ContentslistsavailableatScienceDirect
Resuscitation
j o ur na l h o me p a g e:ww w . e l s e v i er . c o m / l o c a t e / r e s u s c i t a t i o n
European
Resuscitation
Council
Guidelines
for
Resuscitation
2015
Section
4.
Cardiac
arrest
in
special
circumstances
Anatolij
Truhláˇr
a,b,∗,
Charles
D.
Deakin
c,
Jasmeet
Soar
d,
Gamal
Eldin
Abbas
Khalifa
e,
Annette
Alfonzo
f,
Joost
J.L.M.
Bierens
g,
Guttorm
Brattebø
h,
Hermann
Brugger
i,
Joel
Dunning
j,
Silvija
Hunyadi-Antiˇcevi ´c
k,
Rudolph
W.
Koster
l,
David
J.
Lockey
m,w,
Carsten
Lott
n,
Peter
Paal
o,p,
Gavin
D.
Perkins
q,r,
Claudio
Sandroni
s,
Karl-Christian
Thies
t,
David
A.
Zideman
u,
Jerry
P.
Nolan
v,w,
on
behalf
of
the
Cardiac
arrest
in
special
circumstances
section
Collaborators
1aEmergencyMedicalServicesoftheHradecKrálovéRegion,HradecKrálové,CzechRepublic
bDepartmentofAnaesthesiologyandIntensiveCareMedicine,UniversityHospitalHradecKrálové,HradecKrálové,CzechRepublic
cCardiacAnaesthesiaandCardiacIntensiveCare,NIHRSouthamptonRespiratoryBiomedicalResearchUnit,SouthamptonUniversityHospitalNHSTrust,
Southampton,UK
dAnaesthesiaandIntensiveCareMedicine,SouthmeadHospital,NorthBristolNHSTrust,Bristol,UK eEmergencyandDisasterMedicine,SixOctoberUniversityHospital,Cairo,Egypt
fDepartmentsofRenalandInternalMedicine,VictoriaHospital,Kirkcaldy,Fife,UK gSocietytoRescuePeoplefromDrowning,Amsterdam,TheNetherlands
hBergenEmergencyMedicalServices,DepartmentofAnaesthesiaandIntensiveCare,HaukelandUniversityHospital,Bergen,Norway iEURACInstituteofMountainEmergencyMedicine,Bozen,Italy
jDepartmentofCardiothoracicSurgery,JamesCookUniversityHospital,Middlesbrough,UK kCenterforEmergencyMedicine,ClinicalHospitalCenterZagreb,Zagreb,Croatia lDepartmentofCardiology,AcademicMedicalCenter,Amsterdam,TheNetherlands
mIntensiveCareMedicineandAnaesthesia,SouthmeadHospital,NorthBristolNHSTrust,Bristol,UK nDepartmentofAnesthesiology,UniversityMedicalCenter,JohannesGutenberg-Universitaet,Mainz,Germany oBartsHeartCentre,StBartholomew’sHospital,BartsHealthNHSTrust,QueenMaryUniversityofLondon,London,UK pDepartmentofAnaesthesiologyandCriticalCareMedicine,UniversityHospitalInnsbruck,Austria
qWarwickMedicalSchool,UniversityofWarwick,Coventry,UK
rCriticalCareUnit,HeartofEnglandNHSFoundationTrust,Birmingham,UK
sDepartmentofAnaesthesiologyandIntensiveCare,CatholicUniversitySchoolofMedicine,Rome,Italy tBirminghamChildren’sHospital,Birmingham,UK
uDepartmentofAnaesthetics,ImperialCollegeHealthcareNHSTrust,London,UK vAnaesthesiaandIntensiveCareMedicine,RoyalUnitedHospital,Bath,UK wSchoolofClinicalSciences,UniversityofBristol,UK
Introduction
Irrespectiveofthecauseofcardiacarrest,earlyrecognitionand callingforhelp,includingappropriatemanagementofthe deteri-oratingpatient,earlydefibrillation,high-qualitycardiopulmonary resuscitation(CPR)withminimalinterruptionofchest compres-sionsandtreatmentofreversiblecauses,arethemostimportant interventions.
In certain conditions, however, advanced life support (ALS) guidelinesrequiremodification.Thefollowingguidelinesfor resus-citation in special circumstances are divided into three parts:
∗ Correspondingauthor.
E-mailaddress:anatolij.truhlar@gmail.com(A.Truhláˇr).
1 ThemembersoftheCardiacarrestinspecialcircumstancessectionCollaborators
arelistedintheCollaboratorssection.
special causes, special environments and special patients. The firstpartcoverstreatmentofpotentiallyreversiblecausesof car-diacarrest,forwhich specifictreatmentexists,andwhichmust beidentified orexcluded duringanyresuscitation. For improv-ingrecallduringALS,thesearedividedintotwogroupsoffour, based upon theirinitialletter –either H or T– and arecalled the‘4Hsand4Ts’:Hypoxia;Hypo-/hyperkalaemiaandother elec-trolyte disorders; Hypo-/hyperthermia; Hypovolaemia; Tension pneumothorax;Tamponade(cardiac);Thrombosis(coronaryand pulmonary);Toxins(poisoning). Thesecondpartcoverscardiac arrestinspecial environments,whereuniversalguidelineshave tobemodifiedduetospecificlocationsorlocation-specificcauses ofcardiacarrest.Thethirdpartisfocusedonpatientswith spe-cificconditions,and thosewithcertainlong-termcomorbidities whereamodifiedapproachanddifferenttreatmentdecisionsmay benecessary.
http://dx.doi.org/10.1016/j.resuscitation.2015.07.017
Summaryofchangessince2010Guidelines
ThemainchangesintheERCGuidelines2015incomparison withtheGuidelines20101aresummarisedbelow:
Specialcauses
• Survivalafteranasphyxia-inducedcardiacarrestisrareand sur-vivorsoftenhavesevereneurologicalimpairment.DuringCPR, earlyeffectiveventilationofthelungswithsupplementary oxy-genisessential.
• Ahighdegreeofclinicalsuspicionandaggressivetreatmentcan preventcardiacarrestfromelectrolyteabnormalities.Thenew algorithmprovidesclinicalguidancetoemergencytreatmentof life-threateninghyperkalaemia.
• Hypothermic patients without signs of cardiac instability (systolic blood pressure ≥90mmHg, absence of ventricular arrhythmiasorcoretemperature≥28◦C)canberewarmed exter-nallyusingminimallyinvasivetechniques(e.g.withwarmforced airandwarmintravenousfluid).Patientswithsignsofcardiac instabilityshouldbetransferreddirectlytoacentrecapableof extracorporeallifesupport(ECLS).
• Earlyrecognitionandimmediatetreatmentwithintramuscular adrenalineremains the mainstayof emergency treatmentfor anaphylaxis.
• Themortalityfromtraumaticcardiacarrest(TCA)isveryhigh. Themostcommoncauseofdeathishaemorrhage.Itisrecognised thatmostsurvivorsdonothavehypovolaemia,butinsteadhave otherreversiblecauses(hypoxia,tensionpneumothorax,cardiac tamponade)thatmustbeimmediatelytreated.Thenew treat-mentalgorithmforTCAwasdevelopedtoprioritisethesequence oflife-savingmeasures.Chestcompressionsshouldnotdelaythe treatmentofreversiblecauses.Cardiacarrestsofnon-traumatic originleadingtoasecondarytraumaticeventshouldbe recog-nisedandtreatedwithstandardalgorithms.
• Thereislimitedevidenceforrecommendingtheroutine trans-portofpatientswithcontinuingCPRafterout-of-hospitalcardiac arrest(OHCA)ofsuspectedcardiacorigin.Transportmaybe ben-eficialin selectedpatientswhere there is immediatehospital accessto thecatheterisation laboratoryand an infrastructure providing prehospital and in-hospital teams experienced in mechanicalorhaemodynamicsupportandpercutaneous coro-naryintervention(PCI)withongoingCPR.
• Recommendationsforadministrationoffibrinolyticswhen pul-monaryembolismisthesuspectedcauseofcardiacarrestremain unchanged.Routineuseofsurgicalembolectomyor mechani-calthrombectomywhenpulmonaryembolismisthesuspected cause of cardiac arrest is not recommended. Consider these methodsonlywhenthere isaknowndiagnosisofpulmonary embolism.
• Routineuseofgastriclavageforgastrointestinal decontamina-tioninpoisoningisnolongerrecommended.Reducedemphasis isplacedonhyperbaricoxygentherapyincarbonmonoxide poi-soning.
Specialenvironments
• Thespecialenvironmentssectionincludesrecommendationsfor treatmentofcardiacarrestoccurringinspecificlocations.These locationsarespecialisedhealthcarefacilities(e.g.operating the-atre,cardiac surgery, catheterisation laboratory, dialysis unit, dentalsurgery),commercialairplanesorairambulances,fieldof play,outsideenvironment(e.g.drowning,difficultterrain,high altitude,avalancheburial,lightningstrikeandelectricalinjuries) orthesceneofamasscasualtyincident.
• Patientsundergoingsurgicalproceduresinvolvinggeneral anaes-thesia,particularlyinemergencies,areatriskfromperioperative
cardiacarrest.Anewsectioncoversthecommoncausesand rel-evantmodificationtoresuscitativeproceduresinthisgroupof patients.
