This article appeared in a journal published by Elsevier. The attached
copy is furnished to the author for internal non-commercial research
and education use, including for instruction at the authors institution
and sharing with colleagues.
Other uses, including reproduction and distribution, or selling or
licensing copies, or posting to personal, institutional or third party
websites are prohibited.
In most cases authors are permitted to post their version of the
article (e.g. in Word or Tex form) to their personal website or
institutional repository. Authors requiring further information
regarding Elsevier’s archiving and manuscript policies are
encouraged to visit:
Author's personal copy
JointBoneSpine81(2014)281–283
Available
online
at
www.sciencedirect.com
Editorial
Does
signaling
pathway
inhibition
hold
therapeutic
promise
for
osteoarthritis?
a
r
t
i
c
l
e
i
n
f
o
Keywords: Osteoarthritis Arthritis Cartilage Chondrocytes CytokinesOsteoarthritis was long viewed as a degenerative joint dis-easecharacterizedbygradualcartilageattrition.Mechanicalfactors werevery often incriminated and patient managementlimited to pain control. Subsequently, osteoarthritis was defined as a family of diseases involving all the articular and periarticular tissues,includingthemusclesandtendons[1].Therolefor syn-ovitisandsubchondral-bonesclerosishasbeenfirmlyestablished. Research focusedonthejointtissues and documentedchanges in the metabolic activity and phenotype of chondrocytes, syn-oviocytes, and subchondral-bone osteoblasts. Several signaling pathways have been incriminated in these changes. Examples include the nuclear factor kappa-light-chain-enhancer of acti-vated Bcells (NF-B) pathway, whose activation by numerous cytokines(e.g.,IL-1)andgrowthfactorsupregulatesthe expres-sionofmanygenesinvolvedinconnectivetissueinflammationand breakdown;andtheNF-B/HIF-2␣pathway,whichplaysarolein endochondralossification.Activationof theB/HIF-2␣pathway by mechanical factorsor proinflammatory cytokines stimulates productionbythechondrocytesofosteogenicfactors,matrix met-alloproteinase(MMP)-13,andcollagentypeX[2].Anotherrelevant pathwayisJAKs/STATs,whichensures rapidsignaltransduction between membrane receptors and target genes. Janus kinases (JAKs) are involvedin activatingphosphorylation via the tran-scriptionfactorssignaltransducersandactivatorsoftranscription (STATs),whichmigratetothenucleus,where theybindtoDNA sequenceswithintargetgenepromoters.TheJAKs/STATspathway isactivatedbynumerouscytokinessuchasIL-6.JAKs/STATs acti-vationinducestheexpressionofgenesencodingproinflammatory cytokinesandMMPsdirectlyinvolvedincartilagebreakdownand synovialmembraneinflammation[3].Activationofthe Insulin/IGF-1/Pl3k/Akt/forkhead-box class O (FoxO) pathway is related to chondrocyteaging.FoxOfactorsplayacentralroleincell resis-tancetooxidativestress[4]andtheirinhibitionresultsindecreased antioxidantproduction.TheWntpathwayisalsoafocusofactive research.Itsroleiscomplex.Wntpathwayactivationandinhibition
induceosteoarthritisinexperimentalanimals[5].Thesepathways constitutepotentialtreatmenttargets.
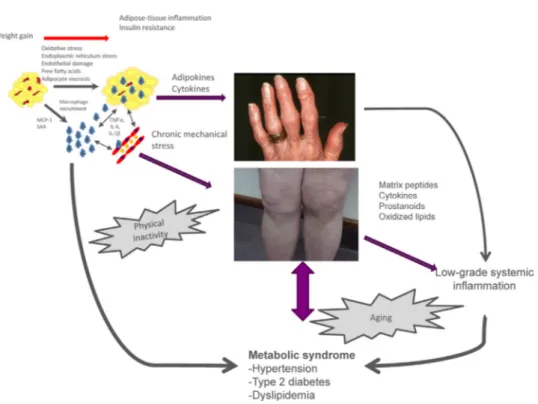
Morerecently,osteoarthritishasbeendescribedasametabolic disease,basedonevidenceofacorrelationbetweenclinicalhand osteoarthritisseverityandobesity[6].Obesitydoesnotincreasethe mechanicalloadsonthehands,andasystemiceffectofobesityon handosteoarthritishasthereforebeensuggested[7].Adiposetissue releasesproinflammatorycytokines(e.g.,tumornecrosisfactor-␣, IL-6)andadipokines(e.g.,leptin,adiponectin,andvisfatin),which exert deleterious effectsonjoint tissues (Fig. 1)[8]. Studies of patientswithobesityhaveshownassociationslinking osteoarthri-tistothemetabolicsyndromeanditscomponents(hypertension, dyslipidemia,andtype2diabetes).Furthermore,associationshave been reported between kneeosteoarthritis and metabolic syn-drome components such as type 2 diabetes and hypertension, independentlyfromthepresenceofobesityorotherknownrisk factorsforosteoarthritis[9].Finally,theaccumulationofmetabolic riskfactorssuchasoverweight,hypertension,dyslipidemia,and hyperglycemiaincreasestheriskofkneeosteoarthritis develop-mentandprogression[10].Thelinkbetweenmetabolicsyndrome andosteoarthritismaybechroniclow-gradesystemic inflamma-tioncharacterizedbyelevatedcirculatinglevelsofreactiveoxygen species,oxidizedlow-densitylipoproteins(LDLs),lipidmediators, oradipokines.
