Activation-dependent modulation of B lymphocyte migration to chemokines
Texte intégral
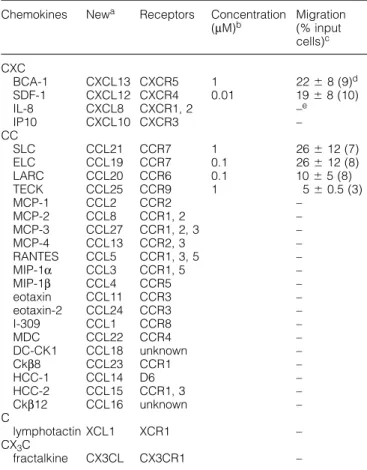
(2) 1286 B cell migration to housekeeping chemokines Table 1. Chemotactic migration of 12 h cultured B lymphocytes to chemokines Chemokines. CXC BCA-1 SDF-1 IL-8 IP10 CC SLC ELC LARC TECK MCP-1 MCP-2 MCP-3 MCP-4 RANTES MIP-1α MIP-1β eotaxin eotaxin-2 I-309 MDC DC-CK1 Ckβ8 HCC-1 HCC-2 Ckβ12 C lymphotactin CX3C fractalkine. Newa. Receptors. Concentration Migration (µM)b (% input cells)c. CXCL13 CXCL12 CXCL8 CXCL10. CXCR5 CXCR4 CXCR1, 2 CXCR3. 1 0.01. CCL21 CCL19 CCL20 CCL25 CCL2 CCL8 CCL27 CCL13 CCL5 CCL3 CCL4 CCL11 CCL24 CCL1 CCL22 CCL18 CCL23 CCL14 CCL15 CCL16. CCR7 1 CCR7 0.1 CCR6 0.1 CCR9 1 CCR2 CCR1, 2 CCR1, 2, 3 CCR2, 3 CCR1, 3, 5 CCR1, 5 CCR5 CCR3 CCR3 CCR8 CCR4 unknown CCR1 D6 CCR1, 3 unknown. 26 ⫾ 12 (7) 26 ⫾ 12 (8) 10 ⫾ 5 (8) 5 ⫾ 0.5 (3) – – – – – – – – – – – – – – – –. XCL1. XCR1. –. CX3CL. CX3CR1. –. 22 ⫾ 8 (9)d 19 ⫾ 8 (10) –e –. aNew nomenclature for chemokines defined at the recent Keystone Symposium on Chemokines and Chemokine Receptors (18–23 January 1999, Keystone, CO). bChemokine concentration for which migration data are shown. cB lymphocyte migration expressed as percentage of input cells that have migrated (100% ⫽ 105 cells). dSD denote donor-to-donor variations and numbers in brackets refer to the number of blood donor samples examined. eNo migration detected (⬍2% of input cells) when chemokines were tested in the range of 0.001–1 µM; six individual experiments were performed.. Current literature shows that both housekeeping and inflammatory chemokines act on B lymphocytes. The potent attractant function of SDF-1 (CXCL12) for B lymphocytes is well described (11–14). (The chemokines are defined by the new nomenclature as listed in Table 1.) In addition, SDF-1 induces mobilization of early B cell precursors in the bone marrow (CD34⫹ pro-/pre-B cells) (15–17) and its crucial role in B lymphopoiesis has been clearly demonstrated in mice that carried a defective gene for either SDF-1 or its receptor CXCR4 (18–21). Of note, CXCR4 is widely expressed on blood and tissue cells, indicating that SDF-1 and its receptor are also important in B lymphocyte-unrelated cell traffic. SLC and ELC which selectively bind to CCR7 are crucial regulators of T lymphocyte homing to secondary lymphoid tissues (22), and, in addition, were shown to induce B lymphocyte migration (12,14,23). B lymphocyte responses to inflammatory chemo-. kines are less clear. MIP-1α is the first chemokine with reported activity on B lymphocytes (24) but the MIP-1α receptors CCR1 and CCR5 were not detected on these cells (25,26). Also, IL-8 and MCP-1 were found to induce B lymphocyte migration and to express the corresponding cell surface receptors CXCR1/CXCR2 and CCR2 respectively (27,28), a finding that is still controversial (13,29). IL-8 and RANTES were shown to induce migration-unrelated cell responses, including B lymphocyte growth and modulation of IgE production (30–32). The recently discovered chemokine BCA-1 is the most efficacious B cell chemoattractant (33,34). It selectively binds to CXCR5, a chemokine receptor with predominant expression on blood and tonsillar B lymphocytes as well as a subpopulation of memory CD4⫹ T lymphocytes (35). CXCR5-deficient mice show reduced numbers of mostly rudimentary Peyer’s patches, have striking defects in splenic follicle formation and lack inguinal lymph nodes, underscoring the importance of BCA-1/CXCR5 in the recruitment of B lymphocytes to these locations (36). By contrast, other lymphoid tissues appeared to be normal. Here, we have investigated in detail the ability of BCA-1 to induce B lymphocyte responses and compared its effect with a large selection of housekeeping and inducible chemokines. Freshly isolated blood and tonsillar B lymphocytes are greatly compromised in their responsiveness which is overcome by short-term cell culture. Treatment with lipopolysaccharide (LPS) or anti-CD40/IL-4 greatly enhanced the migration response without markedly affecting chemokine receptor expression. These studies demonstrate the superior activity of BCA-1, SLC, ELC and SDF-1 on B lymphocytes. Much reduced activities were observed with LARC and TECK. By contrast, inflammatory chemokines appear to be of minor importance in B lymphocyte traffic since none of them was found to be active. The relevance of our findings for B lymphocyte traffic in secondary lymphoid tissues is discussed.. Methods Cytokines and antibodies All chemokines were chemically synthesized according to established protocols (50). Recombinant human IL-4 was purchased from PeproTech (Rocky Hill, NJ). The following monoclonal antibodies (mAb) and polyclonal antibodies were used: FITC-labeled mouse mAb to human CD3 (Sk78-11, IgG1; Becton Dickinson, Mountain View, CA), human CD19 (HD37, IgG1; Dako, Glostrup, Denmark), human CD71 (DF1513, IgG1) and human IgD (HJ9, IgG1; Sigma, St Louis, MO), phycoerythrin (PE)-labeled mouse mAb to human CXCR4 (44717.111, IgG2B) and CCR6 (53103.111, IgG2B; R & D Systems, Minneapolis, MN), mouse mAb to human CD40 (5C3, IgG1) and human CD20 (B8E9, IgG2a; PharMingen, San Diego, CA). MACS goat anti-mouse IgG MicroBeads were from Miltenyi Biotec (Bergisch Gladbach, Germany). For analysis of CXCR5 a rabbit polyclonal antibody was raised against a synthetic peptide corresponding to the N-terminal domain (amino acids 1–55) of human CXCR5, coupled to keyhole limpet hemocyanin. Rabbit anti-human CXCR5 pAb (purified IgG) or unspecific rabbit IgG (Zymed, San Francisco, CA) were detected with PE-labeled goat anti-rabbit IgG.