• Cardiacarrestfollowingmajorcardiacsurgeryisrelatively com-monintheimmediatepost-operativephase.Keytosuccessful resuscitationisrecognitionoftheneedtoperformemergency resternotomy,especially inthecontext oftamponadeor hae-morrhage,whereexternalchestcompressionsmaybeineffective. Resternotomyshouldbeperformedwithin5minifother inter-ventionshavefailed.
• Cardiacarrestfromshockablerhythms(VentricularFibrillation (VF) or pulseless Ventricular Tachycardia (pVT)) during car-diaccatheterisationshouldimmediatelybetreatedwithupto three stacked shocks beforestarting chest compressions. Use ofmechanicalchestcompressiondevicesduringangiographyis recommendedtoensure high-qualitychestcompressionsand reducetheradiation burdentopersonnel duringangiography withongoingCPR.
• Indentalsurgery,donotmovethepatientfromthedentalchair inordertostartCPR.Quicklyreclinethedentalchairintoa hor-izontalpositionandplaceastoolundertheheadofthechairto increaseitsstabilityduringCPR.
• Thein-flightuseofAEDsaboardcommercialairplanescanresult inupto50%survivaltohospitaldischarge.AEDsandappropriate CPRequipmentshouldbemandatoryonboardofall commer-cialaircraftinEurope,includingregionalandlow-costcarriers. Consideranover-the-headtechniqueofCPRifrestrictedaccess precludesaconventionalmethod,e.g.intheaisle.
• Theincidenceofcardiacarrestonboardhelicopteremergency medicalservices(HEMS)andairambulancesislow.Importance ofpre-flightpreparationanduseofmechanicalchest compres-siondevicesareemphasised.
• Suddenandunexpectedcollapseofanathleteonthefieldofplay islikelytobecardiacinoriginandrequiresrapidrecognitionand earlydefibrillation.
• Theduration of submersionis a key determinantofoutcome fromdrowning.Submersionexceeding10minisassociatedwith pooroutcome.Bystandersplayacriticalroleinearlyrescueand resuscitation.Resuscitationstrategiesforthoseinrespiratoryor cardiacarrestcontinuetoprioritiseoxygenationandventilation. • The chances of good outcome from cardiac arrest in diffi-cultterrainormountains maybereduced becauseof delayed access andprolonged transport. Thereis a recognisedrole of airrescueandavailabilityofAEDsinremotebutoften-visited locations.
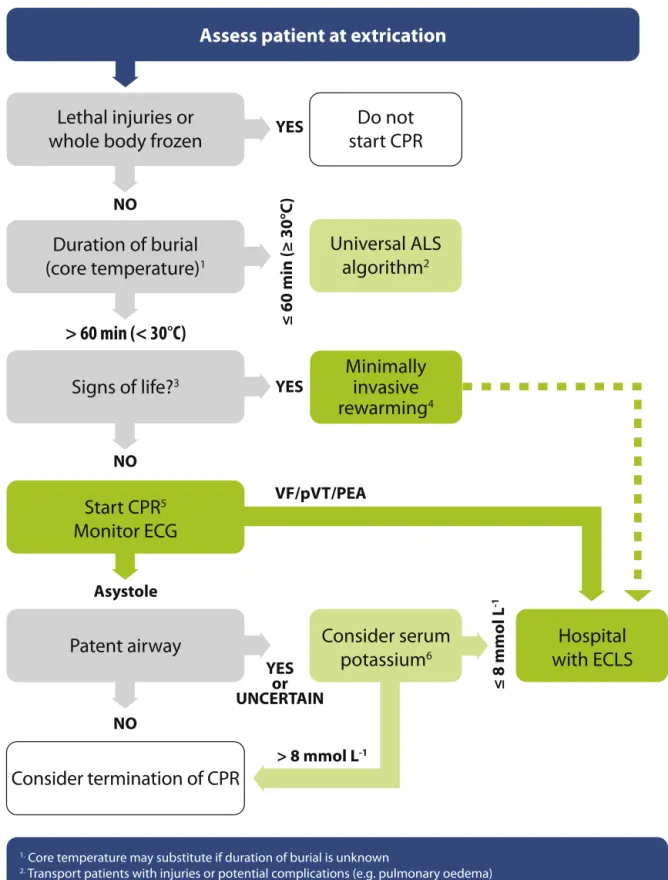
• The cut-off criteria for prolonged CPR and extracorporeal rewarming of avalanche victims in cardiac arrest are more stringenttoreducethenumberoffutilecasestreatedwith extra-corpoereallifesupport(ECLS).ECLSisindicatediftheduration ofburialis >60min(instead of>35min),coretemperatureat extricationis <30◦C (insteadof <32◦C),and serumpotassium athospitaladmissionis≤8mmolL−1(insteadof≤12mmolL−1); otherwisestandardguidelinesapply.
• SafetymeasuresareemphasisedwhenprovidingCPRtothe vic-timofanelectricalinjury.
• Recommendationsformanagementofmultiplevictimsshould preventdelayoftreatmentavailableforsalvageablevictims dur-ingmasscasualtyincidents(MCIs).Safetyatsceneisparamount. Atriagesystemshouldbeusedtoprioritisetreatmentand,ifthe numberofcasualtiesoverwhelmshealthcareresources,withhold CPRforthosewithoutsignsoflife.
Specialpatients
• The section on special patients gives guidance for CPR in patientswithseverecomorbidities(asthma,heartfailurewith
ventricular assist devices, neurological disease, obesity) and thosewithspecificphysiologicalconditions(pregnancy,elderly people).
• Thefirstlinetreatmentforacuteasthmaisinhaledbeta-2 ago-nistswhileintravenousbeta-2agonistsaresuggestedonlyfor thosepatientsinwhominhaledtherapycannotbeusedreliably. Inhaledmagnesiumisnolongerrecommended.
• Inpatientswithventricularassistdevices(VADs),confirmation ofcardiacarrestmaybedifficult.Ifduringthefirst10daysafter surgery,cardiacarrestdoesnotrespondtodefibrillation,perform resternotomyimmediately.
• PatientswithsubarachnoidhaemorrhagemayhaveECGchanges thatsuggestanacutecoronarysyndrome(ACS).Whethera com-putedtomography(CT)brainscanisdonebeforeoraftercoronary angiographywilldependonclinical judgementregarding the likelihoodofasubarachnoidhaemorrhageversusacutecoronary syndrome.
• Nochangestothesequenceofactionsarerecommendedin resus-citationofobesepatients,althoughdeliveryofeffectiveCPRmay bechallenging.Considerchangingrescuersmorefrequentlythan thestandard2-mininterval.Earlytrachealintubationbyan expe-riencedproviderisrecommended.
• Forthepregnantwomanincardiacarrest,high-qualityCPRwith manualuterinedisplacement,earlyALSanddeliveryofthefetus ifearlyreturnofspontaneouscirculation(ROSC)isnotachieved remainkeyinterventions.
A–SPECIALCAUSES Hypoxia
Introduction
Cardiacarrestcausedbypurehypoxaemiaisuncommon.Itis seenmorecommonlyasaconsequenceofasphyxia,whichaccounts formostofthenon-cardiaccausesofcardiacarrest.Therearemany causesof asphyxialcardiacarrest(Table4.1); althoughthere is usuallya combinationof hypoxaemiaand hypercarbia,it isthe hypoxaemiathatultimatelycausescardiacarrest.2
Pathophysiologicalmechanisms
Ifbreathingiscompletelypreventedbyairwayobstructionor apnoea, consciousness will be lost when oxygen saturation in thearterial blood reachesabout 60%. The time taken toreach thisconcentrationisdifficulttopredict,butislikelytobeofthe order 1–2min.3 Based onanimal experiments of cardiac arrest
causedbyasphyxia,pulselesselectricalactivity(PEA)willoccur in3–11min.Asystolewillensueseveralminuteslater.4In
compar-isonwithsimpleapnoea,theexaggeratedrespiratorymovements thatfrequentlyaccompanyairwayobstructionwillincrease oxy-genconsumption resultingin morerapid arterialbloodoxygen desaturationand a shorter time tocardiac arrest. Accordingto Table4.1
Causesofasphyxialcardiacarrest
Airwayobstruction:softtissues(coma),laryngospasm,aspiration Anaemia
Asthma Avalancheburial
Centralhypoventilation–brainorspinalcordinjury Chronicobstructivepulmonarydisease
Drowning Hanging Highaltitude
Impairedalveolarventilationfromneuromusculardisease Pneumonia
Tensionpneumothorax Trauma
Traumaticasphyxiaorcompressionasphyxia(e.g.crowdcrush)
Safar,completeairwayobstructionafterbreathingairwillresult
inPEAcardiacarrestin5–10min.2VFisrarelythefirstmonitored
rhythmafterasphyxialcardiacarrest–inoneofthelargestseriesof hanging-associatedout-of-hospitalcardiacarrests(OHCAs),from Melbourne,Australia,just7(0.5%)of1321patientswereinVF.5
Treatment
Treatingthecauseoftheasphyxia/hypoxaemiaisthehighest prioritybecausethisisapotentiallyreversiblecauseofthe car-diacarrest.Effectiveventilationwithsupplementaryoxygenisa particularpriorityinthesepatients.ThebetteroutcomesforOHCA victimsreceivingcompression-onlyCPR6isnotthecasefor
asphyx-ialcardiac arrests,which have much better survival rates with conventionalCPR.7FollowthestandardALSalgorithmwhen
resus-citatingthesepatients. Outcome
Survivalaftercardiacarrestfromasphyxiaisrareandmost sur-vivorssustainsevereneurologicalinjury.Offivepublishedseries thatincludedatotal of286patientswithcardiacarrest follow-ing hanging where CPR wasattempted (this wasattempted in onlyabout16%ofcases),therewerejustsix(2%)survivorswith afullrecovery;11othersurvivorsallhadseverepermanentbrain injury.5,8–11Inonethird(89;31%)ofthese286patients,rescuers
wereabletoachieveROSC–thuswhenCPRisattempted,ROSC is notuncommonbut subsequentneurologicallyintact survival israre.Thosewhoareunconsciousbuthavenotprogressedtoa cardiacarrestaremuchmorelikelytomakeagoodneurological recovery.11,12
Hypo-/hyperkalaemiaandotherelectrolytedisorders Introduction
Electrolyte abnormalities can cause cardiac arrhythmias or cardiacarrest.Life-threateningarrhythmias areassociatedmost commonlywithpotassiumdisorders,particularlyhyperkalaemia, and less commonly with disorders of serum calcium and magnesium.Consider electrolytedisturbancesin patientgroups atrisk–renalfailure,severeburns,cardiacfailureanddiabetes mellitus.
