R a m a c h a n d r a n . T h e r m o c h i m i c a A c t a
-
3, 343 (1972).3. R. F. Feldman. C e m . and Concr. R e s . 2, 1, 123 (1972).
-
4. R. F. Feldman. Sorption and Length Change Scanning I s o t h e r m s of
Methanol and Water on H y d r a t e d P o r t l a n d C e m e n t . P r o c . , Fifth I n t e r n a t .
Syrnp. Chem. C e m e n t , Vol. 111, 53. Tokyo (1968).
5. P. J. S e r e d a . Cem. and Concr. Res. 2, 6, 717 (1972).