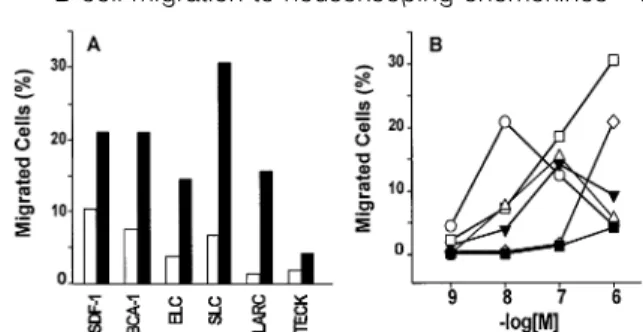
(3) B cell migration to housekeeping chemokines 1287 F(ab⬘)2 (P8172; Sigma) on a FACScan (Becton Dickinson). Selectivity of rabbit anti-human CXCR5 pAb was tested on CXCR5 transfected cells (33). No cross-reactivity was observed with CXCR1-4, CCR2-8, TYMSTR, BOB and HCR transfectants. Cells isolation and culture Mononuclear cell fractions from donor blood buffy coats or erythrocyte concentrates extracts were prepared by standard Ficoll-Hypaque gradient centrifugation and monocytes were depleted by adherence to plastic prior to isolation of B lymphocytes by positive selection with mAb to human CD20 and MACS MicroBeads. Purity of (CD19⫹) B lymphocyte preparations was in the range of 80–90%, as assessed by flow cytometry with mAb to human CD3, CD14 and CD19. Lymphocytes from human tonsil biopsies were isolated by grinding tissue material through a sieve and recovering the lymphocyte fraction by Ficoll-Hypaque gradient separation. Tonsillar B lymphocytes were then purified as described above (MACS MicroBeads). Human peripheral blood lymphocytes and monocytes were isolated from buffy coats or erythrocyte concentrates as described (41). B and T lymphocytes were cultured in RPMI 1640 plus 2 mM L-glutamine, 1% non-essential amino acids, 1% sodium pyruvate, 25 mg/ml penicillin/streptomycin, 5⫻10–5 M β-mercaptoethanol (Gibco, Paisley, UK) and 10% heat-inactivated FCS (Inotech/Biological Industries, Beit Haemek, Israel). B lymphocytes were stimulated by cell culture in the presence of 1 µg/ml of LPS or, alternatively, by addition of mAb to CD40 plus IL-4. Briefly, subconfluent cultures of the murine fibroblast cell line Ltk32 stably expressing CD32 (37) were inhibited by S. caespitosus mitomycin C treatment (Sigma; 2 µg/ml, 30 min at 37°C), washed and coated with 10 µg/ml mAb to CD40 (60 min at 37°C). B lymphocytes were added at 2⫻106 cells/ml and cultured in the presence of 10 ng/ml IL-4. Cell viability and proliferation was assessed by Trypan blue exclusion and CD71 staining respectively. T lymphocytes were stimulated by culturing in the presence of 250 U/ml IL-2 or 1 µg/ml phytohemagglutinin. B lymphocyte responses to chemokines In vitro chemotaxis was assessed in 48-well chambers (Neuro Probe, Cabin John, MD) using polyvinylpyrrolidone-free polycarbonate membranes (Poretics Corp., Livemore, CA) with 3 µm pores as described (41). Cell migration after incubation of the chemotaxis chambers for 120 min at 37°C (under CO2buffered conditions) was evaluated by counting adherent cells on the lower surface of the membranes in five randomly selected fields at 1000-fold magnification in triplicate wells for each chemokine concentration. Changes in the cytosolic free Ca2⫹ concentration ([Ca2⫹]i) were measured in Fura-2loaded cells as described (33,41). Chemokine receptor expression In flow cytometric studies, 5⫻105 cells were incubated for 30 min at 4°C with PBS/2% normal AB serum containing either PE-labeled mAb (CXCR4 and CCR6), FITC-labeled mAb (CD3, CD19 IgD and CD71) or unlabeled rabbit polyclonal IgG (CXCR5). After two washes with PBS, cell treated with unlabeled primary antibodies were incubated as above with. Fig. 1. Migration of freshly isolated and 12 h cultured B lymphocytes. (A) Enhanced migration of B lymphocytes after 12 h tissue culture. Freshly isolated (open bars) or 12 h cultured (filled bars) peripheral blood B lymphocytes were examined for migration to SDF-1 at 10 nM, ELC and LARC each at 100 nM, and BCA-1, SLC and TECK each at 1 µM; mean of triplicate values, expressed as percent of input cells that have migrated, of a representative experiment is shown. (B) Differences in the potency of chemokines in B lymphocyte chemotaxis. After 12 h of cell culture, B lymphocytes were examined for their migration responses to SDF-1 (s), SLC (u), ELC (n), BCA-1 (e), LARC (.) and TECK (j).One typical experiment out of four is presented; SD of triplicate values ⬍3%.. labeled secondary antibody, PE-labeled goat anti-rabbit IgG F(ab⬘)2, washed twice with PBS and analyzed using a FACScan (Becton Dickinson) and the CellQuest software. Lymphocyte subpopulations were gated by forward and side scatter analysis as well as lymphocyte marker (CD3, CD19) expression. For chemokine receptor transcript analysis, total cellular RNA was isolated from B lymphocytes using TriZol (GibcoBRL Life Technologies Inc.) and analyzed by ribonuclease protection using multiprobe templates (PharMingen) as instructed by the supplier. In short, 32P-labeled riboprobes from each template set was mixed with 5 µg RNA samples, incubated at 56°C for 12–14 h, and then treated with a mixture of RNases A and T1, and proteinase K. RNase-protected, 32P-labeled RNA fragments were separated on denaturing polyacrylamide gels and evaluated by PhosphorImager (Molecular Dynamics, Sunnyvale, CA) analysis. Northern blot analysis was performed as described (41). The 32P-labeled DNA probes (~109 c.p.m./mg DNA) that were used correspond to the 550 bp BamHI-cut 5⬘ end in CXCR4, the 550 bp BstXI-cut 5⬘ end in CXCR5, the 725 bp EcoRI-cut 5⬘ end in CCR6 and the 430 bp BstXI-cut 5⬘ end in CCR7. Results B lymphocyte migration to constitutively produced chemokines Human peripheral B lymphocytes were evaluated for their capacity to migrate in response to 27 human chemokines in the modified Boyden chemotaxis assay. B lymphocyte preparations obtained from buffy coats or erythrocyte concentrates consisted of 80–90% CD19⫹ cells with minor contaminations of CD3⫹ (3–6%) and CD14⫹ (7–14%) cells. Freshly prepared B lymphocytes responded poorly or not at all to the chemokines (Fig. 1A). Best responses were routinely obtained with SDF-1, inducing the migration of up to 10% of total input cells, followed by BCA-1, SLC and ELC (up to 7, 6 and 4% migrated cells respectively), whereas migration to LARC and TECK was consistently low but clearly detectable.