Theelectrolyte values for definitions havebeen chosenas a guidetoclinicaldecision-making.Theprecisevaluesthattrigger treatmentdecisionswilldependonthepatient’sclinicalcondition andrateofchangeofelectrolytevalues.Thereislittleorno evi-denceforthetreatmentofelectrolyteabnormalitiesduringcardiac arrest.Guidanceduringcardiacarrest isbasedonthestrategies usedinthenon-arrestpatient.
Preventionofelectrolytedisorders
Whenpossible,identifyandtreat life-threateningelectrolyte abnormalitiesbeforecardiacarrestoccurs.Monitorrenalfunction inpatientsatriskandavoidcombinationofdrugsthatmay exac-erbatehyperkalaemia.Preventrecurrenceofelectrolytedisorders byremovinganyprecipitatingfactors(e.g.drugs,diet).
Potassiumdisorders
Potassium homeostasis. Extracellularpotassium concentration is regulated tightly between 3.5 and 5.0mmolL−1. A large con-centration gradient normally exists between intracellular and extracellularfluidcompartments.Thispotassiumgradientacross cellmembranescontributestotheexcitabilityofnerveandmuscle cells,includingthemyocardium.Evaluationofserumpotassium musttakeintoconsiderationtheeffectsofchangesinserumpH. WhenserumpHdecreases(acidaemia),serumpotassiumincreases becausepotassiumshiftsfromthecellulartothevascularspace;a processthatisreversedwhenserumpHincreases(alkalaemia).
Hyperkalaemia. Thisisthemostcommonelectrolytedisorder asso-ciatedwithcardiacarrest.Itisusuallycausedbyimpairedexcretion bythekidneys,drugsorincreasedpotassiumrelease fromcells and metabolic acidosis. Hyperkalaemia occurs in up to 10% of hospitalisedpatients.13–15 Chronickidneydisease(CKD)is
com-moninthegeneralpopulationandtheincidenceofhyperkalaemia increasesfrom2to42%asglomerularfiltrationrate(GFR)drops from60to20mLmin−1.16 Patientswithend-stagerenaldisease
areparticularlysusceptible,particularlyfollowinganOHCA.17
Pro-longedhyperkalaemiaisanindependentriskfactorforin-hospital mortality.18 Acute hyperkalaemia is more likely than chronic
hyperkalaemia tocause life-threatening cardiac arrhythmias or cardiacarrest.
Definition. Thereisnouniversaldefinition.Wehavedefined hyperkalaemiaasa serumpotassiumconcentrationhigherthan 5.5mmolL−1; inpractice,hyperkalaemiais acontinuum.Asthe potassium concentration increases above this value the risk of adverse events increases and the need for urgent treatment increases.Severehyperkalaemiahasbeendefinedasaserum potas-siumconcentrationhigherthan6.5mmolL−1.
Causes. Themaincausesofhyperkalaemiaare:
• renalfailure(i.e.acutekidneyinjuryorchronickidneydisease); • drugs (e.g. angiotensin converting enzyme inhibitors (ACE-I), angiotensin II receptor antagonists (ARB), potassium-sparing diuretics,non-steroidalanti-inflammatorydrugs,beta-blockers, trimethoprim);
• tissuebreakdown(e.g.rhabdomyolysis,tumourlysis, haemoly-sis);
• metabolicacidosis(e.g.renalfailure,diabeticketoacidosis); • endocrinedisorders(e.g.Addison’sdisease);
• diet(maybesolecauseinpatientswithadvancedchronickidney disease)and
• spurious–pseudo-hyperkalaemia(suspectincaseswithnormal renalfunction,normalECGand/orhistoryofhaematological dis-order).Pseudo-hyperkalaemiadescribesthefindingofaraised serum (clotted blood) K+ value concurrently with a normal plasma(non-clottedblood)potassiumvalue.Theclottingprocess releasesK+fromcellsandplatelets,whichincreasestheserum K+concentrationbyanaverageof0.4mmol/L.Themostcommon causeofpseudo-hyperkalaemiaisaprolongedtransittimetothe laboratoryorpoorstorageconditions.19,20
The risk of hyperkalaemia is even greater when there is a combinationoffactorssuchastheconcomitantuseof angiotensin-convertingenzymeinhibitorsorangiotensinIIreceptorblockers andpotassium-sparingdiuretics.
Recognition of hyperkalaemia. Exclude hyperkalaemia in all patientswithanarrhythmiaorcardiacarrest.Patientsmaypresent withweaknessprogressing toflaccid paralysis,paraesthesia,or depresseddeeptendonreflexes.Alternatively,theclinicalpicture can be overshadowed by the primary illness causing hyper-kalaemia.The firstindicator ofhyperkalaemia may alsobethe presenceofECGabnormalities,arrhythmias,orcardiacarrest.The useofabloodgasanalysertomeasurepotassiumcanreducedelays inrecognition.21,22
TheeffectofhyperkalaemiaontheECGdependsonthe abso-luteserumpotassiumaswellastherateofincrease.23Thereported
frequencyofECGchangesinseverehyperkalaemiaisvariable,but mostpatientsappeartoshowECGabnormalitiesataserum potas-siumconcentrationhigherthan6.7mmolL−1.23,24Thepresenceof
ECGchangesstronglycorrelateswithmortality.25Insomecases,
theECG maybenormal orshowatypical changes includingST elevation.
TheECG changes associated withhyperkalaemiaare usually progressiveandinclude:
• firstdegreeheartblock(prolongedPRinterval>0.2s); • flattenedorabsentPwaves;
• tall,peaked(tented)Twaves(i.e.TwavelargerthanRwavein morethan1lead);
• ST-segmentdepression;
• S&Twavemerging(sinewavepattern); • widenedQRS(>0.12s);
• ventriculartachycardia; • bradycardia;
• cardiacarrest(PEA,VF/pVT,asystole).
Treatment of hyperkalaemia. There are five key treatment strategiesforhyperkalaemia22:
• cardiacprotection;
• shiftingpotassiumintocells; • removingpotassiumfromthebody;
• monitoringserumpotassiumandbloodglucose; • preventionofrecurrence.
Whenhyperkalaemiaisstronglysuspected,e.g.inthepresence ofECGchanges,startlife-savingtreatmentevenbeforelaboratory resultsare available. The treatmentstrategy for hyperkalaemia hasbeenreviewedextensively.13,22,26Followthehyperkalaemia
emergency treatment algorithm (Fig. 4.1).22 Avoid salbutamol
monotherapy,whichmaybeineffective.Thereisinsufficient evi-dencetosupporttheuseofsodiumbicarbonatetodecreaseserum potassium.Consider theneedforearlyspecialistorcriticalcare referral.
Themainrisksassociatedwithtreatmentofhyperkalaemiaare: • Hypoglycaemia followinginsulin-glucose administration (usu-allyoccurswithin1–3hoftreatment,butmayoccurupto6h afterinfusion).27Monitorbloodglucoseandtreathypoglycaemia
promptly.
• Tissuenecrosissecondarytoextravasationofintravenous cal-ciumsalts.Ensuresecurevascularaccesspriortoadministration. • Intestinal necrosis or obstruction following use of potassium exchangeresins.Avoidprolongeduseofresinsandgivelaxative. • Reboundhyperkalaemiaaftertheeffectofdrugtreatmenthas wornoff(i.e.within4–6h).Continuetomonitorserumpotassium foraminimumof24hafteranepisode.
Patientnotincardiacarrest Assesspatient:
• UsesystematicABCDEapproachandcorrectanyabnormalities, obtainIVaccess.
