HAL Id: hal-03218463
https://hal.archives-ouvertes.fr/hal-03218463
Submitted on 5 May 2021
HAL is a multi-disciplinary open access
archive for the deposit and dissemination of
sci-entific research documents, whether they are
pub-lished or not. The documents may come from
teaching and research institutions in France or
abroad, or from public or private research centers.
L’archive ouverte pluridisciplinaire HAL, est
destinée au dépôt et à la diffusion de documents
scientifiques de niveau recherche, publiés ou non,
émanant des établissements d’enseignement et de
recherche français ou étrangers, des laboratoires
publics ou privés.
Effect of silver and strontium incorporation route on
hydroxyapatite coatings elaborated by rf-SPS
Marine Chambard, Djamel Remache, Yannick Balcaen, Olivier Dalverny, Joël
Alexis, Robin Siadous, Reine Bareille, Sylvain Catros, Pascal Fort, David
Grossin, et al.
To cite this version:
Marine Chambard, Djamel Remache, Yannick Balcaen, Olivier Dalverny, Joël Alexis, et al.. Effect of
silver and strontium incorporation route on hydroxyapatite coatings elaborated by rf-SPS. Materialia,
Elsevier, 2020, 12, pp.100809. �10.1016/j.mtla.2020.100809�. �hal-03218463�
OATAO is an open access repository that collects the work of Toulouse
researchers and makes it freely available over the web where possible
Any correspondence concerning this service should be sent
to the repository administrator:
tech-oatao@listes-diff.inp-toulouse.fr
This is an author’s version published in: http://oatao.univ-toulouse.fr/27198
To cite this version:
Chambard, Marine and Remache, Djamel
and Balcaen,
Yannick
and Dalverny, Olivier
and Alexis, Joël
and Siadous,
Robin and Bareille, Reine and Catros, Sylvain and Fort, Pascal and
Grossin, David and Gitzhofer, François and Bertrand, Ghislaine
Effect of silver and strontium incorporation route on hydroxyapatite
coatings elaborated by rf-SPS. (2020) Materialia, 12. 1-12. ISSN
25891529
Official URL:
https://doi.org/10.1016/j.mtla.2020.100809
Effect
of
silver
and
strontium
incorporation
route
on
hydroxyapatite
coatings
elaborated
by
rf-SPS
Marine
Chambard
a,b,∗,
Djamel
Remache
c,
Yannick
Balcaen
c,
Olivier
Dalverny
c,
Joël
Alexis
c,
Robin
Siadous
d,
Reine
Bareille
d,
Sylvain
Catros
d,
Pascal
Fort
e,
David
Grossin
a,
François
Gitzhofer
b,
Ghislaine
Bertrand
aa CIRIMAT, Université de Toulouse, CNRS, INP- ENSIACET 4 allée Emile Monso - BP44362, 31030 Toulouse cedex 4, France b CREPE, Department of Chemical and Biotechnological Engineering, Université de Sherbrooke, Sherbrooke, QC, Canada c LGP, Université de Toulouse, INP/ENIT, 47, avenue d’Azereix, Tarbes, F-65016, France
d Inserm, U1026, Tissue Bioengineering, Bordeaux University, Bordeaux, France e PROJECTION PLASMA SYSTEME (2PS), ZI du Colombier, 12220 Montbazens, France
Keywords:
rf - Suspension plasma spraying doped hydroxyapatite coating mechanical properties biological properties bactericidal properties
a
b
s
t
r
a
c
t
Hydroxyapatitecoatingshavebeencurrentlyusedonhipprosthesesfortheirabilitytopromotefaster osseointe-grationandbonegrowth.Nevertheless,post-operativeinfectionsremainarecurringproblem.Toovercomethis issue,dopingwithantibacterialelementshasbecomeanewtrend.Inthiswork,hydroxyapatitecoatings elab-oratedbyradio-frequencysuspensionplasmaspraying(rf-SPS)weredopedwithsilverandstrontium.Several dopingstrategieswereexploredthankstotheversatilityofferedbySPScomparedwithconventionalspraying. Firstway:calciumphosphatedopedpowdersweresynthesizedbycoprecipitationandthendispersedintowater beforeplasmaspraying;secondway:undopedpowderwasdispersedintoaqueousmediuminwhichnitratesor nanoparticlesofthedopant(s)wererespectivelydissolved/dispersed.XRDrevealedahighlevelofcrystallinity ratio(ISO13779)andhydroxyapatiteproportionformostofthecoatings,withthepresenceofAg/Ag2O
nanopar-ticleswhateverthedopingroute.SEM-EDSandSTEMhavedemonstratedamorehomogeneousdistributionofthe strontiumwithinthecoatingmadefromthedopedpowder.Adherenceofthecoatingswasestimatedvia a3-point bendingtest,whilebacteriologicaltestswithS. aureus andproliferationofmesenchymalstemcells(hMSC)were performed.Theresultsindicatedapreferentialincorporationofstrontiumintothesecondaryphases,showed efficientbactericidalproperties,excellentmechanicalpropertiesincomparisonwithanAPSreferencecoating, andnoevidenceofcytotoxiceffect.Thisopensthewayofanewtypeofcoatingswithafinerstructureanda higherhomogeneitythroughabettercontrolofphysicochemicalpropertiesusingasuspensionastheprecursor.
1. Introduction
Theincreaseintheworld’spopulationanditsaginginducesan ever-increasingdemandfororthopedicprostheses[1].However,inabout1% ofthenewlyimplantedprostheses[2],aninfectioncausedbybacterial agentssuchasstaphylococciorenterobacteria istriggered, requiring newsurgery.Suchaninterventionisbothexpensiveandtraumatic. Cal-ciumphosphatecoatingshavebeensuccessfullyusedfor30yearsin or-thopaedicsurgerysincetheysuppressmicro-movements,supportbone in-growthandprovideosseostabilitytouncementedhipimplants[3,4]. Hydroxyapatiteisparticularlyusedascalciumphosphateasitis struc-turallyandchemicallysimilartobonemineral,andlessresorbablethan othercalciumphosphatephases[5]suchastricalciumphosphate(TCP), tetracalciumphosphate(TTCP),amorphouscalciumphosphate(ACP)or
∗Correspondingauthorat:CIRIMAT,Université deToulouse,CNRS,INP-ENSIACET4alléeEmileMonso-BP44362,31030Toulousecedex4,France.
calciumoxide(CaO),whosepresenceislimitedeventhoughtypicalin plasma-sprayedcoating.
Thedevelopmentofhipprostheseswhichcanpreventbacterial infec-tionsisthereforeacurrentchallenge.Themainstrategywouldconsistin addingadopingelementwithantibacterialandanti-inflammatory prop-ertiessuchasmetalelements(Cu,Ag,Zn…)tohydroxyapatitethatis coatedontheimplant.Amongtheavailabledopants,silverisofgreat in-terestbecauseofitsreactivitywiththiolicmoieties(especiallyagainst gram-bacteria)[6]makingitoneofthemosteffectiveantibacterial agentknownatlowconcentration.Eventhoughfirstclinicalresults im-plyingsilverdopedhydroxyapatitecoatingarepromising[7],ahigh concentrationofsilvercanbetoxicandreportedlycausesargyria, hep-atopathy,andnephropathy[8,9].Albersetal.[10]conductedastudy inwhichtheydemonstratedthatthecytotoxicitythresholdofsilverfor osteoblastsandosteoclastswas2to4timeslowerthanitsantibacterial
efficacyagainstS.epidermidis. Thissamestudyhasalsoputforwarda
significantincreaseinthecytotoxicityofsilverparticleswhentheirsize isreducedfromthemicro(3𝜇m)tothenanometer(50nm).In
paral-lel,otherstudieshavedemonstratedthatsilverbecomescytotoxicwhen theconcentrationinthebloodexceeds2𝜇gmL−1, buthasnoadverse
effectonhMSCaslongastheionicconcentrationintheapatitedoesnot exceed0.7wt%[11,12].Inconclusion,manyparametersmustbe con-sidered,anditisnecessarytocontrolboththesilverconcentrationin thecoatinganditsreleaseoncetheprosthesisisimplanted.Thisway, itshouldbepossibletocounteracttheharmfulnessofsilver.Fielding etal.showedthatstrontium,whenusedinreasonableproportion(3– 7 at%[13]),canbalancethecytotoxicityofsilverviaosteoblast stimula-tionandosteoclastinhibition[3,14],andotherwouldevenlenditsome antibacterialproperties[15]. Inaddition,Ratnayakeetal.[16]reports thatsilverandstrontium-substitutedhydroxyapatitehasitsmechanical propertiesreinforced,eventhoughitssolubilitywithinbodyfluids in-creases.Othersworksconfirmedtheefficiencyandinterestofsilverand strontiumco-doping[17,18].Thisco-dopingseemsthereforepromising forpotentialclinicalapplications,eventhoughaspecialattentionshall begiventoitsimpactontheresorptionofHAin biologicalmedium, whichhasbeenshowntobeweakenedaboveacertainsubstitution de-gree[16]anddeterminesthesustainabilityof thedevicewithin the body.