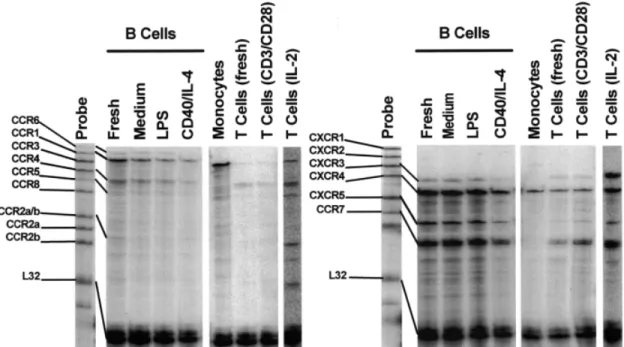
(4) 1288 B cell migration to housekeeping chemokines (up to 3 and 2% migrated cells respectively). Background migration (migration in the absence of chemokines) was ⬍1% migrated cells with freshly isolated B lymphocytes. No other chemokines, including a selection that covers all chemokine receptor selectivities as well as those with unknown receptors (Table 1), was active when tested at a concentration range of 1 nM to 1 µM. A similar situation was observed with freshly isolated tonsillar B lymphocytes (not shown). As with peripheral blood B lymphocytes, clear migration responses were obtained with SDF-1, BCA-1, SLC and ELC, followed by borderline activities with LARC and TECK, whereas all other chemokines listed in Table 1 were inactive. Marked upregulation of chemotaxis responses (⬎2-fold) was achieved when peripheral blood B lymphocytes were cultured for 12 h in the absence of stimulatory agents (Fig. 1A). The same set of chemokines was found to be active, indicating that culturing of B lymphocytes did not change their selectivity for chemokines. As shown by a representative experiment in Fig. 1(B), SDF-1 had the highest potency, inducing maximal migration at 10 nM, followed by ELC and LARC (maximal migration at 100 nM), whereas maximal responses to BCA-1, SLC and TECK required 1 µM or higher concentrations. Interestingly, ELC was routinely found to be more potent than SLC in B cell chemotaxis which may reflect different roles (transendothelial migration versus tissue localization) played by these chemokines in the recruitment of lymphocytes into secondary lymphoid tissues. An extensive evaluation performed with up to 10 individual B cell preparations per chemokine demonstrates that SDF-1, BCA-1, SLC and ELC are by far the most active B lymphocyte chemoattractants, inducing at optimal concentrations the migration of 19–26% of input cells (Table 1). Responses to LARC and TECK remained much smaller. Also, substantial donor-to-donor variations in B lymphocyte responsiveness was evident. Of note, the chemokines for which we demonstrated highest activity on B lymphocytes are known to be involved in the regulation of inflammationunrelated lymphocyte traffic. Transient up-regulation of B lymphocyte migration during in vitro stimulation Based on the modulatory effect of in vitro culture we next examined the influence of B lymphocyte activation on responsiveness to chemokines and chemokine receptor expression (see below). Freshly isolated peripheral B lymphocytes were treated with LPS or anti-CD40 mAb plus IL-4 and analyzed after 12, 36 and 72 h of culture, and cells cultured in the absence of stimuli were taken as point of reference. For optimal CD40 signaling, B lymphocyte preparations were cultured on monolayers of CD32-transfected murine fibroblast cells, Ltk32, that were coated with anti-CD40 mAb (37). Under both stimulatory conditions signs of B lymphocyte activation became evident during 72 h of culture, showing increased numbers of blastoid cells and enhanced CD71 expression (20 and 35% CD71⫹ cells in LPS- and anti-CD40/IL-4-stimulated B lymphocyte preparations respectively, as opposed to untreated cultures with ⬍5% of CD71⫹ cells; data not shown). Viability of stimulated or unstimulated B lymphocytes did not drop below 90%, as assessed by Trypan blue exclusion, and also the fraction of naive/memory (IgD⫹/IgD–) B lymphocytes remained unchanged during 72 h of culture. To avoid allo-. Fig. 2. Enhanced responsiveness to chemokines upon treatment of B lymphocytes with LPS or anti-CD40/IL-4. Peripheral blood B lymphocytes were cultured in the absence of stimuli (A), in the presence of LPS (B) or anti-CD40 plus IL-4 (C), and examined for migration to SDF-1 (s), SLC (u), ELC (n), BCA-1 (e), LARC (.) and TECK (j) after 12, 36 or 72 h of culture. Background migration in the absence of chemokines is also shown (⫻). Concentrations of chemokines were as used in Fig. 1(A). One typical experiment out of three is shown and SD of triplicate values ⬍3%.. geneic stimulation, B lymphocytes from each donor buffy coat or erythrocyte concentrate were cultured separately and then pooled just before running the in vitro chemotaxis assay. As shown in Fig. 2, culturing of B lymphocytes in the absence of stimulation did not substantially alter the migration responses to optimal concentrations of chemokines in the course of 12–72 h. By contrast, LPS or anti-CD40/IL-4 treatment greatly enhanced the B lymphocyte responsiveness to SDF-1, BCA-1, SLC and ELC. The effect of LPS was rapid, inducing maximal migration at 12 h of culture (30–50% migrated cells), whereas with anti-CD40/IL-4-stimulated cells maximal migration was seen at 36 h of culture (30–60% migrated cells). Of note, the observed stimulatory effect was transient and declined toward base level migration within 72 h of culture. By 5 or 7 days of culture, stimulated B lymphocytes were indistinguishable in their chemokine responsiveness from those cultured under non-stimulatory conditions (not shown). By contrast, responses to LARC and TECK were not affected by B lymphocyte stimulation and remained low (5–15% migrated cells). Again, as in unstimulated cultures, no additional chemokine was found to mobilize LPS- or