• Checkserumpotassium. • RecordanECG.
Monitorcardiacrhythminpatientswithseverehyperkalaemia. Treatmentisdeterminedaccordingtoseverityofhyperkalaemia. Approximate values are provided to guide treatment. Follow hyperkalaemiaemergencytreatmentalgorithm(Fig.4.1).
Mildelevation(5.5–5.9mmolL−1).
• Addresscauseofhyperkalaemiatocorrectandavoidfurtherrise inserumpotassium(e.g.drugs,diet).
• If treatment is indicated, remove potassium from the body: potassiumexchangeresins-calciumresonium15–30g,orsodium polystyrenesulfonate(Kayexalate)15–30g,giveneitherorallyor byretentionenema/PR(perrectum)(onsetin>4h).
Moderateelevation(6.0–6.4mmolL−1)withoutECGchanges. • Shift potassium intracellularly with glucose/insulin: 10 units
short-actinginsulinand25gglucoseIVover15–30min(onset in15–30min;maximaleffectat30–60min;durationofaction 4–6h;monitorbloodglucose).
• Removepotassiumfromthebody(seeabove;considerdialysis guidedbyclinicalsetting).
Fig.4.1.Emergencytreatmentofhyperkalaemia.PRperrectum;ECGelectrocardiogram;VTventriculartachycardia. ReproducedwithpermissionfromRenalAssociationandResuscitationCouncil(UK).
Severeelevation(≥6.5mmolL−1)withoutECGchanges. • Seekexperthelp.
• Giveglucose/insulin(seeabove).
• Givesalbutamol10–20mgnebulised(onsetin15–30min; dura-tionofaction4–6h).
• Removepotassiumfromthebody(considerdialysis). Severeelevation(≥6.5mmolL−1)withtoxicECGchanges. • Seekexperthelp.
• Protect the heart with calcium chloride: 10mL 10% cal-cium chloride IV over 2–5min to antagonise the toxic effects of hyperkalaemia at the myocardial cell mem-brane. This protects the heart by reducing the risk of VF/pVT but does not lower serum potassium (onset in 1–3min).
• Useshiftingagents(glucose/insulinandsalbutamol).
• Removepotassiumfromthebody(considerdialysisatoutsetor ifrefractorytomedicaltreatment).
Modificationstocardiopulmonaryresuscitation. Thefollowing modificationstostandardALSguidelinesarerecommendedinthe presenceofseverehyperkalaemia:
• Confirmhyperkalaemiausingabloodgasanalyserifavailable. • Protecttheheart:give10mLcalciumchloride10%IVbyrapid
bolusinjection.
• Shiftpotassiumintocells:Giveglucose/insulin:10units short-actinginsulin and 25gglucoseIV byrapid injection.Monitor bloodglucose.
• Givesodiumbicarbonate:50mmolIVbyrapidinjection(ifsevere acidosisorrenalfailure).
• Remove potassium from body: Consider dialysis for hyper-kalaemiccardiacarrestresistanttomedicaltreatment.Several dialysismodalitieshavebeenusedsafelyandeffectivelyin car-diacarrest,butthismayonlybeavailableinspecialistcentres.28
Consideruseofamechanicalchestcompressiondeviceif pro-longedCPRisneeded.
Indications for dialysis. The main indications for dialysis in patientswithhyperkalaemiaare:
• severe life-threatening hyperkalaemia with or without ECG changesorarrhythmia;
• hyperkalaemiaresistanttomedicaltreatment; • end-stagerenaldisease;
• oliguricacutekidneyinjury(<400mLday−1urineoutput); • markedtissuebreakdown(e.g.rhabdomyolysis).
Specialconsiderationsformanagement ofcardiacarrestin a dialysisunitareaddressedinthesectionSpecialenvironments(see cardiacarrestinadialysisunit).
Hypokalaemia. Hypokalaemiaisthemostcommonelectrolyte dis-orderinclinicalpractice.29Itisseeninupto20%ofhospitalised
patients.30Hypokalaemiaincreasestheincidenceofarrhythmias
andsuddencardiacdeath(SCD).31Theriskisincreasedinpatients
withpre-existingheartdiseaseandinthosetreatedwithdigoxin. Definition. Hypokalaemiaisdefinedasaserumpotassiumlevel <3.5mmolL−1.Severehypokalaemiaisdefinedasaserum potas-siumlevel<2.5mmolL−1andmaybeassociatedwithsymptoms.
Causes. Themaincausesofhypokalaemiainclude: • gastrointestinalloss(e.g.diarrhoea);
• drugs(e.g.diuretics,laxatives,steroids);
• renallosses(e.g.renaltubulardisorders,diabetesinsipidus, dial-ysis);
• endocrinedisorders (e.g.Cushing’s syndrome, hyperaldostero-nism);
• metabolicalkalosis; • magnesiumdepletion; • poordietaryintake.
Treatmentstrategiesusedforhyperkalaemiamayalsoinduce hypokalaemia.
Recognitionofhypokalaemia. Excludehypokalaemiainevery patientwithanarrhythmiaorcardiacarrest.Indialysispatients, hypokalaemiamayoccurattheendofahaemodialysissessionor duringtreatmentwithperitonealdialysis.
Asserumpotassium concentrationdecreases,thenervesand muscles are predominantly affected,causing fatigue, weakness, leg cramps, constipation. In severe cases (serum potassium <2.5mmolL−1),rhabdomyolysis,ascendingparalysisand respira-torydifficultiesmayoccur.
ECGfeaturesofhypokalaemiaare: • Uwaves;
• Twaveflattening; • STsegmentchanges;
• arrhythmias,especiallyifpatientistakingdigoxin; • cardiacarrest(PEA,VF/pVT,asystole).
Treatment. This depends on the severity of hypokalaemia and thepresenceof symptomsand ECG abnormalities.Gradual replacementofpotassiumispreferable,butinanemergency, intra-venous potassium is required. The maximumrecommended IV doseofpotassiumis 20mmolh−1,but morerapidinfusion(e.g. 2mmolmin−1for10min,followedby10mmolover5–10min)is indicatedforunstablearrhythmiaswhencardiacarrestis immi-nent.ContinuousECGmonitoringisessentialduringIVinfusion andthedoseshouldbetitratedafterrepeatedsamplingofserum potassiumlevels.
Manypatientswhoarepotassiumdeficientarealsodeficient inmagnesium.Magnesiumisimportantforpotassiumuptakeand forthemaintenanceofintracellularpotassiumvalues,particularly inthemyocardium.Repletionofmagnesiumstoreswillfacilitate morerapid correctionofhypokalaemiaandis recommendedin severecasesofhypokalaemia.32
Calciumandmagnesiumdisorders
Therecognitionandmanagementofcalciumandmagnesium disordersissummarisedinTable4.2.
Hypo-/hyperthermia Accidentalhypothermia
Definition. Every year approximately 1500 people die of pri-maryaccidental hypothermia intheUnitedStates.33 Accidental
hypothermiaisdefinedasaninvoluntarydropofthebodycore temperature<35◦C.TheSwissstagingsystemisusedtoestimate coretemperatureatthescene.Itsstagesarebasedonclinicalsigns, whichroughlycorrelatewiththecoretemperature:
• hypothermia I; mild hypothermia (conscious, shivering, core temperature35–32◦C);
• hypothermiaII;moderatehypothermia(impairedconsciousness withoutshivering,coretemperature32–28◦C);
• hypothermiaIII;severehypothermia(unconscious,vitalssigns present,coretemperature28–24◦C);
• hypothermiaIV;cardiacarrestorlowflowstate(noorminimal vitalsigns,coretemperature<24◦C);
• hypothermiaV;deathduetoirreversiblehypothermia(core tem-perature<13.7◦C).34
Diagnosis. Hypothermiaisdiagnosed inanypatientwitha core temperature<35◦C,orwheremeasurementunavailable,ahistory of exposuretocold, orwhen thetrunkfeels cold.33 Accidental
hypothermiamaybeunder-diagnosedincountrieswitha temper-ateclimate.Whenthermoregulationisimpaired,forexample,in theelderlyandveryyoung,hypothermiamayfollowamildinsult. Theriskofhypothermiaisincreasedbyalcoholordrugingestion, exhaustion, illness,injury or neglectespecially whenthere is a decreaseinthelevelofconsciousness.