Currently,conventionalpowderplasmaspraying(APS)isthe tech-niqueused bymost of themanufacturerstoproduce coateddevices whichmeettheISOstandardsandtheFDArequirements[19,20].But thistechniquehasitslimits.Firstly,becauseoftheuseoflarge pow-ders,itdoesnotallowtoreachamicrostructureasfineasthatofthe mineralbone,eventhoughresearchhashighlightedthestimulationof osteoblastsseededonnanostructuredcalciumphosphates[21]. And sec-ondly,thistechniqueoffersonlytwowaysofintegratingthesedopants intothecoatings:eitherviathesynthesisofadopedpowderorby mix-ingpowdersofhydroxyapatiteandofdopantsinoxidizedormetallic form[3,22,23].Thelattersolutionleadstoaratherpoorstructuraland chemicalhomogeneityofthecoating,andtheformerhaslimitationin thechemicalcompositionofthedopedpowder.Indeed,thesynthesisof thesilver-dopedpowderwhilepreservingthestoichiometryofthe hy-droxyapatiteischallengingduetothedifferenceinchargeandsizeof thesilverionAg+. Typically,silverisonlyincorporatedatamaximum
of10wt.%[6]. Usingasuspensioninsteadofapowderoffersan in-terestingalternativesinceitallowsononehandsprayingsmaller(even nanometric)particlesleadingtofinerstructures[24], improvingthe ho-mogeneityofboththecoatingandthedoping,andontheotherhand itenrichesthedopingwaysbyinvolvingtheuseofaliquiddispersive medium.Inaddition,bydopingthedispersivemedium,abettercontrol oftheincorporatedamountofdopantswhileimprovingtheir distribu-tionwithinthecoatingisexpected.Thequestionis:towhatextentdoes thedopingprotocolusedinrf-SPSaffectthebiologicalandmechanical propertiesofthecoating?
Thispaperaimstoevaluatethemechanicalandbiologicalproperties ofsilverandstrontium-hydroxyapatitecoatingssynthesizedbyrf-SPS, implementingdifferentdopingroutes.Thedopingofthesuspensionwas performedeitherusingapowderdopedbycoprecipitationordissolving silverandstrontiumnitratesoraddingsilvernanoparticlesinthe dis-persivemedium.A3-pointbendingtestwasimplementedtodetermine themechanicaladherenceofthecoatings,whiletheadhesionand pro-liferationofhMSCwasevaluatedtoassesstheirbioactivity,andthe de-velopmentofstaphylococcusaureusbacteriawastestedtoestablishtheir bactericidalactivity.
2. Experimental
2.1. PreparationofunsubstitutedandAg/Sr-substitutedHA
Apurehydroxyapatite(Hap)powderandanotheronesubstituted with Ag and Sr (Ag/Sr-Hap) were synthesized to produce the
sus-pensionsdestinedtorf-SPSplasmaspraying.Calciumnitrate tetrahy-drate (Ca(NO3)2, 4 H2O – Sigma Aldrich), diammonium phosphate ((NH4)2HPO4-Fischer),silvernitrate(Ag(NO3),Fischer),strontium ni-trate(Sr(NO3)2,Sigma-Aldrich)andNH4OHsolutionat20wt.% (Fis-cher)wereusedaschemicalprecursorsforCa2+,PO
42−,Ag+,Sr2+and
OH−.Theunsubstitutedcalciumphosphatewasobtainedby
coprecip-itationaccordingtothefollowingbalancedchemicalequation(Eq1):
10 Ca (N O 3 ) 2 + 6 (N H 4 ) 2 HP O 4 + 8 N H 4 OH → C a 10(P O 4 ) 6 O H 2 + 6 H 2 O + 20 N H 4 N O 3 (1) Thepowdersynthesisprotocolswereadaptedfromthosedescribed byKannan[25].Asolutionofdiammoniumphosphatewasaddedtoa solutionofnitrate(s)suchastheCa/Pand(Ca+Sr+Ag)/Pratiosofthe powdersHapandAg/Sr-Haprespectivelywerebothsetto1.667to pro-motetheformationofstoichiometrichydroxyapatite.ThepHwas main-tainedat9allalongthesynthesisbyadding20wt.%NH4OHtoobtain theappropriateacidityofthephosphateion.Theinitialmolar concen-trationofthedopantswassettomol.%Ag=Ag/(Ca+Sr+Ag)=10.0and mol.%Sr=Sr/(Ca+Sr+Ag)=5.0.Afterthematurationtime(24h),the powderswerefilteredandwashedwith10timesthereactionmedium volumeofwaterbeforefreeze-drying(pressureof0.10mbar,−80°Cfor 3days).Themol%ofthedopantwithinthesynthesizedpowderwas measuredat1.41mol.%forAgand5.27mol.%forSr.Twopowders weresynthesizedfromthisprotocol,namelyHapandAg/Sr-Hap.
2.2. Suspensionpreparation
Priortospraying,thepreviouslydescribedpowders(Hapor Ag/Sr-Hap)weredispersedintowateratasolidloadof13wt.%,andan ul-trasonicprobewasplungedintothesuspensionfor5minat30Wto breakuptheaggregates,leadinguptotwosuspensions,namedafter thepowders:HapandAg/Sr-Hap.
Then, two more Ag and Sr doped suspensions were produced, where the liquid medium was doped instead of the powder: one with silver nanoparticles (AgNP), the other one with silver nitrates
(Ag+),andbothwithstrontiumnitrates(Sr2+).UndopedHapwasthen
added tothis dopedmedium,giving risetotwo suspensionsnamed Hap+[Ag+]/[Sr2+]andHap+AgNP+[Sr2+].
In summary, four suspensions were prepared: with the undoped (Hap),with thedopedpowder(Ag/Sr-Hap),withtheundoped pow-der and nitrates (Hap+[Ag+]/[Sr2+]) and with the undoped
pow-der and a mixture of strontium nitrate and silver nanoparticles (Hap+AgNP+[Sr2+]),whichleadtothesynthesisofcoatingsidentified
withthesamereferenceastheirprecursor.
2.3. Coatings 2.3.1. Substrates
TitaniumalloyTi-6Al-4V(grade5)wasselectedasthesubstrate ma-terialforitssufficientmechanicalstrength,anti-corrosionandbioinert properties[26,27].Platesof50×55×1.6mm3adaptedtotheprocess wereused.The3-pointbendingsamples(50×10×1.6mm3)werelaser pre-cutexceptattwoslotsinthetitaniumplates,aswellascylindersof 10mmindiameterusedforbiologicalandbacteriologicaltests.
Prior toplasma spraying, the plates were cleaned with solvents (ethanolandacetoneinultrasonicbath)andgrit-blastedwithalumina (F120)atanincidenceangleof90°,a120mmblastingdistanceand apressureof4.5bar.Thearithmeticroughnessofthesurface(Sa)was determinedwithaprofilometertobeapproximately1.5±0.2µm.
2.3.2. Plasmasprayingparameters
Rf-SPS coatings were produced at the Université de Sherbrooke (Québec)withaplasmageneratedbya50-kWRFplasmatorch operat-ingat3MHz(PL-50modelfromTeknaPlasmaSystemInc.)equipped
withasupersonicnozzle.Thesuspensionwasinjectedbyaperistaltic pumpatafeedingrateof12.5mLmin−1andthroughasupersonic
noz-zlewitha45mmoutputdiameter[28]. Theliquidatomisationprobe washome-designed,andspraysofwaterdropletswithamean diame-terD50of11µmwereproduced.Thecompositionoftheplasmawasa mixtureofargonanddioxygenbasedonpreviousworkperformedby Loszachetal.[29], andthegeneratedpowerwas35kW,withapressure withinthechamberadjustedto90torr.Theworkingdistancefromthe torchtothesampleholderwassetto185mm,andthedistanceswept bythesampleholderundertheplasmajetwassetto80mm.8 preheat-ingpassesfollowedby30sprayingpasseswerenecessarytoachievea thicknessofabout100µm.
2.4. Characterizationofpowdersandcoatings
AllthefollowingcharacterizationswereperformedinlinewithISO standard(i)ISO13,779–2:2008and(ii)ISO13,779–3:2008[19,20].
TheparticlesizedistributionwasmeasuredwithaMalvern Master-sizerHydro2000Sgranulometer(UK)inaqueousmedia,toreproduce thestateofthesuspensionbeforeplasmaspraying.Themeasuringcell waskeptunderagitationat1750rpmforalltrialstoprevent sedimen-tation.