antiCD40/IL-4-treated B lymphocytes. Transient increases in the intracellular Ca2⫹ concentrations ([Ca2⫹]i) are frequently taken as a measure of chemokine receptor signaling in leukocytes and the height of the [Ca2⫹]i responses were shown to correlate with receptor binding affinities of chemokines. However, none of the chemokines listed in Table 1 did induce [Ca2⫹]i mobilization in B lymphocytes and this unresponsiveness was not affected by LPS or anti-CD40/IL-4 treatment. In control experiments, [Ca2⫹]i signals to phorbol myristate acetate were readily observed (not shown). Chemokine receptor expression in B lymphocytes The large majority of freshly isolated B lymphocytes express chemokine receptors for SDF-1 (CXCR4), BCA-1 (CXCR5) and LARC (CCR6) as shown by flow cytometry (Fig. 3). We could not determine the expression of CCR7 (SLC and ELC) and CCR9 (TECK) since the corresponding antibodies were not available to us. The level of surface CXCR4 and CXCR5 did not change markedly during cell culture and was not.
(5) B cell migration to housekeeping chemokines 1289 Discussion. Fig. 3. Chemokine receptor expression on B lymphocytes. Flow cytometry was performed with antibodies to CXCR4, CXCR5 and CCR6 with freshly isolated or cultured B lymphocytes as described in Fig. 2. Filled histograms show staining with chemokine receptorspecific antibodies and open histograms are control stainings with isotype-matched antibodies.. influenced by cell culture under stimulatory conditions. By contrast, clear changes in surface staining were observed for CCR6 when B lymphocytes were treated with LPS or antiCD40/IL-4, resulting in 2.5- and 4-fold reduction in the corresponding mean fluorescence intensities. No other chemokine receptor for which antibodies were available was expressed at similar level as CXCR4, CXCR5 or CCR6 on stimulated or unstimulated B lymphocytes (except for CXCR3 on tonsillar B lymphocytes; data not shown). The expression of surface CXCR4 and CXCR5 correlates well with the level of receptor transcripts, and strong signals for CCR7, as observed by RNase protection analysis, indicates also prominent expression of this receptor (Fig. 4). By contrast, transcripts for CCR6 were much less abundant. Also, stimulation of B lymphocytes for 12 h with either LPS or anti-CD40/IL-4 did not markedly affect transcript expression when signals are adjusted for differences in RNA loadings (L32 expression). If anything, the level of receptor transcripts is rather reduced than enhanced upon B lymphocyte stimulation. In control experiments, RNA from T lymphocytes was analyzed by the RNase protection method. As expected, transcripts for CXCR4 and CCR7 are prominent in freshly isolated or short-term activated T lymphocytes, whereas transcripts of chemokine receptors for inflammatory chemokines (CCR1, CCR2, CCR5 and CXCR3), in addition to CXCR4 and CCR7, were readily observed in RNA from IL-2 cultured T lymphocytes. These results were confirmed by Northern blot analysis, showing strong hybridization with DNA probes for CXCR4, CXCR5 and CCR7, whereas weak but clearly detectable signals were observed with a DNA probe for CCR6 (not shown). Again, in vitro stimulation of B lymphocytes had no marked effect. Of note, RNase protection analysis did also reveal transcripts for CXCR3, CCR1 and CCR4 in B lymphocytes.. Freshly isolated blood or tonsillar B lymphocytes are greatly reduced in their capacity to respond to chemokines which resembles the variable but consistently low chemokine responses obtained with T lymphocytes when examined shortly after isolation from blood or inflamed tissues (38–40). Best responses of freshly isolated, uncultured B lymphocytes were routinely seen with SDF-1 inducing migration of up to 10% of total input cells, whereas migration to other chemokines was either moderate (BCA-1, SLC, ELC, LARC and TECK) or undetectable. Short-term culture under non-stimulatory conditions greatly enhanced the migration response, being at least 2-fold (SDF-1 and BCA-1) but reaching ⬎10-fold in some experiments for other chemokines (e.g. LARC and SLC). The reason for the enhancement effect is not clear to us at present but is certainly not related to chemokine receptor expression since the majority of freshly isolated B lymphocyte express surface CXCR4, CXCR5 and CCR6, and contain transcripts for many chemokine receptors, including CXCR4, CXCR5, CCR6 and CCR7. Clearly, chemokine receptor expression by itself is not sufficient for effective chemotaxis responses. Of note, short-term culture did not change the selection of chemokines to which B lymphocytes responded and, therefore, we screened the large selection of chemokines (Table 1) on B lymphocytes that were cultured for 12 h. The housekeeping chemokines BCA-1, SDF-1, SLC and ELC are the most efficacious attractants for cultured blood and tonsillar B lymphocytes. SDF-1 and ELC are the ones with highest potency, inducing maximal migration at 10–100 nM, whereas maximal responses to SLC and BCA-1 occurred at higher concentrations. Additional chemokines with reduced but consistently positive effects are TECK and LARC. No other chemokines (Table 1), including prominent members of all structural (CXC, CC, C and CX3C) and functional (inflammatory and housekeeping) subfamilies as well as those with unknown chemokine receptor selectivity, were found to be active on freshly isolated or overnight cultured (peripheral/ tonsillar) B lymphocytes. Responsiveness of T lymphocytes to chemokines is greatly influenced by cellular activation (1,2), but similar studies in B lymphocytes