These recent datahave changed themanner in which both patients and healthcare professionals view osteoarthritis. Clini-cians andresearchers arenow seekingtoclassify patientswith osteoarthritis based on the disease phenotype. Potential cate-goriesincludemetabolicosteoarthritis,earlygenetic osteoarthri-tis, age-related osteoarthritis, and posttraumatic osteoarthritis. Phenotype-basedclassificationwillresultinmanagement strate-giestailoredtothediseasecharacteristics,therebyincreasingthe responseratestoeachtreatment.
Thetherapeuticobjectiveinpatientswithosteoarthritisisto controlthesymptomsviaacombinationofpharmacologicaland nonpharmacologicaltreatments[11].Themain nonpharmacolog-icaltreatmentsareweightlossandphysicalexercisedesignedto maintainmusclefunctionandtodecreasebodyfat.Nonsteroidal anti-inflammatorydrugsandparacetamolarethemostwidelyused drugsbutcarryahighriskofseveresideeffectsthatlimitstheiruse inpatientswithosteoarthritisandcomorbidities.
In contrastto patientswithrheumatoid arthritis (RA), those withosteoarthritishavenotyetbenefitedfromtreatment break-throughs capable of significantly slowing disease progression.
doi:10.1016/j.jbspin.2014.03.002
Author's personal copy
282 Editorial/JointBoneSpine81(2014)281–283
Fig.1.Pathophysiologyofosteoarthritis(OA)associatedwithobesity.MCP-1:monocytechemotacticprotein-1;SAA:serumamyloidA;IL-1:interleukin-1beta; TNF-␣:tumornecrosisfactoralpha;IL-6:interleukin-6.Sincearelationshipwasdemonstratedbetweenhandosteoathritisandobesity,obesity-relatedOAisconsideredasa metabolicdiseasesinvolvingsystemicbetweenthetwoconditionsthroughthereleaseofproinflammatorycytokinesbutalsoadipokineslikesleptin,adiponectotvisfatin whichcandirectlyactivatechondrocyteslikemechanicalstressorproinflammatorycytokines.Obesityalsopromotesanumberofco-morbidities,includingthemetabolic syndrome,whichmayincreasetheriskofOA.Finally,OA,particularlyage-relatedOA,isassociatedtochroniclow-gradeinflammationthroughthemediatorreleasedby jointtissuesbutalsobyotherageingtissue.
BiotherapiestargetingIL-1 or TNF have shownlittle efficacyin patientswithhand or kneeosteoarthritis [12]. In addition,the severesideeffectsofthesetargetedtreatmentslimitthe accept-abilityoftheirlong-termuse,astheyaredisproportionatewiththe severityofosteoarthritis.Nevertheless,patientswith osteoarthri-tismaybenefitfromtherapeuticadvancesachievedinRA,notably fromthedevelopmentofsmallmoleculesthatspecificallyinhibit theJAKs.AnexampleisCP-690,550(tofacitinib).JAK/STAT path-wayinhibitionisnowatreatmenttargetnotonlyinRA,butalso inosteoarthritis.TofacitinibinhibitstheactivityofJAKs1,2,and3, exhibitinggreateraffinityforJAK3thanforJAKs1and2. Tofac-itinibhasbeen evaluatedin six phase IIstudies and six phase IIIstudiesincludingabout5000patientswithRA[3].Theresults showedsignificantdecreasesinsymptoms anddisease activity, evenin patientshavingfailedprior biotherapies[13]. The inci-denceofsideeffects(infectionsandgastricperforation)wassimilar tothatseenwithbiotherapies[14].Toourknowledge,tofacitinib hasnotyetbeenevaluatedinpatientswithosteoarthritis. Nev-ertheless,theresultsoftrialsinRAsuggestthattofacitinibmay eventuallybecomethefirstspecificsignalingpathwayinhibitor forevaluationinpatientswithosteoarthritis.Finally,therecently identifiedRunx1inhibitorTD-198946preventsthedevelopment ofcartilagedamagein micewithosteoarthritis inducedby sur-gical meniscectomy and section of the median knee ligament [15].
