The effect of inulin and/or wheat bran in the diet during early life on intestinal health of broiler chicks
B. Li#, J. Leblois#, B. Taminiau※, L. Willems*, Y. Beckers#, J. Bindelle#, N. Everaert#
#Precision livestock and nutrition unit, Gembloux Agro-Bio Tech, TERRA, Teaching and Research Centre, University of Liège, Passage des Déportés, 2. 5030 Gembloux, Belgium-bli@student.ulg.ac.be;
* Molecular Biology, Gembloux Agro-Bio Tech, University of Liège, Passage des Déportés, 2. 5030 Gembloux, Belgium
FARAH - Department of Food Sciences – Microbiology, University of Liège, Avenue de ※
Cureghem 180, 4000 Liege, Belgium Abstract
Diets enriched in inulin (IN) and wheat bran (WB) were supplied to chicks from d-1 after hatch to investigate effects on the intestinal health and microbiota, individually and in combination. Four treatments: Control (CON), IN, WB, WB+IN were assigned with 6 replicate pens per treatment. On d7 and d11, average BW of chicks fed WB, WB+IN were significantly higher than that of chicks fed IN, CON. The WB+IN group also showed greater villus height (V), crypt depth (C) in the jejunum and ileum, and either the jejunal the ratio V/C. The WB group significantly increased jejunal villus height compared to CON group. In contrast, jejunal villus height and crypt depth were lower in IN group which also had a lower ileal crypt depth. Significantly higher expression of Claudin-1 in the jejunum and ileum was observed in IN group compared to WB, WB+IN groups. The abundance of Anaerostipes and Faecalibacterium were higher in IN group and the abundance of VadinBB60_unclassified was higher in WB+IN group compared to the other three groups. In addition, Flavonifractor, Intestinimonas were found lower in IN and WB+IN groups compared to CON group. In conclusion, WB and WB+IN as an ingredient of the starter diet could ameliorate growth performance and intestinal morphology, but weakened the protection of intestinal barrier during the starter period, while IN and WB+IN diet had a stronger effect on microbiota composition.
Introduction
Animal gut has a various and abundant microbiota, some of them can cause detrimental effects on host health and performance (Schauer.,1997; Farthing.,2004). The relationship between gut microbiota and host was considered to a balance between mutualism and pathogenicity (Farthing et al., 2004). Dietary strategies are focusing on the interaction between diet-microbiota in host which have proved by previous studies, but usually focused on finisher period on chickens (Torok et al., 2011). Another strategy, gaining more and more attention, is the modulation of the digestive tract and the colonization of the microbiota during starter period. Therefore, the aim in present study was to determine the effects of inulin and wheat bran, individually and in combination, from d-1 after hatch until d-11 (starter period) on intestinal health and microbiota. Materials and methods
Experimental design: Nine hundred and sixty 1-d old male broiler chicks (Ross 308) were housed in 24 floor pens covered with wood shavings as litter, each containing 40 birds. Pens were randomly assigned to 1 of the following 4 groups, (a) IN (4% inulin); (b) WB (10% wheat bran); (c) IN+WB (4% inulin+10% wheat bran); (d) CON (without inulin and wheat bran). Average BW were recorded on d0, d7 and d11. One bird per pen was euthanized and sampled on d-11. Cecal content and 5-cm jejuna, ileal segments were collected and snapfrozen in liquid nitrogen,
then stored at -80 °C for future analysis. Another 5-cm jejunal and ileal segments were taken and stored in 4% paraformaldehyde for intestinal morphology analysis.
Intestinal histomorphology: The paraffin sections and the measurement were performed like previous study (Rebolé et al., 2010).
Gene expression: Total RNA was isolated using Promega system (Z3741) and the RT-PCR process were performed by Takara system (RR037A and RR820L) according to the manufacturer’s instructions. The 2-ΔΔCt method was used automatically by ABI StepOnePlus
system to analyze target genes of a sample relative to this gene expression of the pool.
Gut microbiota analyses: DNA extraction of the cecal content was performed using the QIAamp DNA Stool Mini Kit (Qiagen). 16S rDNA profiling, targeting V1-V3 hypervariable region and sequenced on Illumina MiSeq as described previously (Neyrinck et al., 2016).
Statistical analysis: All data was subjected to analysis of variance using SPSS software, and significant differences among treatment means were determined by Duncan’s multiple range test. The data of intestinal morphology were analysed using nested designs for ANOVA models. The results were considered statistically significant when P <0.05.
Results and discussion
Table 1. Effect of inulin and/or wheat bran diets on the villus height, crypt depth, ratio V/C in the jejunum and ileum of broiler chicks on d-11
Values are
mean, 6
chicks per groups and 15 units of villus and crypt per chick, n=75-90 after unusual values being canceled. a-c Means in the same row not sharing a common superscript are significantly different (P< 0.05).