have not been performed. Here we show that cell activation by addition of LPS or via CD40 signaling (in the presence of IL-4) greatly increased B lymphocyte chemotaxis to BCA-1, SDF-1, SLC and ELC. The increase in responsiveness to chemokines was up to 3-fold, as compared to cultured but unstimulated B lymphocytes, reaching the impressive mobilization of 30–60% of total input cells. Evidently, activated B lymphocytes are prime targets for chemokine action, surpassing by far the migration responses seen with inflammatory chemokines on activated effector/memory T lymphocytes, monocytes or neutrophils. Furthermore, the migration enhancement was rapid (maximal migration at 12 or 36 h for stimulation with LPS or anti-CD40/IL-4 respectively) and transient, reaching base level migration at 36 h (LPS) or 72 h (anti-CD40/IL-4) of culture. Again, these effects were not due to changes in chemokine receptor expression. Moreover, we did not observe [Ca2⫹]i mobilization responses in B lymphocytes, regardless of origin (blood, tonsils) or state of activation (LPS, anti-CD40/IL-4), demonstrating that in vitro.
(6) 1290 B cell migration to housekeeping chemokines. Fig. 4. Chemokine receptor transcript analysis in freshly isolated or cultured B lymphocytes. Total RNA extracted from freshly isolated peripheral blood B lymphocytes (Fresh) or B lymphocytes that were cultured for 12 h in medium alone (Medium) or in the presence of either LPS or anti-CD40/IL-4 were analyzed by the RNA protection method. Each lane was loaded with 10 µg of RNA that was pooled from 10 independent B lymphocyte stimulation experiments. RNA from freshly isolated monocytes, T lymphocytes, anti-CD3/anti-CD28-treated (3 days) T lymphocytes or IL-2-treated (12 days; separate experiment) T lymphocytes were included as control. Shown are 32P-signals of protected chemokine receptor RNA probes obtained after hybridization with total cellular RNA, RNase digestion, PAGE and analysis by PhosphorImager.. cell migration does not depend on transient [Ca2⫹]i fluxes. In this respect, B lymphocytes differ from T lymphocytes, since [Ca2⫹]i mobilization responses to SLC are readily detected upon stimulation of peripheral blood lymphocytes with phytohemagglutinin or IL-2 (41). B lymphocyte responses to LARC and TECK were consistently low and remained unchanged during cell culture. Also, none of the other 21 chemokines tested were found to attract stimulated B lymphocytes, including MIP-1α, MCP-1 and IL-8 which were previously reported to be active (24,27,28). Our findings do not rule out that some of these chemokines are selective for subpopulations of B lymphocytes which were not present in sufficient numbers for chemotaxis analysis in our cell preparations. How do our findings relate to the recirculation properties of mature B lymphocytes? For initiation of a humoral response B lymphocytes need to enter secondary lymphoid tissues which involves the migration across specialized endothelia such as high endothelial venules (HEV) in lymph nodes (6,7,42). SDF-1, SLC and ELC were shown to induce rapid and transient integrin activation on T lymphocytes, and, thus, may also do so on B lymphocytes which express the corresponding receptors (43,44). Murine SLC is reported to be expressed on HEV and, therefore, this chemokine may play a pivotal role in the transendothelial migration process (45). In agreement, naive (IgD⫹) B lymphocytes from CCR7deficient mice show severely impaired homing into lymph nodes and Peyer’s patches of normal mice, and show aberrant retention kinetics in the periateriolar lymphoid sheath of wildtype spleen (22). However, some CCR7–/– B lymphocytes do enter lymph nodes and Peyer’s patches, and form large. secondary follicles in the spleen, indicating that the SLC/ELC system is not solely responsible for B lymphocyte homing into secondary lymphoid tissues. Once inside, B lymphocytes do not stay in the T cell-rich areas but migrate into the B cell follicles (46). Since chemokine binding induces rapid cellular desensitization that involves receptor inactivation (internalization and/or interruption of G-protein coupling), exposure of B lymphocytes to high levels of SLC and ELC present in the T cell-rich areas may shut down their responsiveness to these chemokines (12,14,41,45). Consequently, B lymphocytes may leave the T cell zones and enter B cell follicles which selectively express BCA-1 and SDF-1 (11,34,47,48). In this respect, BCA-1 plays a prominent role since B cell follicle formation is greatly impaired or even absent in lymph nodes and Peyer’s patches of CXCR5-deficient mice (36). This is in contrast to the large majority of T lymphocytes which lack CXCR5 and, consequently, are not recruited into B cell follicles (35).The germinal center reaction is initiated through T–B cell interactions in the follicular rim regions (46), a process which involves antigen binding to the B cell receptor, co-stimulation and T cell-derived cytokines (8,9). In this process CD40 signaling in B lymphocytes is a key event as evidenced by the absence of Ig class switching and abortive germinal center formation in patients with X-linked hyper-IgM syndrome (49). We show here that activation of B lymphocytes by antiCD40/IL-4 or LPS increases the efficacy of their migration response to chemokines which may be the mechanism for selectively trapping those B lymphocytes that are engaged in T–B cell interactions. Possibly, resting B lymphocytes which do not recognize the B cell antigen become desensitized by local BCA-1 and SDF-1, and, therefore, are not retained in.
