Line Tension Effect Upon Static Wetting
6
0
0
Texte intégral
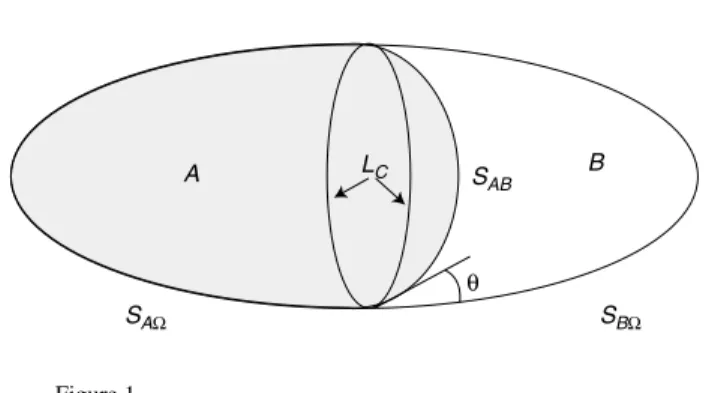
Figure
Documents relatifs