Alow-readingthermometerisneededtomeasurethecore tem-peratureandconfirmthediagnosis.Thecoretemperatureinthe lower third of the oesophagus correlates wellwithheart tem-perature. Tympanic measurement usinga thermistor technique isareliablealternativebutmaybeconsiderablylowerthancore temperature if the environment is very cold, the probe is not wellinsulated,ortheexternalauditorycanalisfilledwithsnow orwater35,36Widelyavailabletympanicthermometersbasedon
infraredtechniquedonotsealtheearcanalandarenotdesignedfor lowcoretemperaturereadings.37Thein-hospitalcoretemperature
measurement site shouldbethesamethroughoutresuscitation andrewarming.Bladderandrectaltemperatureslagbehindcore temperature;38,39 for this reason, measurement of bladder and
Table4.2
Calciumandmagnesiumdisorderswithassociatedclinicalpresentation,ECGmanifestationsandrecommendedtreatment
Disorder Causes Presentation ECG Treatment
Hypercalcaemia
Calcium>2.6mmolL−1 Primaryortertiary
hyperparathyroidism Malignancy Sarcoidosis Drugs Confusion Weakness Abdominalpain Hypotension Arrhythmias Cardiacarrest ShortQTinterval ProlongedQRSinterval FlatTwaves AVblock Cardiacarrest FluidreplacementIV Furosemide1mgkg−1IV Hydrocortisone200–300mgIV Pamidronate30–90mgIV Treatunderlyingcause
Hypocalcaemia
Calcium<2.1mmolL−1 Chronicrenalfailure Acutepancreatitis Calciumchannelblocker overdose
Toxicshocksyndrome Rhabdomyolysis Tumourlysissyndrome
Paraesthesia Tetany Seizures AV-block Cardiacarrest ProlongedQTinterval Twaveinversion Heartblock Cardiacarrest Calciumchloride10%10–40mL Magnesiumsulphate50% 4–8mmol(ifnecessary)
Hypermagnesaemia
Magnesium>1.1mmolL−1 Renalfailure
Iatrogenic Confusion Weakness Respiratorydepression AV-block Cardiacarrest ProlongedPRandQT intervals Twavepeaking AVblock Cardiacarrest
Considertreatmentwhen magnesium>1.75mmolL−1
Calciumchloride10%5–10mL repeatedifnecessary Ventilatorysupportifnecessary Salinediuresis–0.9%salinewith furosemide1mgkg−1IV
Haemodialysis Hypomagnesaemia
Magnesium<0.6mmolL−1 GIloss
Polyuria Starvation Alcoholism Malabsorption Tremor Ataxia Nystagmus Seizures Arrhythmias–torsade depointes Cardiacarrest ProlongedPRandQT intervals ST-segmentdepression T-waveinversion FlattenedPwaves IncreasedQRSduration Torsadedepointes Severeorsymptomatic:2g50% magnesiumsulphate(4mL; 8mmol)IVover15min Torsadedepointes:2g50% magnesiumsulphate(4mL; 8mmol)IVover1–2min Seizure:2g50%magnesium sulphate(4mL;8mmol)IVover 10min
rectaltemperaturehasbeende-emphasisedinpatientswithsevere
hypothermia.
Decisiontoresuscitate. Coolingofthehumanbodydecreases
cel-lular oxygen consumption by about 6% per 1◦C decrease in
coretemperature.40At28◦C,oxygenconsumptionisreducedby
approximately50%andat22◦Cbyapproximately75%.At18◦Cthe braincantoleratecardiacarrestforupto10timeslongerthanat 37◦C.Thisresultsinhypothermiaexertingaprotectiveeffectonthe brainandheart,41andintactneurologicalrecoverymaybepossible
evenafterprolongedcardiacarrestifdeephypothermiadevelops beforeasphyxia.
Bewareofdiagnosingdeathinahypothermicpatientbecause hypothermia itself may produce a very slow, small-volume, irregular pulse and unrecordable blood pressure. In a deeply hypothermicpatient(hypothermiaIV)signsoflifemaybeso mini-malthatitiseasytooverlookthem.Therefore,lookforsignsoflife foratleast1minanduseanECGmonitortodetectanyelectrical cardiacactivity.Neurologicallyintactsurvivalhasbeenreported afterhypothermiccardiacarrestwithacoretemperatureaslowas 13.7◦C42andCPRforaslongassixandahalfhours.43
IntermittentCPR,asrescueallows,mayalsobeofbenefit.44If
continuousCPRcannotbedelivered,apatientwithhypothermic cardiacarrestandacoretemperature<28◦C(orunknown),should receive5minofCPR,alternatingwithperiods≤5minwithoutCPR. Patientswithacoretemperature<20◦C,shouldreceive5minof CPR,alternatingwithperiods≤10minwithoutCPR.45
Intheprehospitalsetting,resuscitationshouldbewithheldin hypothermicpatientsonlyifthecauseofcardiacarrestisclearly attributabletoalethalinjury,fatalillness,prolongedasphyxia,or ifthechestisincompressible.46Inallotherhypothermicpatients,
thetraditionalguidingprinciplethat‘nooneisdeaduntilwarm anddead’shouldbeconsidered.Inremoteareas,the impracticali-tiesofachievingrewarminghavetobeconsidered.Inthehospital settinginvolveseniordoctorsanduseclinicaljudgementto deter-minewhentostopresuscitatingahypothermicvictimincardiac arrest.
Modificationstocardiopulmonaryresuscitation
• Donotdelaycarefultrachealintubationwhenitisindicated.The advantagesofadequateoxygenationandprotectionfrom aspi-rationoutweightheminimalriskoftriggeringVFbyperforming trachealintubation.47
• Checkforsignsoflifeforupto1min.Palpateacentralarteryand assessthecardiacrhythm(ifECGmonitoravailable). Echocardi-ography,near-infraredspectroscopyorultrasoundwithDoppler maybeusedtoestablishwhetherthereis(anadequate)cardiac outputorperipheralbloodflow.48,49Ifthereisanydoubt,start
CPRimmediately.
• Hypothermiacancausestiffnessofthechestwall,making ven-tilationsand chestcompressionsdifficult.Considertheuseof mechanicalchestcompressiondevices.50
• OnceCPRisunderway,confirmhypothermiawithalow-reading thermometer.
• The hypothermic heart may be unresponsive to cardioac-tivedrugs,attemptedelectricalpacinganddefibrillation.Drug metabolismisslowed,leadingtopotentiallytoxicplasma con-centrationsofanydruggiven.51 Theevidencefor theefficacy
of drugs in severe hypothermia is limited and based mainly onanimalstudies.Forinstance,inseverehypothermiccardiac arrest,theefficacyofamiodaroneisreduced.52Adrenalinemay
survival.53,54Vasopressorsmayalsoincreasethechancesof
suc-cessfuldefibrillation,butwithacoretemperature<30◦C,sinus rhythmoften degradesbackintoVF.Given that defibrillation andadrenalinemayinducemyocardialinjury, itisreasonable towithholdadrenaline,other CPRdrugs andshocksuntilthe patienthasbeenwarmedtoacoretemperature≥30◦C.Once 30◦Chasbeenreached,theintervalsbetweendrugdosesshould bedoubledwhen compared to normothermia(i.e.adrenaline every6–10min).Asnormothermia(≥35◦C)isapproached,use standarddrugprotocols.
Treatmentof arrhythmias. As coretemperature decreases,sinus bradycardia tendstogive way toatrial fibrillationfollowed by VFand finallyasystole.55,56 Arrhythmias otherthan VFtendto
revertspontaneouslyascoretemperatureincreases,andusually donotrequireimmediatetreatment.Bradycardiaisphysiologicalin severehypothermia.Cardiacpacingisnotindicatedunless brady-cardiaassociatedwithhaemodynamiccompromisepersistsafter rewarming.Thetemperatureatwhichdefibrillationshouldfirstly beattempted,andhowoftenitshouldbeattemptedintheseverely hypothermicpatient,hasnotbeenestablished.IfVFisdetected, defibrillateaccordingtostandardprotocols.IfVFpersistsafterthree shocks,delayfurtherattemptsuntilcoretemperatureis≥30◦C.57
CPRandrewarmingmayhavetobecontinuedforseveralhoursto facilitatesuccessfuldefibrillation.
Insulation. Generalmeasuresforallvictimsincluderemovalfrom thecoldenvironment, preventionof furtherheatlossandrapid transfertohospital.58Inthefield,apatientwithmoderateorsevere
hypothermia(hypothermia≥II)shouldbeimmobilisedand han-dledcarefully,oxygenatedadequately,monitored(includingECG andcoretemperature),andthewholebodydriedandinsulated.51
Removewetclotheswhileminimisingexcessivemovementof thevictim. Removalof wetclothing oruseof a vapour barrier seemstobeequallyeffectivetolimitheatloss.59Conscious
vic-tims(hypothermiaI)canmobiliseasexerciserewarmsaperson morerapidlythanshivering.60Patientswillcontinuecoolingafter
removalfromacoldenvironment(i.e.afterdrop),whichmayresult inalife-threateningdecreaseincoretemperaturetriggeringa car-diacarrestduringtransport(i.e.‘rescuedeath’).Prehospitally,avoid prolongedinvestigationsandtreatment,asfurtherheatlossis diffi-culttoprevent.Patientswhostopshivering(e.g.hypothermiaII–IV, andsedatedoranaesthetisedpatients)willcoolfaster.
Prehospitalrewarming. Rewarmingmaybepassive,activeexternal, oractiveinternal.InhypothermiaIpassiverewarmingis appropri-ateaspatientsarestillabletoshiver.Passiverewarmingisbest achievedbyfullbodyinsulationwithwoolblankets,aluminium foil,capandawarmenvironment.InhypothermiaII–IVthe appli-cationofchemicalheatpackstothetrunkhasbeenrecommended. Inconsciouspatientswhoareabletoshiver,thisimprovesthermal comfortbutdoesnotspeedrewarming.61Ifthepatientis
uncon-sciousandtheairwayisnotsecured,arrangetheinsulationaround thepatientlyinginarecovery(lateraldecubitus)position. Rewarm-inginthefieldwithheatedintravenousfluidsandwarmhumidified gasesisnotfeasible.51Intensiveactiverewarmingmustnotdelay
transport toa hospital where advanced rewarming techniques, continuousmonitoringandobservationareavailable.