Average BW on d-7 and d-11 of chicks fed WB and WB+IN were significantly higher when compared to chicks fed IN and CON (unshown). Probably due to the excellent performance of gut histomorphology for WB and WB+IN diets. A shorter villi and deeper crypts may bring about a poor nutrient absorption (Xu et al., 2003). Obviously, WB+IN diet significantly increased all the parameters of histomorphology in the jejunum and ileum, except the ratio in the ileum and WB diet also significantly increased jejunal villus height. Conversely, the jejunal villus height and crypt depth in IN group was lower, compared to the other three groups, same as ileal crypt depth compared to CON diet. Previous studies reported WB and its derivative in the diet increased BW (JøRgensen et al., 1996; Courtin et al., 2008), but IN did not (Alzueta et al., 2010; Świątkiewicz et al., 2011) which supported our discovery. However, still some studies found beneficial BW gain and gut histomorphology in the IN treatment (Rebolé et al., 2010; Nabizadeh., 2012). These apparent contradictions might due to effects such as inclusion level, animal characteristics, composition of diet, husbandry conditions. Even inulin alone treatment did not
CON IN WB WB+IN SEM P Villus height, μm Jejunum 831.62c 760.11d 940.59b 1072.62a 9.11 <0.001 Ileum 462.64b 475.03ab 484.22ab 579.08a 3.52 <0.001 Crypt depth, μm Jejunum 152.90b 146.07c 149.90bc 164.25a 1.20 <0.001 Ileum 126.31b 119.25c 120.94bc 133.75a 1.10 <0.001 Ratio V/C Jejunum 5.48b 5.21b 5.86ab 6.61a 0.19 0.042 Ileum 3.90 4.02 4.01 4.35 0.09 0.295
make a contribution to the development of host’s BW and gut histomorphology, but significantly higher expression of Claudin-1 in the jejunum and ileum was observed in IN group (Fold=0.72; 1.06) compared to WB (Fold=0.61; 0.70) and WB+IN (Fold=0.54; 0.67) groups, with CON group (Fold=0.63; 0.93) having intermediate values. Claudin-1 has been thought to create a primary barrier to restrict access to substances, prevent the attachment of pathogenic organisms. It seems that while wheat bran-containing treatment promoted growth, while also increased the risk of diseases (Dhawan et al., 2005; Turner., 2009).
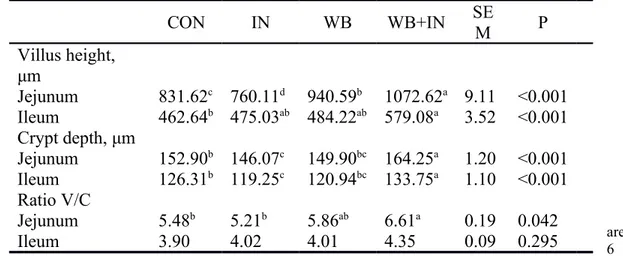
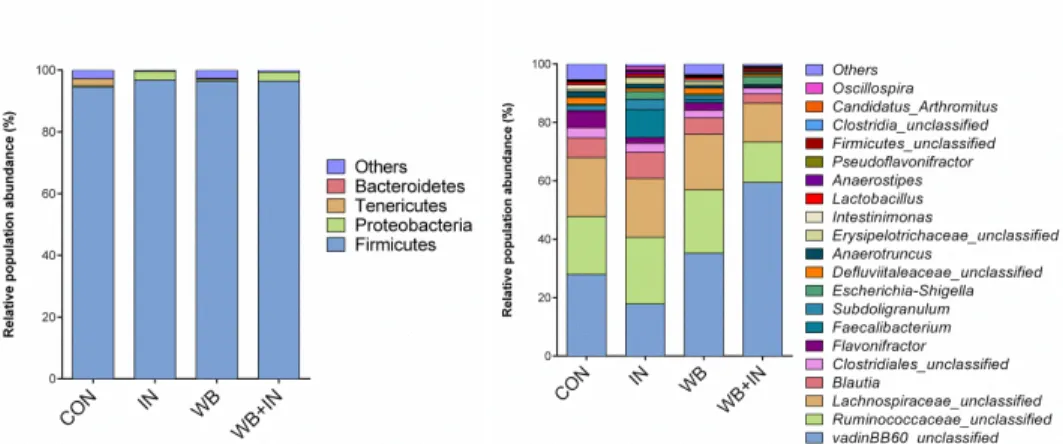
Figure 1: Mean phylotype distribution (phylum and genus levels) expressed as mean cumulated relative abundance in the cecal content of chicks on d-11.
Alpha-diversity showed no difference of bacterial diversity, richness and evenness among treatments (unshown). In our study, vadinBB60 is the most predominant genus and the abundant was lower in IN group (17.79%). Only Neyrinck et al (2016) found the inulin decreased vadinBB60 group in the mice which proved our result, but its function has not been reported. Moreover, the relative abundances of Anaerostipes (0.98%) and Faecalibacterium (9.56%) were higher in IN group compared to the other three groups, also Flavonifractor, Intestinimonas were lower in IN (0.67%; 0.33%) and WB+IN (0.36%; 0.26%) groups compared to CON group (5.71%; 2.04%). All these bacteria are related to SCFA, especially for butyrate (Schoefer et al., 2003., Sokol et al., 2008; Eeckhaut et al., 2010; Kläring et al., 2013). As prebiotics, IN and WB were the fermented in the host, there is no doubt that the composition of the relevant bacteria will be changed. Unfortunately, the weak correlation between these bacteria and dietary treatment, might due to the natural individual variations.