Theelementalcompositionsofpowdersandcoatingsweremeasured byaninductivelycoupledplasmaopticalemissionspectrometerJobin YvonUltima2(Horiba,Japan).Theroughnessofthesubstratesandthe coatingswasevaluatedusing aSensofarS-Neoxconfocal microscope (Barcelona,Spain)atx10magnification.Theaveragevaluewas deter-minedfrom5scanningzonesof1750× 1300µmat0,90°,180° and 270°.Thecrystallinephaseanalysisof thepowdersandthecoatings wereinvestigatedusing D8diffractometer(Bruker AXSGmbH, Karl-sruhe,Germany)withCuK𝛼 radiation(𝜆 =1.5406Å)producedat40kV
and50 mA.Datawererecordedin the20 ° - 60 ° 2𝜃 rangewith a
stepsizeof0.02° and38sperstep.Thepatternswereidentified us-ingMatch!softwareandsemi-quantitativeanalyseswererealizedusing theReferenceIntensityRatiomethod[30], usingthefollowingJCPDS data(HA,hydroxyapatite,JCPDSno.00–009–0432;𝛼-TCP,tricalcium
phosphateCa3(PO4)2, JCPDSno.04–018–9895;𝛽-TCP,tricalcium
phos-phateCa3(PO4)2, JCPDSno.00–070–2065;TTCP,tetracalcium
phos-phate,Ca4P2O9, JCPDSno.00–025–1137;CaO,calciumoxide,JCPDS no.04–011–9020).ThecrystallinityratioR1(Eq2)wascalculated ac-cordingtotheformulaproposedinthestandardISO13,779–3,involving theuseofafullycrystallisedstandard(hydroxyapatitepowdercalcined at1000°Cfor15h):
R1=
Integratedintensityof10linesofthesample
Integratedintensityof10linesofthestandard× 100 (2) FourierTransformInfrared FTIR analysiswas carried out with a PerkinElmer 1700spectrometer(USA)onscraped coatingsdispersed inKBrpelletsinaratioof0.3percentmass,ataresolutionof4cm−1
inthe400-4000cm−1range.Chemicalstateofdopantswasevaluated
byX-rayphotoelectronspectroscopy(XPS)withaVGESCALAB220i-XL (ThermoFischerScientific,US)spectrometer,witha150µm-lateral reso-lution.Theobservationofsurfaceandmicrostructurewasconductedby SEMwithaFEIQuanta450(Bruker,Germany),usingtheback-scattering modeat12.5kVand10mmworkingdistance,andbySTEMwitha JEM-ARM200FColdFEG(Jeol,Japan).Todeterminetheporosityfromthe cross-sectionimages,thesampleswerecut,thenmountedandvacuum impregnatedwithcoldepoxyresin.Theywerethengroundandpolished inaccordancewiththeproceduredescribedbyGeelsetal.[31].Image analysiswereperformedwithImageJon10differentviews, implement-ingmanualthresholdinganddespeckling.Contactanglemeasurements werecarriedoutwithaDigidropMCATgoniometer(GBX,Ireland).An equalvolumeofmilliQwater(2 µL)wasplacedon everysampleby meansofamicropipetteformingadroponthesurface,andthecontact anglewasrecorded10safterdropdeposition.
2.5. Calcium,silverandstrontiumrelease
The evolution of the dopedcoatings in physiological media and specificallyinaggressivemediummimickingthelocalinflammatory sit-uationappearingafterimplantationwasassessedbyimplementing dis-solutiontestsinastrongacidicmedium.Toperformthedissolutiontest, acylinderofeachspecimenexceptfortheHap+AgNP+[Sr2+]condition
wasimmersedin50mLofHNO3acidifiedwaterat37°C,pH3.5and subjectedtoconstantshakinginaDissolutestSotaxCE6unit (Switzer-land)ata9mLmin−1flowrateinclosedcirculation.Anextracoating,
synthesizedinsimilarplasmaconditions,fromasuspensioncontaining astrontium-dopedhydroxyapatitepowder(3.39±0.04wt.%)and sil-verunderionicandmetallicform,wasannealedat600°Cfor2hand testedwiththeotherstohighlighttheeffectofcrystallinityonthe dis-solutionbehaviorofthedopedcoatings.Analiquotof10mLofmedium wascollectedateachtimepointstartingat30min,with10mLoffresh solutionaddedateachtimepoint,for5days.ThepHwasmeasured foreachaliquot.ThealiquotcontainingthereleasedAg+andSr2+was
analysedviaatomicabsorptionspectrophotometer(ContrAA300 Ana-lytikJena,Germany)forthestrontiumandcalcium,andinductively coupledplasmamassspectrometer(ICP-MS7500ce,Agilent Technolo-gies,USA)forthesilver.
2.6. Mechanicalproperties
Theadherenceofacoatingisoneofthecriticalcharacteristicsthatis requiredwhendevelopingnewsolutions.Thetestselectedinthepresent studyisathree-pointbendingtest,elaboratedbyRocheetal.[32]and isdescribedintheISO14679:2001standard[33].Apolyepoxide stiff-enerof 25 ×4×5mm3 is placedandadheredin thecenterofthe surfaceofthecoatedtestpiece,whosesurfaceareais50×10mm2.The wholepieceisthenplacedinanInstrontestingmachineandisloadedin bendingatacrossheadspeedof0.50mm.min−1,untilfailure.Adhesion
energy(mJ)isthencalculatedfromthefailureforceandthedeflection. Then,theresidualcoatingpercentageisevaluatedwithEDS cartogra-phy.Ifthepercentageofresidualcoatingisbeyond65%,therupture isdesignedascohesive,below35%meansadhesive, andin between standsforamixedrupturetype.Inordertoensurereproducibilityof theresults,fourreplicateswereused.
2.7. Resazurinassay,cellproliferation
Humanmesenchymal stem cells wereisolated from humanbone marrowaspiratesobtainedfromtheUniversityHospitalofBordeaux af-terapprovalbythelocalethicalcommissionandwrittenconsentofthe patient.TheAlamarBlue○R cellviabilityreagent(Thermofischer,USA) wasusedtoevaluatecellproliferation.Thesampleswerefirstplaced intoanalphaMinimumEssentialMedium(𝛼-MEM)(Sigma,USA)fora
soakingpurposein48-wellplates.1mLof𝛼-MEM+10%Fetalbovine serum(FBS)holdingadensityof50×103cellsml−1werethenseeded
ontothesamples.Ateachtimepoint,themediumwaswithdrawnfrom thewelland1mLof𝛼-MEM+10%serum+10%alamarBluesolution at3.5mgmL−1wereadded.Afterincubation,200µloftheresulting
su-pernatantwastransferredtoa96-wellplateandreadbyaplatereader at590nm.Cellproliferationwasevaluatedat4,7and14 days,and theviabilityofthecellsCV(Eq3)wasestimatedthroughthefollowing formula:
CV=%proliferationoftestedgroup
%proliferationofreference × 100 (3) Foranygivenexperiment,eachdatapointrepresentsthemean± standarderroroffiveindividualcultures.Datawereprocessedby anal-ysisof variancewithtwo-wayANOVA testandBonferroni’sposttest tocompareallmaterialswitheachotherateachtime.Pvalues<0.05
Table1
Physicochemicalcharacteristicsofprecursors.
D 10 D 50 D 90 wt.% Ag wt.% Sr Ca/P (Ca + Sr + Ag)/P Hap 2.8 ± 0.1 9.3 ± 0.3 24 ± 3 – – 1.68 ± 0.03 –
Ag/Sr-Hap 2.2 ± 0.1 4.6 ± 0.1 9.5 ± 0.2 1.40 ± 0.02 4.30 ± 0.07 1.49 ± 0.03 1.59 ± 0.03 Hap + Ag NP + [Sr 2+ ] 2.8 ± 0.1 9.3 ± 0.3 24 ± 3 0.40 ± 0.01 4.37 ± 0.05 1.68 ± 0.03 1.75 ± 0.03 Hap + [A g + ]/[Sr 2+ ] 2.8 ± 0.1 9.3 ± 0.3 24 ± 3 1.54 ± 0.01 4.32 ± 0.02 1.68 ± 0.03 1.77 ± 0.03
2.8. Antimicrobialactivity
TheantimicrobialactivityofdopedHA-coatedsampleswas deter-minedbychallengingwithS.Aureus(ATCC33,591,MRSA).The bac-terialstocksolutionwaspreparedextemporaneouslyinsteriledistilled waterbeforedilutioninaculturemedium.Thepreparedsampleswere sterilizedbyautoclavingat121°Cfor15minandplacedinto24-well plates,coveredwith2mLofbacterialsuspension103UFCmL−1and
incubatedbyrotation for24hat 37°C.Each samplewastakenout at6,24and48handrinsedwith4mLsterilewaterandplacedinto 3mLofthesamesolutionandshakenbyvortexfollowedby1minof ultrasounds.Afterdilution,1mLofthesupernatantwasseededin Tryp-caseSoyAgar(TSA)andthenumberofvisiblecellswasdeterminedby quantifyingCFUs.APScoatingwasusedasacontrolgroup;eachtest wasperformedintriplicate.Thedegreeofthemicrobialcellreduction
BR(Eq4)wasestimatedbythefollowingformula:
BR= ∑
𝑛=3Log(Nbbacteriainstandardgroup)−
∑
𝑛=3Log(Nbbacteriaintestgroup)
3
(4)
3. Results
Inordertoassessthequalityofthecoatingsinrespectwiththe com-mercialstandard,the mechanicalandbiologicaltests werealso per-formedwithanatmosphericplasmaspray(APS)hydroxyapatite stan-dardcoating,servingasReference.
3.1. Physico-chemicalanalysis 3.1.1. Suspension
The suspensions showed very similar characteristics, reported in
Table1.ThemeanD50iscomprisedbetween4.6±0.1µmforthedoped powderand9.3±0.9µmfortheundopedHap.Quitesimilar stron-tiumconcentrationsofabout4.35wt.%weredeterminedwhateverthe dopingstrategy.1.40and1.54wt.%wererespectively measuredfor silverinthecaseofthesuspensionsformulatedeitherfromthedoped Ag/Sr-HappowderorfromtheundopedHappowderaddedwith ni-trates (Hap+[Ag+]/[Sr2+]).Alowercontentof 0.40wt.%wasfound
forthesuspensionproducedfromtheundopedpowderHapmixedwith AgNP nanoparticles(Hap+AgNP+Sr2+).Thecation/Pratiosvary from
1.68±0.03fortheundopedpowderto1.59±0.02fortheAg/Sr-doped powderandto1.76± 0.03forthedopedsuspensions.XRDpatterns ofundopedanddopedsynthesizedpowders(Fig.1)showthetypical crystallographicapatiticstructureofCa10(PO4)6(OH)2(JCPDS00–009– 432),wherenosecondarycrystallinephaseisdetected.Despitean ob-viousdiminutionofthecrystallitesize,thecrystallinityratioisslightly betterforthedopedpowderwithavalueof97±3%forAg/Sr-Hap, comparedwiththevalueof92±3%forHappowder.