(7) B cell migration to housekeeping chemokines 1291 the area of T cell contact. A similar situation was observed for T lymphocytes which show an activation-dependent enhancement of the migration responses to SLC and SDF-1 (14,41). Also, here, T lymphocytes that are engaged in antigen recognition may be retained in the T cell areas of secondary lymphoid tissues through enhanced responsiveness to SLC/ ELC, whereas resting cells become desensitized and, consequently, are free to leave this location. We show that the enhancement of B lymphocyte migration to chemokines is transient which, again, resembles the regulation of T lymphocyte responses to SLC and SDF-1 (14,41). Following T–B cell interactions, activated B lymphocytes migrate into germinal centers for further maturation or exit follicules as plasma cells, and reduction in responsiveness to chemokines may be the decisive factor for allowing this process to occur. In agreement, tonsillar B lymphocytes with naive or memory phenotype were found to migrate to SDF-1 whereas germinal center cells did not respond (11). By contrast to areas of T cell contact (follicular rim or mantle zones), BCA-1 and SDF-1 production is low in germinal centers, suggesting that some unknown chemokine(s) may recruit activated B lymphocytes to this microenvironment. The mechanism by which antigen-experienced cells, including memory and effector T/B lymphocytes, exit secondary lymphoid tissues is not known but may involve modulation of responsiveness to chemokines.. Acknowledgements The authors wish to thank R. Stuber Roos for expert technical help, Dr I. Clark-Lewis (University of British Columbia, Vancouver, Canada) for providing the chemokines, Dr A. Lanzavecchia (Basel Institute for Immunology, Basel, Switzerland) for the gift of recombinant human IL-2, Dr M. Caversaccio (University Hospital, Bern, Switzerland) for tonsil biopsies and Dr A. Zuercher (University Hospital, Bern, Switzerland) for Ltk32 cells. Donor blood buffy coats were provided by the Swiss Central Laboratory for Blood Transfusion Service, SRK, Bern, Switzerland. This work was supported by NF grant 31-055996.98 to B. M who is a recipient of a Professor Max Cloe¨ tta Foundation career development award.. Abbreviations BCA-1 ELC GC HEV LARC LPS PE PHA SDF-1 SLC TECK. B cell-attracting chemokine 1 EBI1 ligand chemokine germinal center high endothelial venule. liver and activation-regulated chemokine lipopolysaccharide phycoerythrin phytohemagglutinin stromal cell-derived factor 1 secondary lymphoid-tissue chemokine thymus-expressed chemokine. References 1 Loetscher, P., Moser, B. and Baggiolini, M. 2000. Chemokines and their receptors in lymphocyte traffic and HIV infection. Adv. Immunol. 74:127. 2 Baggiolini, M. 1998. Chemokines and leukocyte traffic. Nature 392:565.. 3 Sallusto, F., Lanzavecchia, A. and Mackay, C. R. 1998. Chemokines and chemokine receptors in T-cell priming and Th1/ Th2-mediated responses. Immunol. Today 19:568. 4 Melchers, F., Rolink, A. G. and Schaniel, C. 1999. The role of chemokines in regulating cell migration during humoral immune responses. Cell 99:351. 5 Butcher, E. C. and Picker, L. J. 1996. Lymphocyte homing and homeostasis. Science 272:60. 6 Springer, T. A. 1994. Traffic signals for lymphocyte recirculation and leukocyte emigration: The multistep paradigm. Cell 76:301. 7 Ager, A. 1997. Regulation of lymphocyte migration into lymph nodes by high endothelial venules. Biochem. Soc. Trans. 25:421. 8 MacLennan, I. C. 1994. Germinal centers. Annu. Rev. Immunol. 12:117. 9 Lindhout, E., Koopman, G., Pals, S. T. and De Groot, C. 1997. Triple check for antigen specificity of B cells during germinal centre reactions. Immunol. Today 18:573. 10 Tarlinton, D. 1998. Germinal centers: getting there is half the fun. Curr. Biol. 8:R753. 11 Bleul, C. C., Schultze, J. L. and Springer, T. A. 1998. B lymphocyte chemotaxis regulated in association with microanatomic localization, differentiation state, and B cell receptor engagement. J. Exp. Med. 187:753. 12 Ngo, V. N., Tang, H. L. and Cyster, J. G. 1998. Epstein–Barr virusinduced molecule 1 ligand chemokine is expressed by dendritic cells in lymphoid tissues and strongly attracts naive T cells and activated B cells. J. Exp. Med. 188:181. 13 Vicente-Manzanares, M., Montoya, M. C., Mellado, M., Frade, J. M. R., Del Pozo, M. A., Nieto, M., De Landazuri, M. O., Martı´nez, C. and Sa´ nchez-Madrid, F. 1998. The chemokine SDF1α triggers a chemotactic response and induces cell polarization in human B lymphocytes. Eur. J. Immunol. 28:2197. 14 Nagira, M., Imai, T., Yoshida, R., Takagi, S., Iwasaki, M., Baba, M., Tabira, Y., Akagi, J., Nomiyama, H. and Yoshie, O. 1998. A lymphocyte-specific CC chemokine, secondary lymphoid tissue chemokine (SLC), is a highly efficient chemoattractant for B cells and activated T cells. Eur. J. Immunol. 28:1516. 