Signaling pathways hold promise as treatment targets in osteoarthritis,astheirinhibitionoractivationregulatesthe expres-sion of a set of target genes directly involved in joint tissue metabolic dysfunctions. Nevertheless, the first clinical trials in RAhaveshownthatspecificregulationoftheJAK/STATpathway inducesnumerous side effects.Thus, safety concerns maylimit theuseofspecificinhibitorsoractivatorsofsignalingpathways inosteoarthritis.Therisk/benefitratiomustremainatthecenter oftreatmentdecisions,asosteoarthritisisaslowlyprogressiveand
moderatelyseverediseasethatisassociatedwithmultiple comor-bidities.
Disclosureofinterest
YHisthefounderandashareholderofArtialisSAandSynolyne Pharma,twospin-offoftheUniversityofLiège.Hehasreceived consultingfeesfromTilmanSAandtheLaboratoiresExpanscience. HehasalsoreceivedspeakerfeesfromIBSAandBioIberica.
References
[1]LoeserRF,GoldringSR,ScanzelloCR,GoldringMB.Osteoarthritis:adiseaseof
thejointasanorgan.ArthritisRheum2012;64:1697–707.
[2]SaitoT,KawaguchiH.HIF-2alphaasapossibletherapeutictargetof
osteoarthri-tis.OsteoarthritisCartilage2010;18:1552–6.
[3]FeistE,BurmesterGR.SmallmoleculestargetingJAKs–anewapproachinthe
treatmentofrheumatoidarthritis.Rheumatology(Oxford)2013;52:1352–7.
[4]AkasakiY,HasegawaA,SaitoM,etal.DysregulatedFOXOtranscription
fac-torsinarticularcartilageinagingandosteoarthritis.OsteoarthritisCartilage
2014;22:162–70.
[5]LoriesRJ,CorrM,LaneNE.ToWntornottoWnt:theboneandjointhealth
dilemma.NatRevRheumatol2013;9:328–39.
[6]YusufE,NelissenRG,Ioan-FacsinayA,etal.Associationbetweenweightor
bodymassindexandhandosteoarthritis:asystematicreview.AnnRheumDis
2010;69:761–5.
[7]CondeJ,ScoteceM,LopezV,etal.Adipokines:novelplayersinrheumatic
diseases.DiscovMed2013;15:73–83.
[8]SellamJ,BerenbaumF.Isosteoarthritisametabolicdisease?JointBoneSpine
2013;80:568–73.
[9]PuenpatomRA,VictorTW.Increasedprevalenceofmetabolicsyndromein
individualswithosteoarthritis:ananalysisofNHANESIIIdata.PostgradMed
2009;121:9–20.
[10]YoshimuraN,MurakiS,OkaH,etal.Accumulationofmetabolicriskfactorssuch
asoverweight,hypertension,dyslipidaemia,andimpairedglucosetolerance
raisestheriskofoccurrenceandprogressionofkneeosteoarthritis:a3-year
follow-upoftheROADstudy.OsteoarthritisCartilage2012;20:1217–26.
[11]ZhangW,NukiG,MoskowitzRW,etal.OARSIrecommendationsforthe
man-agementofhipandkneeosteoarthritis:partIII:changesinevidencefollowing
systematiccumulativeupdateofresearchpublishedthroughJanuary2009.
Author's personal copy
Editorial/JointBoneSpine81(2014)281–283 283
[12]ChevalierX,EymardF,RichetteP.Biologicagentsinosteoarthritis:hopesand
disappointments.NatRevRheumatol2013;9:400–10.
[13]CoombsJH,BloomBJ, BreedveldFC,etal. Improvedpain,physical
func-tioning and health status in patients with rheumatoid arthritis treated
with CP-690,550, an orally active Janus kinase (JAK) inhibitor: results
fromarandomised,double-blind,placebo-controlledtrial.AnnRheumDis
2010;69:413–6.
[14]KremerJM,CohenS,WilkinsonBE,etal.AphaseIIbdose-rangingstudyofthe
oralJAKinhibitortofacitinib(CP-690,550)versusplaceboincombinationwith
backgroundmethotrexateinpatientswithactiverheumatoidarthritisandan
inadequateresponsetomethotrexatealone.ArthritisRheum2012;64:970–81.
[15]YanoF,HojoH,OhbaS,etal.Anoveldisease-modifyingosteoarthritisdrug
candidatetargetingRunx1.AnnRheumDis2013;72:748–53.
YvesHenrotin∗ UnitédeRecherchesurl’OsetleCartilage,Arthropôle
Liège,Institutdepathologie,niveau+5,CHU Sart-Tilman,4000Liège,Belgium
∗Tel.:+3243662516.
E-mailaddress:yhenrotin@ulg.ac.be Accepted27February2013 Availableonline4April2014