Conclusion
In conclusion, WB and WB+IN as an ingredient of the starter diet could ameliorate growth performance and intestinal morphology, but weakened the protection of intestinal barrier during the starter period, while IN and WB+IN diet had a stronger effect on microbiota composition. References
Farthing, M. J, 2004: Bugs and the gut: an unstable marriage. Best Practice & Research Clinical Gastroenterology. 18(2), 233-239.
Schauer, D. B, 1997: Indigenous microflora: paving the way for pathogens?. Current Biology. 7(2), R75-R77.
Torok, V. A., R. J. Hughes, L. L. Mikkelsen, R. Perez-Maldonado, K. Balding, R. MacAlpine, K. Ophel-Keller, 2011: Identification and characterization of potential performance-related gut microbiotas in broiler chickens across various feeding trials. Applied and Environmental Microbiology. 77(17), 5868-5878.
Rebolé, A., L. T. Ortiz, M. Rodríguez, C. Alzueta, J. Treviño, S. Velasco, 2010: Effects of inulin and enzyme complex, individually or in combination, on growth performance, intestinal microflora, cecal fermentation characteristics, and jejunal histomorphology in broiler chickens fed a wheat-and barley-based diet. Poultry Science. 89(2), 276-286.
JøRgensen, H., X. Q. Zhao, K. E. B. Knudsen, B. O. Eggum, 1996: The influence of dietary fibre source and level on the development of the gastrointestinal tract, digestibility and energy metabolism in broiler chickens. British Journal of Nutrition. 75(03), 379-395.
Courtin, C. M., W. F. Broekaert, K. Swennen, O. Lescroart, O. Onagbesan, J. Buyse, G. Huyghebaert, 2008: Dietary inclusion of wheat bran arabinoxylooligosaccharides induces beneficial nutritional effects in chickens. Cereal Chem. 85(5), 607-13.
Świątkiewicz, S., J. Koreleski, A. Arczewska-Włosek, 2011: Effect of inulin and oligofructose on performance and bone characteristics of broiler chickens fed on diets with different concentrations of calcium and phosphorus. British poultry science. 52(4), 483-491.
Alzueta, C., M. L. Rodriguez, L. T. Ortiz, A. Rebole, J. Trevino, 2010: Effects of inulin on growth performance, nutrient digestibility and metabolisable energy in broiler chickens. British poultry science. 51(3), 393-398.
Nabizadeh, A., 2012: The effect of inulin on broiler chicken intestinal microflora, gut morphology, and performance. Journal of Animal and Feed Sciences. 21(4), 725-734.
Turner, J. R., 2009: Intestinal mucosal barrier function in health and disease. Nature Reviews Immunology. 9(11), 799-809.
Dhawan, P., A. B. Singh, N. G. Deane, Y. No, S. R. Shiou, C. Schmidt, R. D. Beauchamp, 2005: Claudin-1 regulates cellular transformation and metastatic behavior in colon cancer. The Journal of clinical investigation. 115(7), 1765-1776.
Neyrinck, A. M., U. Etxeberria, B. Taminiau, G. Daube, M. Hul, A. Everard, N.M. Delzenne, 2017: Rhubarb extract prevents hepatic inflammation induced by acute alcohol intake, an effect related to the modulation of the gut microbiota. Molecular nutrition & food research. 61(1). Sokol, H., B. Pigneur, L. Watterlot, O. Lakhdari, L.G. Bermúdez-Humarán, J.J Gratadoux, C. Grangette, 2008: Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proceedings of the National Academy of Sciences. 105(43), 16731-16736.
Eeckhaut, V., F. Van Immerseel, F. Pasmans, E. De Brandt, F. Haesebrouck, R. Ducatelle, P. Vandamme, 2010: Anaerostipes butyraticus sp. nov., an anaerobic, butyrate-producing bacterium from Clostridium cluster XIVa isolated from broiler chicken caecal content, and emended description of the genus Anaerostipes. International journal of systematic and evolutionary microbiology. 60(5), 1108-1112.
Kläring, K., L. Hanske, N. Bui, C. Charrier, M. Blaut, D. Haller, T. Clavel, 2013: Intestinimonas butyriciproducens gen. nov., sp. nov., a butyrate-producing bacterium from the mouse intestine. International journal of systematic and evolutionary microbiology. 63(12), 4606-4612.
Schoefer, L., R. Mohan, A. Schwiertz, A. Braune, M. Blaut, 2003: Anaerobic degradation of flavonoids by Clostridium orbiscindens. Applied and environmental microbiology. 69(10), 5849-5854.
Xu, Z. R., C. H. Hu, M. S. Xia, X. A. Zhan, M. Q. Wang, 2003: Effects of dietary fructooligosaccharide on digestive enzyme activities, intestinal microflora and morphology of male broilers. Poultry science. 82(6), 1030-1036.