15 D’Apuzzo, M., Rolink, A., Loetscher, M., Hoxie, J. A., Clark-Lewis, I., Melchers, F., Baggiolini, M. and Moser, B. 1997. The chemokine SDF-1, stromal cell-derived factor 1, attracts early stage B cell precursors via the chemokine receptor CXCR4. Eur. J. Immunol. 27:1788. 16 Aiuti, A., Webb, I. J., Bleul, C., Springer, T. and Gutierrez-Ramos, J. C. 1997. The chemokine SDF-1 is a chemoattractant for human CD34⫹ hematopoietic progenitor cells and provides a new mechanism to explain the mobilization of CD34⫹ progenitors to peripheral blood. J. Exp. Med. 185:111. 17 Kim, C. H. and Broxmeyer, H. E. 1998. In vitro behavior of hematopoietic progenitor cells under the influence of chemoattractants: stromal cell-derived factor-1, steel factor, and the bone marrow environment. Blood 91:100. 18 Nagasawa, T., Hirota, S., Tachibana, K., Takakura, N., Nishikawa, S., Kitamura, Y., Yoshida, N., Kikutani, H. and Kishimoto, T. 1996. Defects of B-cell lymphopoiesis and bonemarrow myelopoiesis in mice lacking the CXC chemokine PBSF/ SDF-1. Nature 382:635. 19 Ma, Q., Jones, D. and Springer, T. A. 1999. The chemokine receptor CXCR4 is required for the retention of B lineage and granulocytic precursors within the bone marrow microenvironment. Immunity 10:463. 20 Tachibana, K., Hirota, S., Iizasa, H., Yoshida, H., Kawabata, K., Kataoka, Y., Kitamura, Y., Matsushima, K., Yoshida, N., Nishikawa, S., Kishimoto, T. and Nagasawa, T. 1998. The chemokine receptor CXCR4 is essential for vascularization of the gastrointestinal tract. Nature 393:591. 21 Zou, Y. R., Kottmann, A. H., Kuroda, M., Taniuchi, I. and Littman, D. R. 1998. Function of the chemokine receptor CXCR4 in haematopoiesis and in cerebellar development. Nature 393:595. 22 Fo¨ rster, R., Schubel, A., Breitfeld, D., Kremmer, E., Renner-Mu¨ ller, I., Wolf, E. and Lipp, M. 1999. CCR7 coordinates the primary immune response by establishing functional microenvironments in secondary lymphoid organs. Cell 99:23..
(8) 1292 B cell migration to housekeeping chemokines 23 Kim, C. H., Pelus, L. M., White, J. R., Applebaum, E., Johanson, K. and Broxmeyer, H. E. 1998. CKβ-11 macrophage inflammatory protein-3β EBI1-ligand chemokine is an efficacious chemoattractant for T and B cells. J. Immunol. 160:2418. 24 Schall, T. J., Bacon, K., Camp, R. D. R., Kaspari, J. W. and Goeddel, D. V. 1993. Human macrophage inflammatory protein α (MIP-1α) and MIP-1β chemokines attract distinct populations of lymphocytes. J. Exp. Med. 177:1821. 25 Wu, L. J., Paxton, W. A., Kassam, N., Ruffing, N., Rottman, J. B., Sullivan, N., Choe, H., Sodroski, J., Newman, W., Koup, R. A. and Mackay, C. R. 1997. CCR5 levels and expression pattern correlate with infectability by macrophage-tropic HIV-1, in vitro. J. Exp. Med. 185:1681. 26 Lee, B., Sharron, M., Montaner, L. J., Weissman, D. and Doms, R. W. 1999. Quantification of CD4, CCR5, and CXCR4 levels on lymphocyte subsets, dendritic cells, and differentially conditioned monocyte-derived macrophages. Proc. Natl Acad. Sci. USA 96:5215. 27 Tan, J. Q., Moller, B., Storgaard, M., Mukaida, N., Bonde, J., Grunnet, N., Black, F. T., Larsen, C. G., Matsushima, K. and Thestrup-Pedersen, K. 1997. Chemotaxis and IL-8 receptor expression in B cells from normal and HIV-infected subjects. J. Immunol. 158:475. 28 Frade, J. M. R., Mellado, M., Del Real, G., Gutierrez-Ramos, J. C., Lind, P. and Martinez, C. 1997. Characterization of the CCR2 chemokine receptor: Functional CCR2 receptor expression in B cells. J. Immunol. 159:5576. 29 Qin, S. X., LaRosa, G., Campbell, J. J., Smith-Heath, H., Kassam, N., Shi, X. J., Zeng, L., Butcher, E. C. and Mackay, C. R. 1996. Expression of monocyte chemoattractant protein-1 and interleukin-8 receptors on subsets of T cells: Correlation with transendothelial chemotactic potential. Eur. J. Immunol. 26:640. 30 Kimata, H., Yoshida, A., Ishioka, C., Lindley, I. and Mikawa, H. 1992. Interleukin 8 (IL-8) selectively inhibits immunoglobulin E production induced by IL-4 in human B cells. J. Exp. Med. 176:1227. 31 Kimata, H. and Lindley, I. 1994. Interleukin-8 differentially modulates interleukin-4- and interleukin-2-induced human B cell growth. Eur. J. Immunol. 24:3237. 32 Kimata, H., Yoshida, A., Ishioka, C., Fujimoto, M., Lindley, I. and Furusho, K. 1996. RANTES and macrophage inflammatory protein 1α selectively enhance immunoglobulin (IgE) and IgG4 production by human B cells. J. Exp. Med. 183:2397. 33 Legler, D. F., Loetscher, M., Roos, R. S., Clark-Lewis, I., Baggiolini, M. and Moser, B. 1998. B cell-attracting chemokine 1, a human CXC chemokine expressed in lymphoid tissues, selectively attracts B lymphocytes via BLR1/CXCR5. J. Exp. Med. 187:655. 34 Gunn, M. D., Ngo, V. N., Ansel, K. M., Ekland, E. H., Cyster, J. G. and Williams, L. T. 1998. A B-cell-homing chemokine made in lymphoid follicles activates Burkitt’s lymphoma receptor-1. Nature 391:799. 