Anomalous Behaviour of a Protein During Sds/Page Corrected by Chemical Modification of Carboxylic Groups
Texte intégral
Figure
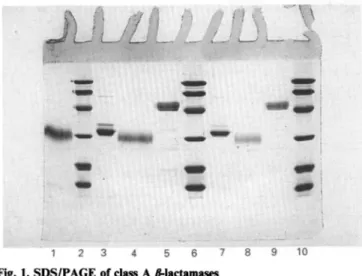
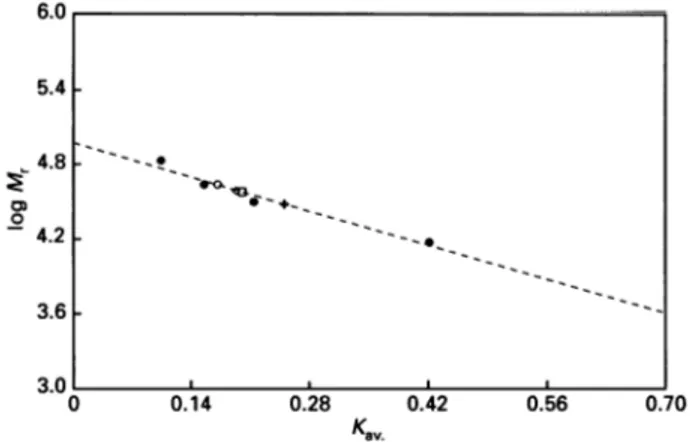
Documents relatifs
If environmental and housing conditions are responsible for these differences, the effect on performance are partly associated to the stimulation of the immune system
4b and 4c, which show also a small shift of the carboxylic stretching band to lower wave numbers, in agreement with an increase of the C-O bond length can be attributed to
number of amino acids (length), molecular weight, isoelectric point, FoldIndex [32], mean (reduced) net charge at pH 7, mean hydrophilicity [33], GRAVY (grand average of
RhoA-V14 produced a robust decrease in BRET compared with vector- transfected cells (Fig. 5a), indicating conformational change, and activation of the biosensor, as shown by in
Où que j'aille, dans n'importe quelle ruelle, sur n’importe quelle place, d'un bâtiment à l'autre, tout demeurait obstinément désert et calme comme si un
Try to learn new words not in isolation but in phrases. Write down adjectives together with nouns they are often associated with and vice versa, e.g. royal family; rich
Sensitivity analysis showed that changes in treatment efficacy produced effects only for less-well proven treatments (spray, inhaler, and bupropion) and revealed a strong influence of
In Theorems 1.5 through 1.8 we prove that the above Diophantine classes remain generic or null when additional assumptions on the frac- tal (and the measure supported on it)