3.1.2. Coatings
ThequantitativeXRDanalysis(Table2)establishesthatHAisthe majorphaseforallcoatings(Fig.2).86wt.%ofHAtogetherwitha crys-tallinityratioof70%wereassessedforthecoatingselaboratedfromHap undopedpowder,withorwithoutdopingagentsinthesuspension.The secondaryphasesinthedopedcoatingswereTTCPandCaOinquantities twiceaslargeasinthecoatingproducedfromtheundopedpowderthat
alsocontains𝛼-TCP.Incontrast,forthecoatingmadewiththedoped
powderAg/Sr-Hap,anamountof62wt.%ofHAphasewith33wt.%of
𝛼-TCPweremeasured,aswellasalowcrystallinityratioof57%in
re-gardtothevaluerecommendedintheISO13779–2standardthatmust beover45%.Asmallpeakwasnoticeableat2𝜃=36° inAg/Sr-Hapand Hap+[Ag+]/[Sr2+]patterns, whichcouldbe attributedtoAg (JCPDS
No.4–0783)orAg2O(JCPDSno.19−1155),indicatingthatthesilver ionswerepossiblyturnedintometallicoroxidizedsilverduringplasma spraying.FTIRanalysisvalidatedtheapatitestructureofthecoatings sinceallcharacteristicbands(𝜈1=938cm−1,𝜈
2=420cm−1,𝜈3=1017 cm−1,and𝜈
4=567cm−1)arepresent,butalsoevidencedadiminution
ofthehydroxylationdegree(𝜈LOH−=631and𝜈SOH−=3570cm−1)for alldopedcoatings(Fig.3)[34].Spectradisplayedpoorlydefinedbands thatconfirmedthepoorcrystallinityoftheAg/Sr-Hapcoating.Aweak relativeintensityofthebandsassignedtoOH−groupevidencesalow
hydroxylationdegreeforbothAg/Sr-HapandHap+[Ag+]/[Sr2+]
coat-ingsincomparisonwiththeundopedHapandHap+AgNP+Sr2+
coat-ings.Thisindicatesthatsilvernanoparticlespromotethemaintenance ofapatitehydroxylationduringplasmaspraying,probablybecausethey arelikelytoconsumeheatinalargerextentthroughtheirmeltingand vaporizationsteps.
Thecrosssectionsofthecoatings(Fig.4)observedbySEMshowsa goodqualityofinterfaceandauniformmicrostructure,withsome het-erogeneouszonesandcracks, characteristicofplasmasprayingas al-readymentionedinliterature[27].Theporosityevolvesfromabout3% forHap+[Ag+]/[Sr2+]andHap+AgNP+Sr2+coatingsto5%for
Ag/Sr-Hapand7%fortheundopedcoatingHap.Itseemsthatthepresence of nitrateswithin thesuspensiondiminishestheporosity ofthe pro-ducedcoatings.Asimilartrendcan benoticedfortheroughness. In-deed,Sa parametersequalto4.4±0.1µmweremeasured forboth Hap+[Ag+]/[Sr2+]andHap+AgNP+Sr2+coatingsandhighervaluesof
about5.4±0.4µmwererecordedforbothHapandAg/Sr-Hap coat-ings.Thepresence ofsilveror silveroxideparticleswas clearly evi-dencedintobothAg/Sr-HapandHap+[Ag+]/[Sr2+]microstructures us-ingSEMimagesathighmagnificationinbackscatteringmode(Fig.5) Thesenanoparticlesweremainlylocatedatthesplats-poresbordersas showninFig.5b,supportingthehypothesisthatsilverdoesnot favor-ablysubstituteintheapatiticstructure.TheSEMimageanalysisofthese particlesresultedinameandiameterof230±95nmwhenthedoped powderisimplementedinthespraysystemand115±91nmwhen sil-verisintroducedasnitrateinthesuspension.Butconsideringthelimits oftheSEM,evensmallerparticlesizesaround1.5µmindiametercould bedetectedbySTEM.Finally,EDScartography(Fig.6)performedon theHap+[Ag+]/[Sr2+]confirmedtheincorporationofstrontiumtoan
extentdependingonthemeltingdegreeoftheparticleatimpact.Indeed, splatsshowedlowvariationsofstrontiumcontentfromonetotheother, probably duetotheimplementationprocesswhich ledtoavariable concentrationofstrontiumintohydroxyapatiteparticles,dependingon theirsizeandtheirmeltingdegree.TheEDSmappingperformedonthe Ag/Sr-Hapcoatingshowedontheotherhandahomogenous distribu-tionofstrontium(Fig.6cand6g)alloverthemicrostructure,confirming itssubstitutionintheapatite.
Elementalquantificationofthescrapedcoatingsrevealsalossofthe silvercontentduringplasmaspraying:74%lossoftheinitialsilver pro-portionfortheAg/Sr-Hapcoating,80%forHap+[Ag+]/[Sr2+]coating
Fig.1.X-Raydiffractogramsofundopedand doped synthesizedpowderscomparedwitha commercialfullycrystallisedhydroxyapatite.
Table2
CrystallographiccompositionobtainedbyRiRmethodandcrystallinityratioofcoatings(ISO 13,779-3[20]).
Hap Ag/Sr-Hap Hap + [A g + ]/[Sr 2+ ] Hap + Ag NP + [Sr 2+ ]
Annealed doped coating HA (wt%) 86 ± 4 63 ± 13 83 ± 2 86 ± 5 98 𝛽-TCP (wt%) – – – – 1.4 𝛼-TCP (wt%) 5.9 ± 1.9 33 ± 11 – – – TTCP (wt%) 7.4 ± 1.6 4.0 ± 2.2 15 ± 2 12 ± 4 – CaO (wt%) 0.7 ± 0.3 0.1 ± 0.2 1.9 ± 0.6 1.6 ± 0.6 1.0 R 1 (%) 71 ± 5 57 ± 11 68 ± 5 70 ± 4 90 Rp (%) 5.2 4.6 5.6 5.4 3.2
Fig.2. X-Raydiffractogramsofdopedand un-dopedrf-suspensionplasmasprayedcoatings.
ratioishighlyover1.667foreverycoatingentailingtheundoped pow-der(Hap,Hap+AgNP+Sr2+,Hap+[Ag+]/[Sr2+]).Theoneproducedfrom
thedopedpowderisveryclosetostoichiometrywhenconsideringthe (Ca+Sr+Ag)/PratioinsteadoftheCa/Pratio.Strontiumdidnotsuffer anysignificantloss,sinceitsconcentrationrangesfrom4.24±0.08to 4.62±0.07wt.%inthecoatings.
Thecontactanglesvarysignificantlyfromonecoatingtotheother, asdisplayedinTable3.Butoverall,theyallstandbelow60° whichis consideredastheupperthresholdtofavorizecellattachment accord-ingtoYeungetal.[35].Almosthydrophobicbehaviorcanbeassigned tocoatingsproducedfrom suspensionscontaining nitrates: 47.6° for
Hap+[Ag+]/[Sr2+]and38.8° forHap+AgNP+Sr2+.Incontrastcoatings
producedwiththeHapandAg/Sr-Happowdersaremorehydrophilic (17.8° and22.1° respectively).
3.2. Cationsrelease
ThekineticsofreleaseofCaandSrarereportedinFig.7aand7b respectively.BothfiguresrevealedthatalargereleaseofCaandSrin theacidicmediumwasnoticedduringthefirst500minofimmersion. After10htheconcentrationsofcalciumandstrontiumleveledoff to evolve very slowlyup to4days. At theend of theassay the
disso-Fig.3. FTIRspectraofthecoatings,with phos-phatePO43-bandsrepresentedwiththe 𝜈1,𝜈2,
𝜈3 and𝜈4 signsandOH− bandsrepresented
with𝜈sOH−and𝜈LOH−.
Fig.4. SEMimagesofrespectivelycrosssectionsandtopsurfacesofHap(a,e),Ag/Sr-Hap(b,f),Hap+[Ag+]/[Sr2+](c,g),Hap+AgNP+[Sr2+](d,h)coatingsat
x500magnification.
Fig.6.STEMmicrographofHap+[Ag+]/[Sr2+](a)andAg/Sr-Hap(e)coatingcross-sectionandrespectiveEDSmappingofCa(b-f),Sr(c-g)andAg(d-h).
Table3
Physicalandelementalcharacteristicsofcoatings.
Sa (µm) Porosity (%) wt.% Ag wt.% Sr (Ca + Sr) /P Ø Ag np.(nm) Contact angle (°)
Hap 5.4 ± 0.4 6.7 ± 3.1 0 0 – 0 18 ± 4
Ag/Sr-Hap 5.4 ± 0.3 5.0 ± 2.3 0.394 ± 0.004 4.62 ± 0.07 1.69 ± 0.02 231 ± 95 22 ± 13 Hap + Ag NP + [Sr 2+ ] 4.4 ± 0.1 3.0 ± 1.5 0.064 ± 0.002 4.23 ± 0.08 1.79 ± 0.02 810 ± 646 39 ± 12 Hap + [A g + ]/[Sr 2+ ] 4.4 ± 0.1 2.5 ± 1.6 0.342 ± 0.005 4.55 ± 0.06 1.80 ± 0.02 115 ± 91 48 ± 7
Table4
Mechanicalandbiologicalpropertiesofcoatings.