35 Fo¨ rster, R., Emrich, T., Kremmer, E. and Lipp, M. 1994. Expression of the G-protein-coupled receptor BLR1 defines mature, recirculating B cells and a subset of T-helper memory cells. Blood 84:830. 36 Fo¨ rster, R., Mattis, A. E., Kremmer, E., Wolf, E., Brem, G. and Lipp, M. 1996. A putative chemokine receptor, BLR1, directs. 37. 38. 39. 40 41. 42 43 44. 45. 46 47 48. 49. 50. B cell migration to defined lymphoid organs and specific anatomic compartments of the spleen. Cell 87:1037. Stuart, S. G., Trounstine, M. L., Vaux, D. J., Koch, T., Martens, C. L., Mellman, I. and Moore, K. W. 1987. Isolation and expression of cDNA clones encoding a human receptor for IgG (Fc gamma RII). J. Exp. Med. 166:1668. Loetscher, M., Loetscher, P., Brass, N., Meese, E. and Moser, B. 1998. Lymphocyte-specific chemokine receptor CXCR3: regulation, chemokine binding and gene localization. Eur. J. Immunol. 28:3696. Qin, S. X., Rottman, J. B., Myers, P., Kassam, N., Weinblatt, M., Loetscher, M., Koch, A. E., Moser, B. and Mackay, C. R. 1998. The chemokine receptors CXCR3 and CCR5 mark subsets of T cells associated with certain inflammatory reactions. J. Clin. Invest. 101:746. Loetscher, P., Uguccioni, M., Bordoli, L., Baggiolini, M., Moser, B., Chizzolini, C. and Dayer, J.-M. 1998. CCR5 is characteristic of Th1 lymphocytes. Nature 391:344. Willimann, K., Legler, D. F., Loetscher, M., Roos, R. S., Delgado, M. B., Clark-Lewis, I., Baggiolini, M. and Moser, B. 1998. The chemokine SLC is expressed in T cell areas of lymph nodes and mucosal lymphoid tissues and attracts activated T cells via CCR7. Eur. J. Immunol. 28:2025. Butcher, E. C. and Picker, L. J. 1996. Lymphocyte homing and homeostasis. Science 272:60. Campbell, J. J., Hedrick, J., Zlotnik, A., Siani, M. A., Thompson, D. A. and Butcher, E. C. 1998. Chemokines and the arrest of lymphocytes rolling under flow conditions. Science 279:381. Pachynski, R. K., Wu, S. W., Gunn, M. D. and Erle, D. J. 1998. Secondary lymphoid-tissue chemokine (SLC) stimulates integrin α4β7-mediated adhesion of lymphocytes to mucosal addressin cell adhesion molecule-1 (MAdCAM-1) under flow. J. Immunol. 161:952. Gunn, M. D., Tangemann, K., Tam, C., Cyster, J. G., Rosen, S. D. and Williams, L. T. 1998. A chemokine expressed in lymphoid high endothelial venules promotes the adhesion and chemotaxis of naive T lymphocytes. Proc. Natl Acad. Sci. USA 95:258. Garside, P., Ingulli, E., Merica, R. R., Johnson, J. G., Noelle, R. J. and Jenkins, M. K. 1998. Visualization of specific B and T lymphocyte interactions in the lymph node. Science 281:96. Cyster, J. G., Ngo, V. N., Ekland, E. H., Gunn, M. D., Sedgwick, J. D. and Ansel, K. M. 1999. Chemokines and B-cell homing to follicles. Curr. Top. Microbiol. Immunol. 246:87. Ngo, V. N., Korner, H., Gunn, M. D., Schmidt, K. N., Riminton, D. S., Cooper, M. D., Browning, J. L., Sedgwick, J. D. and Cyster, J. G. 1999. Lymphotoxin α/β and tumor necrosis factor are required for stromal cell expression of homing chemokines in B and T cell areas of the spleen. J. Exp. Med. 189:403. Kroczek, R. A., Graf, D., Brugnoni, D., Giliani, S., Korthuer, U., Ugazio, A., Senger, G., Mages, H. W., Villa, A. and Notarangelo, L. D. 1994. Defective expression of CD40 ligand on T cells causes ‘X-linked immunodeficiency with hyper-IgM (HIGM1)’. Immunol. Rev. 138:39. Clark-Lewis, I., Moser, B., Walz, A., Baggiolini, M., Scott, G. J. and Aebersold, R. 1991. Chemical synthesis, purification, and characterization of two inflammatory proteins, neutrophil activating peptide 1 (interleukin-8) and neutrophil activating peptide 2. Biochemistry 30:3128..
(9)
Figure
Documents relatifs
En deuxième lieu, L’onduleur Quasi Z-source sera inséré dans une installation photovoltaïque connectée au réseau électrique avec un système de stockage hybride
Band-modulation devices, which are the core of this thesis, offer not only a sharp switch but also very low leakage current, high current I ON /I OFF ratio, fast access time
L’expression actuellement utilisée dans l’Eurocode 5 ne prend pas en compte la longueur de fissure équivalente a ; afin de pouvoir comparer les différents modèles
The risk of developing conduction disturbances after coronary artery bypass grafting or valve replacement has been well established from previous studies, leading to permanent
The first exploits the fact that, according to the Gluckstern parametrization of relative error of particle’s momenta, the resolution is proportional to the mass of the
Our goal was to investigate spectral properties of molecular iodine in the region of 514.7 nm with the help of frequency doubled Yb-doped DFB fiber laser and with a
In addition to lymphocytes in draining lymph nodes, the lamina propria and the epithelium, the intestinal tract is equipped with specific lymphoid tissue structures.. This
Un tube d'eau se déplace à une vitesse ½ c par rapport à la référence du laboratoire lorsqu'un faisceau de lumière se déplace dans la même direction et entrer dans le tube..