Transport. Transport patients with hypothermia stage I to the nearesthospital.InhypothermiastageII–IV,signsofprehospital cardiacinstability(i.e.systolicbloodpressure<90mmHg, ventri-culararrhythmia,coretemperature<28◦C)shoulddeterminethe choiceofadmittinghospital.Ifanysignsofcardiacinstabilityare present,transportthepatienttoanECLScentre,contactingthem wellinadvancetoensurethatthehospitalcanacceptthepatient
forextracorporealrewarming.InhypothermiaV,reasonsfor with-holdingorterminatingCPRshouldbeinvestigated(e.g.obvious signsofirreversibledeath,validDNAR,conditionsunsafefor res-cuer,avalancheburial≥60minandairwaypackedwithsnowand asystole).Intheabsenceofanyofthesesigns,startCPRandtransfer thepatienttoanECLScentre.
In-hospitalrewarming. Unless thepatient goesinto VF, rewarm using active external methods (i.e. with forced warm air) and minimallyinvasivelymethods(i.e.withwarmIVinfusions).With acoretemperature<32◦C andpotassium <8mmolL−1,consider ECLSrewarming.33MostECLSrewarmingshavebeenperformed
usingcardiopulmonarybypass, butmore recently,veno-arterial extracorporealmembraneoxygenation(VA-ECMO)hasbecomethe preferredmethodduetoitsrapidavailability,theneedforless anti-coagulation,andthepotentialtoprolongcardiorespiratorysupport afterrewarming.
IfanECLScentreisnotavailable,rewarmingmaybeattempted inhospitalusingadedicatedteamandacombinationofexternal andinternalrewarming techniques(e.g.forcedwarmair,warm infusions,forcedperitoneallavage).62
Continuous haemodynamic monitoring and warm IV fluids areessential.Patientswillrequirelargevolumesoffluidsduring rewarming,asvasodilationcausesexpansionoftheintravascular space.Avoidhyperthermiaduringandafterrewarming.OnceROSC hasbeenachievedusestandardpost-resuscitationcare.
Hyperthermia
Introduction. Hyperthermiaoccurswhenthebody’sabilityto ther-moregulate fails and core temperature exceeds that normally maintainedby homeostaticmechanisms.Hyperthermiamay be exogenous,causedbyenvironmentalconditions,orsecondaryto endogenousheatproduction.
Environment-relatedhyperthermiaoccurswhereheat,usually intheformofradiantenergy,isabsorbedbythebodyataratefaster thancanbelostbythermoregulatorymechanisms.Hyperthermia isacontinuumofheat-relatedconditions,startingwithheatstress, progressingtoheatexhaustion,thentoheatstrokeandfinallyto multipleorgandysfunctionandcardiacarrest.63
Malignant hyperthermia is a rare disorder of skeletal mus-clecalciumhomeostasischaracterisedbymusclecontractureand life-threateninghypermetaboliccrisisfollowingexposureof genet-ically predisposed individuals to halogenated anaesthetics and depolarisingmusclerelaxants.64,65
Heatexhaustion
Definition. Heatexhaustion isa non-life-threatening clinical syndromeofweakness,malaise,nausea,syncope,andother non-specificsymptomscausedbyheatexposure.Thermoregulationis notimpaired.Heatexhaustioniscausedbywaterandelectrolyte imbalanceduetoheatexposure,withorwithoutexertion.Rarely, severeheatexhaustionafterphysicalexertionmaybecomplicated byrhabdomyolysis,myoglobinuria,acuterenalfailure,and dissem-inatedintravascularcoagulation(DIC).
Symptoms. Symptomsareoftenvague,andpatientsmaynot realisethatheatisthecause.Symptomsmayincludeweakness, dizziness,headache,nausea,andsometimesvomiting.Syncopedue tostandingforlongperiodsintheheat(heatsyncope)iscommon andmaymimiccardiovasculardisorders.Onexamination,patients appeartiredandareusuallysweatyandtachycardic.Mentalstatus istypicallynormal,unlikeinheatstroke.Temperatureisusually normaland,whenelevated,usuallydoesnotexceed40◦C.
Diagnosis. Diagnosisisclinicalandrequiresexclusionofother possible causes (e.g. hypoglycaemia, acute coronary syndrome, infections).Laboratorytestingisrequiredonlyifneededtorule outotherdisorders.
Treatment
Fluidsandelectrolytereplacement. Treatmentinvolves remov-ingpatientstoacoolenvironment,lyingthemflat,andgivingIV fluidsandelectrolytereplacementtherapy;oralrehydrationmay notbeeffectiveinrapidlyreplacingelectrolytes,butmaybeamore practicaltreatment.Rateandvolumeofrehydrationareguidedby age,underlyingdisorders, andclinicalresponse.Replacementof 1–2Lcrystalloidsat500mLh−1 isoftenadequate.External cool-ingmeasuresareusuallynotrequired.Considerexternalcoolingin patientswithacoretemperatureof≥40◦C.
Heatstroke
Definition. Heat stroke (HS) is defined as hyperthermia accompaniedbya systemic inflammatory response witha core temperature>40◦C,accompaniedbymentalstatechangeand vary-inglevelsoforgandysfunction.63
TherearetwoformsofHS:
1.Classic(non-exertional)heatstroke(CHS)occursduringhigh environmentaltemperaturesandofteneffectstheelderlyduring heatwaves.66
2.Exertionalheat stroke (EHS) occurs during strenuous physi-calexercisein highenvironmentaltemperaturesand/orhigh humidityandusuallyeffectshealthyyoungadults.67
Mortalityfromheatstrokerangesbetween10and50%.68
Predisposing factors. The elderly are at increased risk for heat-related illness because of underlying illness, medication use, decliningthermoregulatorymechanisms and limited social support.Thereareseveralriskfactors:lackofacclimatisation, dehy-dration,obesity, alcohol,cardiovascular disease,skinconditions (psoriasis, eczema, scleroderma, burn, cystic fibrosis), hyper-thyroidism, phaeochromocytoma and drugs (anticholinergics, diamorphine, cocaine, amphetamine, phenothiazines, sympath-omimetics,calciumchannelblockers,beta-blockers).
Symptoms. Heat strokecan resembleseptic shockand may becausedby similarmechanisms.69 Asinglecentre case series
reported14ICUdeathsin22heatstrokepatientsadmittedtoICU withmultipleorganfailure.70Featuresincluded:
• coretemperature≥40◦C;
• hot,dryskin(sweatingpresentinabout50%ofcasesofexertional heatstroke);
• earlysignsandsymptoms(e.g.extremefatigue,headache, faint-ing,facialflushing,vomitinganddiarrhoea);
• cardiovascular dysfunction including arrhythmias and hypotension71;
• respiratorydysfunctionincludingacuterespiratorydistress syn-drome(ARDS)72;
• central nervous system dysfunction including seizures and coma73;
• liverandrenalfailure74;
• coagulopathy; • rhabdomyolysis.75
Otherclinicalconditionspresentingwithincreasedcore tem-perature need to be considered, including drug toxicity, drug withdrawalsyndrome,serotoninsyndrome,neurolepticmalignant syndrome,sepsis,centralnervoussysteminfection,endocrine dis-orders(e.g.thyroidstorm,phaeochromocytoma).
Treatment. Themainstayoftreatmentis supportivetherapy andrapidlycoolingthepatient.76–78Startcoolinginthe
prehospi-talsettingifpossible.Aimtorapidlyreducethecoretemperature toapproximately39◦C.Patientswithsevereheatstrokeneedto bemanagedinanICUenvironment.Largevolumesoffluidsand correctionofelectrolyteabnormalitiesmayberequired(see hypo-/hyperkalaemiaandotherelectrolytedisorders).
Cooling techniques. Several cooling methods have been described, but there are few formal trials to determine which isoptimal. Simple coolingtechniquesinclude drinkingcold flu-ids,fanningthecompletelyundressedpatientandsprayingtepid wateronthepatient.Icepacksoverareaswheretherearelarge superficialbloodvessels(axillae,groins,neck)mayalsobeuseful. Surfacecoolingmethodsmaycauseshivering.Incooperative sta-blepatients,immersionincoldwatercanbeeffective79;however,
thismaycauseperipheralvasoconstriction,shuntbloodawayfrom theperipheryandreduceheatdissipation.Immersionisalsonot practicalinthesickestpatients.
Furthertechniquestocoolpatientswithhyperthermiaare sim-ilar to those used for targeted temperature management after cardiacarrest(seepostresuscitationcare).80Coldintravenous
flu-idswilldecreasebodytemperature.Gastric,peritoneal,81pleural
orbladderlavagewithcoldwaterwilllowerthecoretemperature. IntravascularcoolingtechniquesincludetheuseofcoldIVfluids,82
intravascularcoolingcatheters83,84andextracorporealcircuits,85
e.g.continuousveno-venoushaemofiltrationorcardiopulmonary bypass.