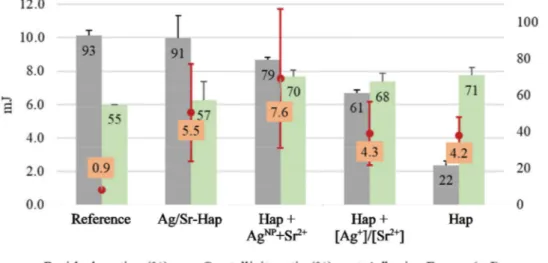
Adhesion energy (mJ) Residual coating proportion (%) Cell viability CV after 14 days (%) Antibacterial reduction BR at 24 h (%) APS Reference 0.9 ± 0.1 93 ± 3 100 – Hap 4.2 ± 1.1 22 ± 3 105.4 – Ag/Sr-Hap 5.5 ± 2.9 91 ± 13 99.42 2.9 ± 1.2 Hap + Ag NP + [Sr 2+ ] 7.6 ± 4.2 79 ± 2 38.06 1.2 ± 0.9 Hap + [A g + ]/[Sr 2+ ] 4.3 ± 1.9 61 ± 2 99.97 5.0 ± 1.2
lutionofAg/Sr-Hapcoatinghasreleasedabout4.5timesmore stron-tiumand1.5timesmorecalciumcomparedwiththecoatingelaborated fromHap+[Ag+]/[Sr2+].TheproportionofSr2+averages5at.%ofthe
cations(Ca2+,Sr2+andAg+)intheAg/Sr-HapandHap+[Ag+]/[Sr2+]
coatings.Butthis ratioreaches24 at.%and9.4 at.%inthe dissolu-tionmediumforAg/Sr-HapandHap+[Ag+]/[Sr2+]respectively,which
meansthatstrontiumispreferentiallyreleasedinthecaseofthe coat-ingmadefromthedopedpowder.AsAg/Sr-Haphasalowcrystallinity andahighproportionofsecondaryphasesevidencedbyXRDandFTIR analyses,itcanbeassumedthatstrontiumislikelyincorporatedinthe secondaryphases.Thisconclusionisreinforcedbythefactthatthese phasesareknowntobemoresolublethanhydroxyapatiteina biolog-icalmedium.Somestudiesalsosuggestapreferentialincorporation,at
highconcentration,ofstrontiumintotheTCPstructure[36,37],thatisa crystallinephasepresentinquitehighproportionintheAg/Sr-Hap coat-ing.Finally,X-raydiffractogramsofthecoatingsurfacesrecordedafter dissolutionthatrevealedthedisappearanceofthesecondaryphasesfor allthecoatings,tendtoconfirmthisinterpretation.ThepHevolution curvesofthemediumincontactwiththedopedsamplesshowthesame trendwithaninitialburstfollowedbyasignificantdecreaseduringthe first500min,followedbyare-increaseandastabilizationatan inter-mediatelevelafter2000min.Theendvaluesdiffer,with6.07forthe annealedsample,6.85forHap+[Ag+]/[Sr2+]and7.02forAg/Sr-Hap.
ThekineticofreleaseofAginthemediumisverydifferentfromthe onesofstrontiumorcalciumandcanbecomparedwiththepH evolu-tioncurve.SilverstronglydissolveswhenthepHisthelowest,andas
Fig.7. Releasedcumulatedquantityofcalcium(a),strontium(b)andsilver(c)inthedopedcoatingandpHevolutionwithtime(d).
pHstartsrisingdue tothedissolutionof thephosphocalcicmatrix,it stopsdissolvingandseemstoprecipitate,ashighlightedbybothcurves (Fig.7cand7d).
3.3. Mechanicalproperties
Theresultsofthe3-pointbendingtestappliedonthesampleswere evaluatedincomparisonwiththecommercialAPSplasma-sprayed hy-droxyapatitecoating(Table4).Allrf-SPScoatingsshowadhesion ener-gies4.6to8.4timesbetterthantheone(0.9mJ)ofthecommercial coat-ing,withthemaxvalue(7.6mJ)measuredforHap+AgNP+Sr2+coating.
Nospecifictrendcanbedrawnexceptthatthestandarddeviation in-creaseswith the value itself. Commercialcoating as well as Ag/Sr-HapandHap+AgNP+Sr2+coatingsexhibitedacohesiverupture(over
60%ofthecoatingisstillattachedtothesubstrateafterthetest).For Hap+Ag+/Sr2+amixedmode(61%)rupturewasidentifiedandforthe
undopedcoatingHapitwasanadhesive(22%)rupture.Nodirect cor-relationbetweentheadherenceenergyvaluesandthefailuredynamics wereestablished.
3.4. Invitrocell-materialsinteraction
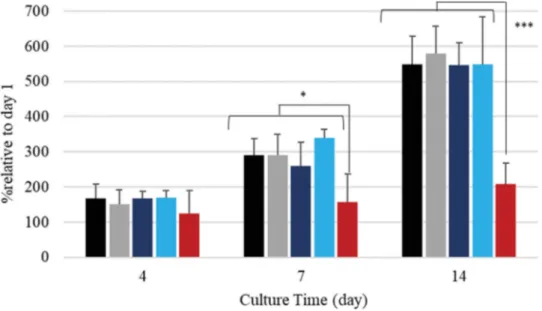
Cellproliferationresultsonthesamplesafter4,7and14daysof incubationareshowninFig.8,TabIV.Ateachtimeinterval,thenumber
ofcellsonundoped(HapandReference)coatingsanddoped(Ag/Sr-Hap andHap+[Ag+]/[Sr2+])coatingswasquiteidenticalandconsiderably
greaterthanthatonHap+AgNP+[Sr2+]coating.Theproliferationrates
reachedmorethan500%oftheirinitialpopulationafter14days,with aslightlybetterresultforHapcoatingthanforthereference.Atday7, Hap+[Ag+]/[Sr2+]showedapromisingburst(340%)sinceitovertook
bothundoped coatings(290%),butthis resultwasnot confirmedat 14days.Theadditionofsilverasnanoparticlesinthesuspensionhad asignificantnegativeimpactoncellproliferation,diminishingthecell viabilitybelow70%.
3.5. Antimicrobialactivity
Bactericidal potential of our coatings toward S. aureus was ex-plored, anddifferent behaviours emerged depending on the doping route(Fig.9).Theundopedcoatings(ReferenceandHap), as antici-pated,didnotshowanyantibacterialeffectivenessandnosignificant differencecouldbedetectedbetweenthem,implyingthatthechange ofmicrostructuralscaledoesnotimpactthebacterialgrowth.If Ag/Sr-Hapgavetroublingresultsafter6hwithasignificantproliferationof bacteria,itfinallymettheantibacterialexpectationswithareduction
BRof2.9after24h(Table4).Hap+[Ag+]/[Sr2+]coatingproveditself
veryefficientsinceitreducedthenumberofviablecellsbyalmost5log after24hofinoculation.Ontheotherhand,Hap+AgNP+[Sr2+]coating
Fig. 8. Proliferation of hMSC with time on plasma-sprayedcoatings(n =5),∗P ≤0.05∗∗∗
P ≤0.001.
Fig.9. Bacterialproliferationof S. aureus on dopedandundopedcoatings(n =3),∗P ≤0.05 ∗∗P ≤0.01.
showedareductionBRof1.2only,whichisinsufficientfroma bacteri-cidalpointofview.