Pharmacologicaltreatment. Therearenospecificdrugtherapies inheatstrokethatlowercoretemperature.Thereisnogood evi-dencethatantipyretics(e.g.non-steroidalanti-inflammatorydrugs orparacetamol)areeffectiveinheatstroke.Diazepammaybe use-fultotreatseizuresandfacilitatecooling.86Dantrolenehasnotbeen
showntobebeneficial.87–89
Malignanthyperthermia
Malignanthyperthermiaisalife-threateninggeneticsensitivity ofskeletalmusclestohalogenatedvolatileanaestheticsand depo-larisingneuromuscularblockingdrugs,occurringduringorafter anaesthesia.90Stoptriggeringagents immediately;giveoxygen,
correctacidosisandelectrolyteabnormalities.Startactivecooling andgivedantrolene.91
Other drugs such as 3,4-methylenedioxymethamphetamine (MDMA, ‘ecstasy’) and amphetamines also cause a condition similartomalignanthyperthermiaandtheuseofdantrolenemay bebeneficial.92
Modifications to cardiopulmonary resuscitation. There are no specificstudiesofcardiacarrestinhyperthermia.Ifcardiacarrest occurs,followstandardguidelinesandcontinuecoolingthepatient. Use the same cooling techniques as for targeted temperature managementaftercardiacarrest(seeSection5Post-resuscitation care).80Attemptdefibrillationusingstandardenergylevels.Animal
studiessuggesttheprognosisispoorcomparedwithnormothermic cardiacarrest.93,94Theriskofunfavourableneurologicaloutcome
increasesby2.26(oddsratio)foreachdegreeofbodytemperature >37◦C.95
Hypovolaemia Introduction
Hypovolaemiaisapotentiallytreatablecauseofcardiacarrest thatusuallyresultsfromareducedintravascularvolume(i.e. hae-morrhage),butrelativehypovolaemiamayalsooccurinpatients withseverevasodilation(e.g.anaphylaxis,sepsis).Hypovolaemia frommediator-activatedvasodilationandincreasedcapillary per-meability is a major factor causing cardiac arrest in severe anaphylaxis.96Hypovolaemiafrombloodloss,isa leadingcause
ofdeathintraumaticcardiacarrest.97Externalbloodlossisusually
obvious,e.g.trauma,haematemesis,haemoptysis,butmaybemore challengingtodiagnosewhenoccult,e.g.gastrointestinal bleed-ingorruptureofanaorticaneurysm.Patientsundergoingmajor surgeryareathigh-riskfromhypovolaemiaduetopost-operative haemorrhageandmustbeappropriatelymonitored(see perioper-ativecardiacarrest).
Dependingonthesuspectedcause,initiatevolumetherapywith warmedblood products and/orcrystalloids,in order to rapidly restoreintravascularvolume.Atthesametime,initiate immedi-ateinterventiontocontrolhaemorrhage,e.g.surgery,endoscopy, endovasculartechniques,98ortreattheprimarycause(e.g.
anaphy-lacticshock).Intheinitialstagesofresuscitationuseanycrystalloid solutionthatisimmediatelyavailable.Ifthereisaqualified sono-grapherabletoperformultrasoundwithoutinterruptiontochest compressions,e.g.duringrhythmcheckorventilations,itmaybe consideredasanadditionaldiagnostictoolinhypovolaemiccardiac arrest.
Treatmentrecommendationsforcardiacarrestandperiarrest situationsin anaphylaxisandtraumaare addressedinseparate sectionsbecauseoftheneedforspecifictherapeuticapproaches. Anaphylaxis
Definition. A precise definition of anaphylaxisis not important foritsemergencytreatment.99TheEuropeanAcademyofAllergy
and Clinical Immunology Nomenclature Committee proposed thefollowing broad definition:100 anaphylaxisis a severe,
life-threatening,generalisedorsystemichypersensitivityreaction.This is characterised by rapidly developing life-threatening airway and/orbreathingand/orcirculation problemsusuallyassociated withskinandmucosalchanges.1,96,101,102
Epidemiology. Anaphylaxisiscommonandaffectsabout1in300 oftheEuropeanpopulationatsomestageintheirlives,withan incidencefrom1.5 to7.9 per100,000person-years.103
Anaphy-laxiscanbetriggeredbyanyofaverybroadrangeoftriggerswith food,drugs,stinginginsects,andlatexthemostcommonly iden-tifiedtriggers.103Foodisthecommonesttriggerinchildrenand
drugsthecommonestinadults.104Virtuallyanyfoodordrugcan
beimplicated,butcertainfoods(nuts)anddrugs(muscle relax-ants,antibiotics,nonsteroidalanti-inflammatorydrugsandaspirin) causemostreactions.105Asignificantnumberofcasesof
anaphy-laxisareidiopathic.Between1992and2012intheUK,admission andfatalityratesfordrug-andinsectsting-inducedanaphylaxis werehighestinthegroupaged60years andolder.Incontrast, admissionsduetofood-triggeredanaphylaxisweremostcommon inyoungpeople,withamarkedpeakintheincidenceoffatalfood reactionsduringthesecondandthirddecadesoflife.106
Theoverallprognosisofanaphylaxisisgood,withacase fatal-ityratiooflessthan1%reportedinmostpopulation-basedstudies. TheEuropeanAnaphylaxisRegistryreportedthatonly2%of3333 caseswereassociatedwithcardiacarrest.107Ifintensivecareunit
admissionisrequired,survivaltodischargeisover90%.Overthe period2005–2009,therewere81paediatricand1269adult admis-sionswithanaphylaxisadmittedtoUKcriticalcareunits.Survival todischargewas95%forchildren,and92%foradults.108
Anaphylaxisandriskofdeathisincreasedinthosewith pre-existing asthma,particularlyifthe asthmais poorly controlled, severeorinasthmaticswhodelaytreatment.109,110When
anaphy-laxisisfatal,deathusuallyoccursverysoonaftercontactwiththe trigger.Fromacaseseries,fatalfoodreactionscauserespiratory arresttypicallywithin30–35min;insectstingscausecollapsefrom shockwithin10–15min;anddeathscausedbyintravenous med-icationoccurmostcommonlywithin5min.Deathneveroccurred morethan6haftercontactwiththetrigger.101,111
Recognition of an anaphylaxis. Anaphylaxis is the likely diagno-sisif a patient whois exposed to a trigger(allergen)develops asuddenillness(usuallywithinminutes)withrapidly develop-ing life-threatening airway and/or breathing and/or circulation problemsusuallyassociatedwithskinandmucosalchanges.The reactionisusuallyunexpected.
TheEuropeanAcademyofAllergyand ClinicalImmunology’s (EAACI)TaskforceonAnaphylaxisstatethatanaphylaxisishighly likelywhenanyoneofthefollowingthreecriteriaisfulfilled96,112:
1.Acuteonsetofanillness(minutestoseveralhours)with involve-mentoftheskin,mucosaltissue,orboth(e.g.generalisedhives, pruritusorflushing,swollenlips–tongue–uvula)andatleastone ofthefollowing:
a.Respiratory compromise, e.g. dyspnoea, wheeze– bronchospasm,stridor,reducedpeakexpiratoryflow(PEF), hypoxaemia.
b.Reducedbloodpressureorassociatedsymptomsofend-organ dysfunction,e.g.hypotonia(collapse),syncope,incontinence. 2.Twoormoreofthefollowingthatoccurrapidlyafterexposure toalikelyallergenforthatpatient(minutestoseveralhours): a.Involvement of the skin–mucosal tissue, e.g. generalised
hives,itch-flush,swollenlips–tongue–uvula.
b.Respiratory compromise, e.g. dyspnoea, wheeze– bronchospasm,stridor,reducedPEF,hypoxaemia.
c.Reducedbloodpressureorassociatedsymptoms,e.g. hypoto-nia(collapse),syncope,incontinence.
d.Persistentgastrointestinalsymptoms,e.g.crampyabdominal pain,vomiting.
3.Reducedbloodpressureafterexposuretoknownallergenfor thatpatient(minutestoseveralhours):
a.Infantsandchildren:lowsystolicbloodpressure(<70mmHg from1monthto1year;<70mmHg+(2×age)from1yearto 10years;<90mmHgfrom11to17years)or>30%decreasein systolicbloodpressure.
b.Adults:systolicbloodpressureof<90mmHgor>30%decrease fromthatperson’sbaseline.
Treatment. The evidence supporting specific interventions for thetreatmentofanaphylaxisis limited.113 A systematicABCDE
approach to recognise and treat anaphylaxis is recommended withimmediateadministrationofintramuscular(IM)adrenaline (Fig.4.2).Treatlife-threateningproblemsasyoufindthem.The basicprinciplesoftreatmentarethesameforallagegroups. Moni-torallpatientswhohavesuspectedanaphylaxisassoonaspossible (e.g.byambulancecrew,intheemergencydepartment,etc.). Min-imum monitoring includes pulse oximetry, non-invasive blood pressureanda3-leadECG.
Patientpositioning. Patientswithanaphylaxiscandeteriorate andareatriskofcardiacarrestifmadetosituporstandup.114All
patientsshouldbeplacedinacomfortableposition.Patientswith airwayandbreathingproblemsmayprefertositup,asthiswill makebreathingeasier.Lyingflatwithorwithoutlegelevationis helpfulforpatientswithalowbloodpressure.