4. Discussion
4.1. Influenceofthesuspensionformulationonthecoatingcomposition
Thechemical stabilityof hydroxyapatite coatings in a biological mediumisessentialtoguaranteetheirsustainability.Amongthe char-acteristicsintimatelyrelatedtothisstability,thecrystallinityandthe phasepurityofthecoatingarepredominantsincetheydetermineboth itsresorbabilityanditsmechanicalproperties,andthose characteris-ticsaremainlydeterminedbythesprayingconditions.The decompo-sitionofstoichiometrichydroxyapatiteandtheformationofsecondary includingamorphousphasesareacommonoccurrenceinconventional plasmaspray.Thehightemperatureoftheplasmaarc andtherapid coolingatthesubstrateimpactleadtotheformationofdecomposition phases(TCP,TTCP,ACPandCaO),whichweakenthecoatingby
re-sorbing significantlyfasterin thebody thanhydroxyapatite[38,39]. Consequently,theISO13779:2018standardrequiresapercentageof thesecondaryphaseslowerthan30wt.%,andacoatingwitha crys-tallinityratiogreaterthan45%.TheX-raydiffractometrydatashownin
Fig.2andTable2indicatethatcoatingsmadewithasuspension contain-ingthedopingelementsasnitratesareconstitutedofTTCPandCaOas secondaryphasesincontrasttotheequivalentundopedHapcoatingthat contains𝛼-TCP,TTCPandCaOforasimilarconcentrationof
hydroxya-patite.Insuspensionplasmaspraying,thedisturbancecausedbythe ad-ditionofsubstancessuchasnitratesormetalnanoparticlesinthe disper-sivemediummodifiestheplasmathermalcharacteristicsandtherefore theheattreatmentsustainedbyphosphocalcicparticles.Theirdegreeof decompositioncanthusbelargelyaffected:inthepresentcase,nitrates increasethethermalfluxavailableleadingtohightemperaturephase formationwhilemaintaininghighcrystallinityratioandHA%as com-paredtoundopedcoating(Hap).Regardingthemicrostructure, param-eterssuchastheporosityandtheroughnessalsoseemtobeimpacted: thepresenceofnitratesandtheuseofanon-stoichiometricdoped
pow-derleadtoadecreaseofporosity,duetoahighermeltingdegreeinthe formercase,andtoalackofcrystallinitywhichfavorsthecompacityof thematteratimpactinthelattercase.ResearchhasshownthatHAisan idealplatformforcationicsubstitutionand,specifically,silverand stron-tiumionscansubstitutecalciumionintheapatitestructure.Asthe ad-ditionofsuchdopantscanalterthephysico-chemicalcharacteristicsof HA,onewouldhaveexpectedpeakshiftsintheXRDanalysisofthe coat-ingsindicatingsignificantincorporationofthedopingionsintheHA latticestructureduetotheuseofeitherthedopedpowder(Ag/Sr-Hap) orthedopedmedium(Hap+AgNP+[Sr2+]orHap+[Ag+]/[Sr 2+]).While
nopeakshiftswereobservedfordopedcoatings,itremainslikelythatSr wasincorporatedinoneofthecalciumphosphatelatticestructureasno separateSrorSrOpeaksweredetectedinthediffractograms.Incontrast, identificationofAgorAg2Opeaksinthediffractogramsandthe obser-vationofthecoatingmicrostructureevidencedthatsilverisnot incor-poratedintheHAlatticestructure.HighmagnificationSEMandSTEM observationsclearlyconfirmedthatsilverisincorporatedas nanoparti-clesinthedopedcoatings,whosesizesdependonthedopingmethod. Indeed,itsionicradius14%higherthanthatofcalcium(128ppm ver-sus112ppmrespectivelyforacoordinationnumberCN=8,[40,41]) combinedwiththechargemismatchrevealtheinstabilityoftheAg+
ionintotheapatitestructure,eitherpreventingitsincorporationduring sprayingorleadingtoitsmigrationoutoftheapatiticstructure,which probablyinducesthesignificantdropofcrystallinitynoticedfor Ag/Sr-Hapcoating.Duetothisphenomenon,alargeamountofsilverdopant islostduringspraying(mainlybecauseofthetemperatureandpressure thatfavorvolatilizationofmetallicsilver)andtheexactamountpresent intothecoatingishardlycontrollable.Toconfirmthechemicalstateof silverinHap+AgNP+[Sr2+]andHap+[Ag+]/[Sr2+]coatings,XPS
anal-ysiswasperformedoncoatingswitha10timeshighersilver concen-trationinordertodetectsilveranddeconvolutethedoublet3d3/2 et 3d5/2 (367,75 eVfor3d5/2 et373,75eVpour3d3/2).Itshowedthat silvernanoparticlesintheHap+[Ag+]/[Sr2+]coatingare90%metallic
and10%oxidized,whiletheyare65%metallicand35%oxidizedinthe Hap+AgNP+[Sr2+]coating.AccordingtotheEllingham’sdiagram,fora
partialoxygenpressureof0.062bar,silveroxideisreducedto metal-licsilveratatemperatureabove398K(approximately125°C).During plasma-spraying,mostparticlesreachmuchhighertemperaturesonthe probe-substratepath.Butoutofthermodynamicequilibrium,the ther-malenergytransmittedtoreducethesilveroxidetometallicsilveris limitedbydynamicconsideration,duetoshortresidencetimeofthe droplets/particles.ForsuspensionscontainingAgNPsilvernanoparticles
andconsideringthatthesenanoparticlesarealreadypartlyoxidized be-foreplasma-spraying,theirconversiontofullymetallic nanoparticles isfurther limitedbecauseof theirlowersurface/volumeratiowhich doesnotfavorthethermaltransfer.ForthedopingmodeinvolvingAg+
ions,andtakingintoaccounttheinstabilityofsilveroxideover metal-licsilverin thesprayingconditions, itcan be assumedthat mostof theionsareinstantlyreducedin metallicsilvereitherwithoutgoing throughanoxidizedstate,or undergoingamoreextensivereduction ofoxidizedparticlesduetotheirsmallersize.Becausethesilverpeak wastooweaktoperformaqualitydeconvolutionoftheXPSspectrum recordedforAg/Sr-Hapcoating,andsincetheconcentrationofsilver couldnotbesufficientlyincreasedintheAg/Sr-Happrecursorpowder, thechemicalstateofthesilverparticlescouldnotbeevaluated.Thanks totheelementalanalysisperformedbySTEM(Fig.6),itwasclearly es-tablishedthatstrontiumishomogeneouslyincorporatedinthecalcium phosphatelatticestructureregardlessofitsionicradius(126ppm),even thoughsomesplatsofdopedcoatingsproducedfromnitratesrevealed concentrationheterogeneitieswithstrontium-richerzoneslocatedatthe grainboundariesofthesplats.Fromtheelementalpointofview,the (Ca+Sr)/P(Table3)ratiosofHap+AgNP+[Sr2+]andHap+[Ag+]/[Sr2+]
arethehighestofall(1.79±0.02and1.80±0.02respectivelyagainst 1.73± 0.03fortheundopedcoating),validatingSrincorporationor phosphorusevaporationduringplasmaspraying.SinceAgleftthe
ap-atitic structure during plasma spraying, it was not included in the ratio.
4.2. Microstructureandmechanicalproperties
Thecoatingbondstrengthisascrucialasitschemistryforthe clini-calsuccessofloadbearingimplants.Theadhesionenergy(Table4)that wasdeterminedinthepresentstudyrepresentstheforcerequiredto de-tachthecoatingfromthesubstrate.Dependingonthefailuremode,the adhesionenergyreflectsthetenacityeitheroftheinterface,eitherofthe nearbymicrostructure.FortheundopedSPScoating(Hap)the percent-ageofresidualcoatingonlyreaches22%,correspondingtoanadhesive failure,whilefortheAPSreferenceitisabout93%,correspondingtoa cohesivefailure.Acohesivetypefailure(residualcoating>60%)means
thatthecohesionofthemicrostructureneartheinterfaceisweakerthan thecoating/substrateinterface.Indeed,itisgenerallythemostfragile areabecauseofthehigherproportionofamorphousphases,duetoa lowerlocalcrystallizationofthematerialcausedbyquenching.This lo-calizedamorphizationhasadoubleeffectontheadhesionenergy:on onehanditpromotestheanchoringofthecoatingbylimitingthe gener-ationofresidualstressesusuallygeneratedbythecrystallizationofthe material,butontheotherhanditweakensthecohesionofthe phospho-calcicmaterialitself[42].Thesetwoantagonisticeffectstherefore con-tributetodefinethefailuremodeandadhesionenergy.Theevolutionof thecrystallinityratiowiththefailuremodeandtheadhesionenergyis illustratedinFig.10.Therefore,foralowcrystallinityratioofthe coat-ing(Ag/Sr-Hap)theruptureistypicallycohesive,whileforahighratio (undopedHap),theruptureisadhesive.Inthecaseoftheundoped coat-ing,sinceitbrokeataloweradhesionenergythanthedopedAg/Sr-Hap coating,itcouldbeassumedthatthereisahigheramountofresidual stressesatitsinterface,certainlycausedbythehighstabilityofundoped hydroxyapatitethatfavorsafastercrystallization.Whenusingnitrates intheprecursorsuspension(Hap+[Ag+]/[Sr2+]andHap+AgNP+Sr2+),
thecrystallinityratiosaresimilar tothatoftheundopedcoating(67 and70%),buttheamountofhightemperaturesecondaryphases(TTCP andCaO)ishigher,whichmakestheruptureevolvefromadhesive to-wardsthemixedandcohesivetype(61%and79%residualcoating re-spectively).Itiscertainlythehighmeltingdegreeofthephosphocalcic particlescausedbytheuseofnitratesthatexplainstheimprovement ofthesurfaceofcontactwiththesubstrate,providingastronger inter-face.However,itwasfoundthattheadhesionenergyismuchhigherfor Hap+AgNP+Sr2+(7.6±4.2mJ)thanforHap+[Ag+]/[Sr2+](4.3±1.9
mJ).Sinceitsfailuremodeis moreadhesive,thedecrease ofthe ad-hesionenergyforHap+[Ag+]/[Sr2+]canbeattributedtoaweakening
oftheanchoringofthedeposit.Itis noteworthytomentionthatthe onlydistinguishingcharacteristicsbetweenthesetwocoatingsarethe concentrationandthesizeofthesilvernanoparticles.Itistherefore as-sumedthatthehigherproportionandthefinersizeofthenanoparticles resultingfromthesilvernitratesbothreducethecontactsurfaceand thusanchorageofthecoating.TheAg/Sr-HapandHap+[Ag+]/[Sr2+]
coatingsexhibitthesamesilverconcentrations,shape,anddispersion ofthesilvernanoparticlesaswellasstrontiumconcentrations,buttheir crystallinityratiosarefarapart.Ag/Sr-Hapistheleast crystallineof allcoatings,justifyingwhyitsinterfaceisstrongerandwhythefailure modeiscohesive.