Remove the trigger(if possible). Stopany drugsuspected of causinganaphylaxis. Removethestingeraftera bee/waspsting. Earlyremovalismoreimportantthanthemethodofremoval.115Do
notdelaydefinitivetreatmentifremovingthetriggerisnotfeasible. Cardiacarrestfollowinganaphylaxis. StartCPRimmediatelyand followcurrentguidelines.ProlongedCPRmaybenecessary. Rescu-ersshouldensurethathelpisonitswayasearlyALSisessential.
Airwayobstruction. Anaphylaxiscancauseairwayswellingand obstruction.Thiswillmakeairwayandventilationinterventions (e.g.bag-maskventilation,trachealintubation,cricothyroidotomy) difficult.Considerearlytrachealintubationbeforeairwayswelling makesthisdifficult.Callforexperthelpearly.
Adrenaline (first line treatment). Adrenaline is the most impor-tantdrugforthetreatmentofanaphylaxis.116,117Althoughthere
are no randomised controlled trials,118 adrenaline is a logical
treatment and there is consistent anecdotal evidence suppor-tingitsusetoeasebronchospasmandcirculatorycollapse.Asan
Fig.4.2.Anaphylaxistreatmentalgorithm.101
alpha-receptor agonist, it reverses peripheral vasodilation and reducesoedema.Itsbeta-receptoractivitydilatesthebronchial air-ways,increasestheforceofmyocardialcontraction,andsuppresses histamineandleukotrienerelease.Activationofbeta-2adrenergic receptorsonmastcellsurfacesinhibittheiractivation,andearly adrenalineattenuatestheseverityofIgE-mediatedallergic reac-tions.Adrenalineismosteffectivewhengivenearlyaftertheonset ofthereaction,119andadverseeffectsareextremelyrarewith
cor-rectIMdoses.
Giveadrenalinetoallpatientswithlife-threateningfeatures. Ifthesefeaturesareabsentbutthereareotherfeaturesofa sys-temicallergicreaction,thepatientneedscarefulobservationand symptomatictreatmentusingtheABCDEapproach.
Intramuscularadrenaline. Theintramuscular(IM)routeisthe bestfor most individuals who have togive adrenalineto treat anaphylaxis.Monitorthepatientassoonaspossible(pulse,blood pressure,ECG,pulseoximetry).Thiswillhelpmonitortheresponse toadrenaline.TheIMroutehasseveralbenefits:
• Thereisagreatermarginofsafety. • Itdoesnotrequireintravenousaccess. • TheIMrouteiseasiertolearn.
• Patientswithknownallergiescanself-administerIMadrenaline. ThebestsiteforIMinjectionistheanterolateralaspectofthe middlethirdofthethigh.Theneedleforinjectionneedstobelong enoughtoensurethattheadrenalineisinjectedintomuscle.120
Thesubcutaneousorinhaledroutesforadrenalinearenot recom-mendedfor thetreatmentof anaphylaxisbecausetheyare less effectivethantheIMroute.121–123
Adrenaline intramuscular dose. Theevidence for the recom-mended doses is limited. The EAACI suggests IM adrenaline (1mgmL−1)shouldbegivenadoseof10mcgkg−1ofbodyweight toamaximumtotaldoseof0.5mg.96
Thefollowingdosesarebasedonwhatisconsideredtobesafe andpracticaltodrawupandinjectinanemergency(equivalent volumeof1:1000adrenalineisshowninbrackets):
>12yearsandadults 500microgramIM(0.5mL)
>6–12years 300microgramIM(0.3mL)
>6months–6years 150microgramIM(0.15mL)
<6months 150microgramIM(0.15mL)
Repeat the IM adrenaline dose if there is no improve-mentinthepatient’s conditionwithin5min.Furtherdosescan be given at about 5-min intervals according to the patient’s response.
Intravenousadrenaline(forspecialistuseonly). Thereisamuch greater risk of causing harmful side effects by inappropriate dosageormisdiagnosisofanaphylaxiswhenusingintravenous(IV) adrenaline.124IVadrenalineshouldonlybeusedbythose
expe-riencedintheuseandtitration ofvasopressorsintheirnormal clinicalpractice(e.g.anaesthetists,emergencyphysicians, inten-sivecaredoctors).Inpatientswithaspontaneouscirculation,IV adrenalinecancauselife-threatening hypertension,tachycardia, arrhythmias,andmyocardialischaemia.IfIVaccessisnotavailable ornotachievedrapidly,usetheIMrouteforadrenaline.Patients whoaregivenIVadrenalinemustbemonitored–continuousECG andpulseoximetryandfrequentnon-invasivebloodpressure mea-surementsasaminimum.PatientswhorequirerepeatedIMdoses ofadrenalinemaybenefitfromIVadrenaline.Itisessentialthat thesepatientsreceiveexperthelpearly.
Adrenalineintravenousdose(forspecialistuseonly).
• Adults:TitrateIVadrenalineusing50microgramboluses accord-ingtoresponse.Ifrepeatedadrenalinedosesareneeded,startan IVadrenalineinfusion.125,126
• Children:IMadrenalineisthepreferredrouteforchildren hav-inganaphylaxis.TheIVrouteisrecommendedonlyinspecialist
paediatricsettingsbythosefamiliarwithitsuse(e.g.paediatric anaesthetists,paediatricemergencyphysicians,paediatric inten-sivists)andifthepatientismonitoredandIVaccessisalready available.Thereisnoevidenceonwhichtobaseadose recom-mendation–thedoseistitratedaccordingtoresponse.Achild mayrespondtoadoseassmallas1mcgkg−1.Thisrequiresvery carefuldilutionandcheckingtopreventdoseerrors.
Adrenalineintravenous/intraosseousdose(incardiacarrestonly). Cardiacarrestwithsuspectedanaphylaxisshouldbetreatedwith standard dosesofIV orintraosseous (IO)adrenalineforcardiac arrest.Ifthisisnotfeasible,considerIMadrenalineifcardiacarrest isimminentorhasjustoccurred.
Oxygen(giveassoonasavailable). Initially,givethehighest concentration of oxygen possible using a mask with an oxy-genreservoir.127 Ensure high-flowoxygen(usuallygreaterthan
10Lmin−1topreventcollapseofthereservoirduringinspiration. Ifthepatient’stracheaisintubated,ventilatethelungswithhigh concentrationoxygenusingaself-inflatingbag.
Fluids(giveassoonasavailable). Largevolumesoffluidmay leakfromthepatient’scirculationduringanaphylaxis.Therewill alsobevasodilation.IfIVaccesshasbeengained,infuseIVfluids immediately.GivearapidIVfluidchallenge(20mLkg−1)inachild or500–1000mLinanadultandmonitortheresponse;givefurther dosesasnecessary.Thereisnoevidencetosupporttheuseof col-loidsovercrystalloidsinthissetting.Considercolloidinfusionas acauseinapatientreceivingacolloidatthetimeofonsetofan anaphylaxisandstoptheinfusion.Alargevolumeoffluidmaybe needed.
IfIVaccessisdelayedorimpossible,theIOroutecanbeusedfor fluidsordrugs.DonotdelaytheadministrationofIMadrenaline whileattemptingIOaccess.
Antihistamines(giveafterinitialresuscitation). Antihistamines are a second line treatment for anaphylaxis. The evidence to support their use is limited, but there are logical reasons for theiruse.128H
1-antihistamineshelpcounterhistamine-mediated vasodilation, bronchoconstriction, and particularly cutaneous symptoms.Thereislittleevidencetosupporttheroutineuseofan H2-antihistamine(e.g.ranitidine,cimetidine)fortheinitial treat-mentofanaphylaxis.
Glucocorticosteroids(giveafterinitialresuscitation). Corticoste-roidsmayhelppreventorshortenprotractedreactions,although theevidenceislimited.129Inasthma,earlycorticosteroidtreatment
isbeneficialinadultsandchildren.Thereislittleevidenceonwhich tobasetheoptimumdoseofhydrocortisoneinanaphylaxis.
Otherdrugs.
Bronchodilators. Thepresentingsymptomsandsignsofsevere anaphylaxisandlife-threateningasthmacanbethesame.Consider furtherbronchodilator therapy withsalbutamol (inhaledor IV), ipratropium(inhaled),aminophylline(IV)ormagnesium(IV)(see asthma).IVmagnesiumisavasodilatorandcanmakehypotension worse.
Cardiac drugs. Adrenalineremains thefirstlinevasopressor for thetreatmentof anaphylaxis. Thereare animal studiesand casereportsdescribingtheuseofothervasopressorsandinotropes (noradrenaline,vasopressin,terlipressinmetaraminol, methoxam-ine,andglucagon)wheninitialresuscitationwithadrenalineand fluidshasnotbeensuccessful.130–142Usethesedrugsonlyin
spe-cialistsettings(e.g.ICU)where thereisexperienceintheiruse. Glucagoncanbeusefultotreat anaphylaxisina patienttaking a beta-blocker.143 Some case reports of cardiac arrest suggest
cardiopulmonarybypass144,145ormechanicalchestcompression