4.3. Physicochemicalpropertiesandbiologicalbehavior
Regardingtheeffectof dopingoncellproliferation,itseemsthat cellsproliferatepreferentiallyontheundopedcoatings(578%forHap and 549% for Reference at day 14). Among the doped substances, Hap+[Ag+]/[Sr2+]andAg/Sr-Hapcoatingsarethemostfavorable(549
and546%respectively),whiletheHap+AgNP+[Sr2+]coatingshowsan intermediatebehavior(209%).Asavalueofcellviabilitybelow70% reportsapotentialcytotoxicmaterial, thiscoatingdoesnotmeetthe
Fig.10. Evolutionofthemechanical proper-tiesofthecoatingswiththecrystallinityratio R1.
requirements.Thismeansthatthecellsproliferatemorefavorablyon thesurfaces wheresilvernanoparticlessizesaresmallanduniformly distributedthanonthosewithlargeragglomerates,withlittleeffectof thesilvercontent.WorkconductedbyAlbersetal.[10]statesthat sil-verharmfulnessismainlycarriedbytheAg+ions.Inaddition,ithas
beenshowninotherstudiesthatthechemicalstabilityofthecoating influencescellproliferation[43].Thisisalsopossiblythecasewith sil-ver,especiallysinceitsdissolutiondependsonthepHofthemedium, whichinturndependsontheamountofOH−andPO
43−ionsreleased
fromthephosphocalcicmatrix.Indeed,silverisfurtherdissolvedwhen thephosphocalcicmatrixis poorlysolublebecausethepHis main-tainedlongeratlowvalues attheperipheryofthesurface,as demon-stratedwiththeannealeddopedsample.Thiscoatingreleasesless cal-cium,strontium,phosphateandhydroxylionsthantheothersbecause ofitshighcrystallinityandhydroxyapatiteproportion.Butitreleases significantlymore silvereventhough theinitialAg concentrationof thedepositissimilar(0.313±0.007wt.%).Crystallinestatethusfirst determinesthesolubilitydegreeofthecoating,solubilitythatwanbe increasedby alarge exchange surfacewiththe medium,defined by roughnessandporosity. Asevidencedduringthedissolutiontest,the solubilityofdopedcoatingsevolvesinthefollowingorder:Ag/Sr-Hap
>Hap+[Ag+]/[Sr2+](≈Hap+AgNP+[Sr2+]).Thisexplainswhy
Ag/Sr-Hapcoatingdoesnotblockcellproliferation:itslowcrystallinity(57%) combinedwitharelativelyhighporosity(5%)isfavorabletothe disso-lutionofitsmatrixandtotheincreaseofpH,sothatitreleasesonlyvery fewAg+ions.ButwhentheCaPmatrixdissolvesquickly,silver
nanopar-ticlesdonot,graduallyconcentratingthematthesurface.Thefactthat cellularproliferationisnotcompromisedmeanscellsaremoresensitive toAg+fluxthantoAgnanoparticles,confirmingtheresultsofAlbers
etal.[10].FortheHap+[Ag+]/[Sr2+]coating,thesolubilityofwhichis
intermediate,thereleaseofAg+ionsisalittlemorepronounced,butthe
nanoparticlesofhomogeneousandfinegrainsizedistributionrelease Ag+ionsinasufficientlydilutedconcentrationnottoalertthecells.On
theotherhand,atequalphosphocalcicmatrixdissolutionandbecause ofthelargesizeofthesilvernanoparticlesdespitetheirlow concentra-tion,theAg+ionfluxescomingfromthesurfaceofHap+AgNP+[Sr2+]
arelocallyimportant,alertingthecellsandblockingtheirdivision.The antibacterialefficacyBRishighfortheHap+[Ag+]/[Sr2+]sample(5.0
at24h),duetoitshighsilverlevel(0.342wt.%)andfavoredAg+ion
release.Ag/Sr-HapcoatinghasaBRof2.9withasimilarsilver concen-tration(0.394wt.%)but,becauseofthehighsolubilityofthematrix, itsreleaseisslower.ThefactthatHap+[Ag+]/[Sr2+]hasahigher
ef-ficiencythanAg/Sr-HapsuggestsastrongereffectoftheAg+ionson
bacteriaincomparisonwiththemetallicAg(s)particlespresentonthe surface.Finally,Hap+AgNP+[Sr2+]samplehasthelowestvalueofBR
(1.2),probablyduetoitssilverconcentrationthatis5to6timeslower thanthepreviousones.
5. Conclusions
Inthisstudy,threesilverandstrontium-dopedhydroxyapatite coat-ingsweresynthesizedbyrf-SPS,implementingsuspensionscontaining strontiuminitsionicstate(freeorincorporatedintheHappowder)and silverundermetallicorionicstateanddispersedeitherinthepowder precursororintheliquidmedium.Strontiumandsilverwerechosen inordertopromotemesenchymalcellproliferationwhiledisablingthe developmentofS.aureusresistantbacteria.Suspensionplasmaspraying providesaccesstoaversatilitythatconventionalplasmasprayinglacks. Bychangingtheformulationoftheprecursorasbytheadditionof ni-tratesormetalnanoparticlesinthedispersingmedium,onecandirectly influencecharacteristicssuchasporosity,crystallinityorcomposition. Somebiologicalpropertiesastheantibacterialefficiencyseemstobe mainlydrivenbythesilvercontentandthereleaseextentofthe sil-verions,whilethehMSCaffinity forthesurfaceseemsratherlinked tothechemicalstabilityofthephosphocalcicmatrixandthesizeof thesilvernanoparticles.TheHap+[Ag+]/[Sr2+]coatingistheonethat
combinesverygoodmechanicalproperties comparedtotheAPS ref-erencecoating,excellentaffinitiesforcells,andbactericidalactivity. Ithas alsocompliantphysicochemicalproperties (crystalline compo-sition,porosity),andapotentialgoodchemical stability.By incorpo-ratingthedopantsinionicformintheprecursor,thefineand homo-geneousdispersionofthedopantsatthemicrometricscalewithinthe apatitestructureisguaranteedanddoesnotcompromisethe mechani-calorbiologicalpropertiesofthephosphocalcicmatrix.Thisstudythus demonstratesthepotentialoftherf-SPStechniqueforthedevelopment ofnanostructured-hydroxyapatitecoatingswithcontrolledantibacterial andanti-inflammatorydoping.
DeclarationofCompetingInterest
Theauthorsdeclarethattheyhavenoknowncompetingfinancial interestsorpersonalrelationshipsthatcouldhaveappearedtoinfluence theworkreportedinthispaper.
Acknowledgements
ThisworkwassupportedbytheAgenceNationaledelaRechercheof FranceunderprojectnamedARCHICAP(ANR-15-CE19–0021)andthe RégionOccitanieinFranceunderprojectnamedREVAMITIC(CLE).
References
[1] ANSM, Etude des facteurs associés aux révisions sur prothèses totales de hanche (PTH) : rôle du mode d ’ancrage (cimentage) et des constituants prothétiques (couple de frottement) dans les révisions chirurgicales, 2015.
[2] Haute Autorité de Santé, Prothèse de hanche ou de genou : diagnostic et prise en charge de l’infection dans le mois suivant l’implantation, (2014) 1–134.
[3] G.A. Fielding, M. Roy, A. Bandyopadhyay, S. Bose, Antibacterial and biological char- acteristics of silver containing and strontium doped plasma sprayed hydroxyapatite coatings, Acta Biomater. 8 (2012) 3144–3152, doi: 10.1016/j.actbio.2012.04.004 . [4] Z. Geng, Z. Cui, Z. Li, S. Zhu, Y. Liang, Y. Liu, X. Li, X. He, X. Yu, R. Wang, X. Yang,
Strontium incorporation to optimize the antibacterial and biological characteristics of silver-substituted hydroxyapatite coating, Mater. Sci. Eng. C 58 (2016) 467–477, doi: 10.1016/j.msec.2015.08.061 .
[5] Elliott, Structure and Chemistry of the Apatites and Other, Elsevier, 1994 .
[6] J. Kolmas, E. Groszyk, D. Kwiatkowska-Rózycka, Substituted hydroxyapatites with antibacterial properties, Biomed. Res. Int. (2014) 2014, doi: 10.1155/2014/178123 . [7] S. Eto, S. Kawano, S. Someya, H. Miyamoto, M. Sonohata, M. Mawatari, First Clinical Experience With Thermal-Sprayed Silver Oxide-Containing Hydroxyapatite Coating Implant, J. Arthroplasty. 31 (2016) 1498–1503, doi: 10.1016/j.arth.2015.12.034 . [8] M. Trop, M. Novak, S. Rodl, B. Hellbom, W. Kroell, W. Goessler, Silver-
coated dressing acticoat caused raised liver enzymes and argyria-like symp- toms in burn patient, J. Trauma Inj. Infect. Crit. Care 60 (2006) 648–652, doi: 10.1097/01.ta.0000208126.22089.b6 .
[9] M.A. Hollinger, Toxicological aspects of topical silver pharmaceuticals, Crit. Rev. Toxicol. 26 (1996) 255–260, doi: 10.3109/10408449609012524 .
[10] C.E. Albers, W. Hofstetter, K.A. Siebenrock, R. Landmann, F.M. Klenke, In vitro cy- totoxicity of silver nanoparticles on osteoblasts and osteoclasts at antibacterial con- centrations, Nanotoxicology 7 (2013) 30–36, doi: 10.3109/17435390.2011.626538 . [11] M. Š upová, Substituted hydroxyapatites for biomedical applications: a review, Ce-
ram. Int. 41 (2015) 9203–9231, doi: 10.1016/j.ceramint.2015.03.316 .
[12]E.S. Thian , P.N. Lim , Z. Shi , B.Y. Tay , K.G. Neoh , Silver-doped apatite as a bioac- tive and an antimicrobial bone material, Key Eng. Mater. 493–494 (2012) 27–30 doi:10.4028/www.scientific.net/KEM.493-494.27 .
[13] C. Capuccini, P. Torricelli, F. Sima, E. Boanini, C. Ristoscu, B. Bracci, G. Socol, Strontium-substituted hydroxyapatite coatings synthesized by pulsed-laser deposi- tion : in vitro osteoblast and osteoclast response, Acta Biomater. 4 (2008) 1885– 1893, doi: 10.1016/j.actbio.2008.05.005 .
[14] C. Lindahl, S. Pujari-Palmer, A. Hoess, M. Ott, H. Engqvist, W. Xia, The influence of Sr content in calcium phosphate coatings, Mater. Sci. Eng. C 53 (2015) 322–330, doi: 10.1016/j.msec.2015.04.015 .
[15] D.S. Brauer, N. Karpukhina, G. Kedia, A. Bhat, R.V. Law, I. Radecka, R.G. Hill, D.S. Brauer, Bactericidal strontium-releasing injectable bone cements based on bioactive glasses, J. R. Soc. Interface (2012) 1–8, doi: 10.1098/rsif.2012.0647 . [16] J.T.B. Ratnayake, M. Mucalo, G.J. Dias, Substituted hydroxyapatites for bone regen-
eration : a review of current trends, J. Biomed. Mater. Res. Part B (2017) 1285–1299, doi: 10.1002/jbm.b.33651 .
[17] H. Qiao, G. Song, Y. Huang, H. Yang, S. Han, Si, Sr, Ag co-doped hydroxyap- atite/TiO2 coating: enhancement of its antibacterial activity and osteoinductivity, RSC Adv. 9 (2019) 13348–13364, doi: 10.1039/c9ra01168d .
[18] Y. Huang, X. Zhang, H. Zhang, H. Qiao, X. Zhang, T. Jia, S. Han, Y. Gao, H. Xiao, H. Yang, Fabrication of silver- and strontium-doped hydroxyapatite/TiO2 nanotube bilayer coatings for enhancing bactericidal effect and osteoinductivity, Ceram. Int. 43 (2016) 992–1007, doi: 10.1016/j.ceramint.2016.10.031 .
[19] International standard ISO_13779, Implants for surgery — Hydroxyapatite - Part 2: Coatings of Hydroxyapatite, (2008).
[20] International standard ISO_13779, Implants for surgery — Hydroxyapatite - Part 3: Chemical Analysis and Characterization of Crystallinity and Phase Purity, (2008). [21]T.J. Webster , C. Ergun , R.H. Doremus , R.W. Siegel , R. Bizios , Enhanced functions of
osteoblasts on nanophase ceramics, Biomaterials 21 (2000) 1803–1810 .
[22] M. Roy, A. Bandyopadhyay, S. Bose, Induction plasma sprayed Sr and Mg doped nano hydroxyapatite coatings on Ti for bone implant, J. Biomed. Mater. Res. Part B Appl. Biomater. 99 (2011) 258–265 B, doi: 10.1002/jbm.b.31893 .
[23] M. Roy, G.A. Fielding, H. Beyenal, A. Bandyopadhyay, S. Bose, Mechani- cal, in vitro antimicrobial, and biological properties of plasma-sprayed silver- doped hydroxyapatite coating, ACS Appl. Mater. Interfaces 4 (2012) 1341–1349, doi: 10.1021/am201610q .
[24] P. Fauchais, Suspension and solution plasma spraying, J. Phys. D Appl. Phys. 46 (2013) 14pp, doi: 10.1088/0022-3727/46/22/224015 .
[25] S. Kannan, J.M.F. Ferreira, Synthesis and thermal stability of hydroxyapatite- 𝛽-tricalcium phosphate composites with cosubstituted sodium, magnesium, and flu- orine, Chem. Mater. 18 (2006) 198–203, doi: 10.1021/cm051966i .
[26]P. Ducheyne , Comprehensive Materials, Elsevier Science„ 2011 .
[27] R.B. Heimann, Plasma-sprayed hydroxylapatite-based coatings: chemical, mechani- cal, microstructural, and biomedical properties, J. Therm. Spray Technol. 25 (2016) 827–850, doi: 10.1007/s11666-016-0421-9 .
[28] M. Roy, A. Bandyopadhyay, S. Bose, Induction plasma sprayed nano hydroxyapatite coatings on titanium for orthopaedic and dental implants, Surf. Coatings Technol. 205 (2011) 2785–2792, doi: 10.1016/j.surfcoat.2010.10.042 .
[29] E. Bouyer, F. Gitzhofer, M.I. Boulos, Suspension plasma spraying for hydroxyapatite powder preparation by RF plasma, IEEE Trans. Plasma Sci. 25 (1997) 1066–1072, doi: 10.1109/27.649627 .
[30] R. d’Haese, L. Pawlowski, M. Bigan, R. Jaworski, M. Martel, Phase evolution of hydroxapatite coatings suspension plasma sprayed using variable parame- ters in simulated body fluid, Surf. Coatings Technol. 204 (2010) 1236–1246, doi: 10.1016/j.surfcoat.2009.10.022 .
[31] K. Geels, D.B. Fowler, W.-.U. Kopp, M. Rückert, Material/Preparation Ta- bles —Methods C-01/T-01 to C-68/T-68, in: Metallographic and Materialographic Specimen Preparation, Light Microscopy, Image Analysis, and Hardness Testing, 2006, pp. 223–226. https://books.google.cl/books?id = oaehZy3Vo1kC .
[32]A.A. Roche , A.K. Behme , J.S. Solomon , A three-point flexure test configuration for improved sensitivity to metal, J. Adhes. Adhes. (1982) 249–254 .
[33] International standard ISO 14679:1997, Adhésifs - Détermination des caractéris- tiques d’adhésion par une méthode de flexion à trois points, (1997).
[34] L. Berzina-Cimdina, N. Borodajenko, Research of calcium phosphates using fourier transform infrared spectroscopy, in: Infrared Spectroscopy - Materials Science, En- gineering and Technology, 2012, pp. 123–148, doi: 10.5772/36942 .
[35] W.K. Yeung, G.C. Reilly, A. Matthews, A. Yerokhin, In vitro biological response of plasma electrolytically oxidized and plasma-sprayed hydroxyapatite coatings on Ti-6Al-4V alloy, J. Biomed. Mater. Res. Part B Appl. Biomater. 101 (2013) 939–949 B, doi: 10.1002/jbm.b.32899 .
[36] H.W. Kim, Y.H. Koh, Y.M. Kong, J.G. Kang, H.E. Kim, Strontium substi- tuted calcium phosphate biphasic ceramics obtained by a powder pre- cipitation method, J. Mater. Sci. Mater. Med. 15 (2004) 1129–1134, doi: 10.1023/B:JMSM.0000046395.76435.60 .
[37] L. Leroux, J.L. Lacout, Preparation of calcium strontium hydroxyapatites by a new route involving calcium phosphate cements, J. Mater. Res. 16 (2001) 171–178, doi: 10.1557/JMR.2001.0028 .
[38]C. Combes , C. Rey , Biomatériaux à base de phosphates de calcium, Tech. l’ingénieur (2013) 1–25 n4950 .
[39] S.V. Dorozhkin, Calcium orthophosphates – Occurrence, properties, biomineraliza- tion, pathological calcification and biomimetic applications, Biomatter 1 (2) (2011) 121–164, doi: 10.4161/biom.18790 .
[40] D.R. Lide, Handbook of Chemistry and Physics, 2004. doi:10.1016/0022-2860(92)85083-s.
[41] A. Fihri, C. Len, R.S. Varma, A. Solhy, Hydroxyapatite: a review of syntheses, struc- ture and applications in heterogeneous catalysis, Coord. Chem. Rev. 347 (2017) 48–76, doi: 10.1016/j.ccr.2017.06.009 .
[42]G. Britain , J. Metauforschung , Fracture mechanics studies of thermal mismatch using a four-point bending specimen, Acta Metall. Mater 40 (1992) S345–S353 .
[43] G. Costa Machado, E. García-Tuñón, R.V. Bell, M. Alini, E. Saiz, M. Peroglio, Calcium phosphate substrates with emulsion-derived roughness: processing, characterisation and interaction with human mesenchymal stem cells, J. Eur. Ceram. Soc. 38 (2018) 949–961, doi: 10.1016/j.jeurceramsoc.2017.06.043 .

![Fig. 4. SEM images of respectively cross sections and top surfaces of Hap (a, e), Ag/Sr-Hap (b, f), Hap+[Ag + ]/[Sr 2 + ] (c, g), Hap+Ag NP +[Sr 2 + ] (d, h) coatings at x500 magnification.](https://thumb-eu.123doks.com/thumbv2/123doknet/14541313.535462/8.892.61.836.898.1092/fig-images-respectively-cross-sections-surfaces-coatings-magnification.webp)
![Fig. 6. STEM micrograph of Hap+[Ag + ]/[Sr 2 + ] (a) and Ag/Sr-Hap (e) coating cross-section and respective EDS mapping of Ca (b-f), Sr (c-g) and Ag (d-h).](https://thumb-eu.123doks.com/thumbv2/123doknet/14541313.535462/9.892.63.835.120.519/fig-stem-micrograph-coating-cross-section-respective-mapping.webp)