Publisher’s version / Version de l'éditeur:
Vous avez des questions? Nous pouvons vous aider. Pour communiquer directement avec un auteur, consultez la
première page de la revue dans laquelle son article a été publié afin de trouver ses coordonnées. Si vous n’arrivez pas à les repérer, communiquez avec nous à PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca.
Questions? Contact the NRC Publications Archive team at
PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca. If you wish to email the authors directly, please see the first page of the publication for their contact information.
https://publications-cnrc.canada.ca/fra/droits
L’accès à ce site Web et l’utilisation de son contenu sont assujettis aux conditions présentées dans le site LISEZ CES CONDITIONS ATTENTIVEMENT AVANT D’UTILISER CE SITE WEB.
Internal Report (National Research Council of Canada. Division of Building
Research), 1985-09-01
READ THESE TERMS AND CONDITIONS CAREFULLY BEFORE USING THIS WEBSITE. https://nrc-publications.canada.ca/eng/copyright
NRC Publications Archive Record / Notice des Archives des publications du CNRC :
https://nrc-publications.canada.ca/eng/view/object/?id=3d3852a3-bb75-4413-92ac-de59104e51f4 https://publications-cnrc.canada.ca/fra/voir/objet/?id=3d3852a3-bb75-4413-92ac-de59104e51f4
NRC Publications Archive
Archives des publications du CNRC
For the publisher’s version, please access the DOI link below./ Pour consulter la version de l’éditeur, utilisez le lien DOI ci-dessous.
https://doi.org/10.4224/20338229
Access and use of this website and the material on it are subject to the Terms and Conditions set forth at
Method of determining the pH of urea formaldehyde foam insulation
NATIONAL RESEARCH COUNCIL OF CANADA
DIVISION OF BUILDING RESEARCH
DBR INTERNAL REPORT NO.
504METHOD OF DETERMINING THE pH OF UREA FORMALDEHYDE FOAM INSULATION by S.J. Rolfe and P.J. Sereda
ANALYZED
Checked by: Approvedby: L.W. Gold Date: September 1985
Prepared for: Records Purposes
ABSTRACT
This note describes the preliminary work that was carried out to establish a suitable method to measure the pH of UFFI foam.
METHOD OF DETERMINING THE pH OF UREA FORMALDEHYDE FOAM INSULATION by
S.J. Rolfe and P.J. Sereda
INTRODUCTION
UFFI foam
i s
produced when a component c o n t a i n i n g a n a c i d c a t a l y s t i s mixed w i t h a component c o n t a i n i n g p a r t i a l l y polymerized r e s i n . Thep r o p o r t i o n i n g i s accomplished a t t h e s i t e and i s s u b j e c t t o s i g n i f i c a n t v a r i a b i l i t y . The a c i d c a t a l y s t remains i n t h e s y s t e m a f t e r t h e foam h a s hardened and most of i t i s e x p e c t e d t o s t a y i n t h e foam a f t e r i t
i s
d r i e d , e x c e p t i n some c a s e s where p a r t of t h e w a t e r s o l u t i o n i s withdrawn as a l i q u i d t o a d j o i n i n g a b s o r b i n g m a t e r i a l s . It i s t h e r e s i d u a l a c i d t h a t remains on t h e c e l l w a l l s and p r o b a b l y i n s i d e t h e polymer forming t h e foam t h a t c a t a l y z e s t h e h y d r o l y s i s r e a c t i o n . The r a t e o f t h i s r e a c t i o n s h o u l d be dependent on t h e pH--
i n c r e a s i n g w i t h d e c r e a s i n g pH. The amount ofa v a i l a b l e a c i d w i l l remain c o n s t a n t , b u t i t s c o n c e n t r a t i o n w i l l d e c r e a s e as more w a t e r i s adsorbed. Thus t h e l o w e s t pH w i l l o c c u r a t some c o n d i t i o n of RH when a d e q u a t e f i l m s of w a t e r a r e p r e s e n t t o s o l v a t e a l l t h e a v a i l a b l e a c i d . T h i s may be a c o n d i t i o n t h a t cannot be measured w i t h a commercial g l a s s e l e c t r o d e d e s i g n e d f o r t h e measurement of s u r f a c e pH. However, i t i s i m p o r t a n t t o measure t h e l o w e s t v a l u e of pH i f p o s s i b l e because i t i s t h i s pH t h a t w i l l i n f l u e n c e t h e r a t e of h y d r o l y s i s .
This n o t e d e s c r i b e s t h e p r e l i m i n a r y work t h a t w a s c a r r i e d o u t t o e s t a b l i s h a s u i t a b l e method t o measure t h e pH of UFFI foam.
APPARATUS
I n i t i a l l y , s e v e r a l p o r t a b l e d i g i t a l and a n a l o g u e pH m e t e r s were t r i e d . The analogue model 74 p o r t a b l e meter k i t made by Markson Ltd. was found most s u i t a b l e . It employed a g l a s s , f l a t s u r f a c e e l e c t r o d e .
C u r r e n t l y , a d i g i t a l i n s t r u m e n t , I o n a l y z e r Model 601A, manufactured by Orion R e s e a r c h , c o u p l e d w i t h Corning S u r f a c e pH e l e c t r o d e Cat. No. 476216, i s i n use. T h i s a p p a r a t u s a p p e a r s t o be more s e n s i t i v e and c a n measure t h e pH even when t h e foam i s a t about 50% RH. Under t h e s e c o n d i t i o n s t h e
r e s p o n s e time ( t i m e t o s t a b i l i z e t h e r e a d i n g ) may be a s l o n g a s 20 seconds.
SAMPLE PREPARATION
Three t y p e s of t e s t specimens were p r e p a r e d :
1. Compacted d i s c s
-
shredded foam (produced by r u b b i n g foam a g a i n s t t h e s u r f a c e of a +-inch mesh T y l e r s i e v e ) was compacted a t a l o a d of25 000 l b f i n a s t e e l mould t o form d i s c s 31.7 mm i n d i a m e t e r and a b o u t 2 mm t h i c k . A mass of 1 t o 3 g of m a t e r i a l was used t o produce one specimen.
2. Packed foam i n p o l y e t h y l e n e b o t t l e s
-
s h r e d d e d foam was packed by hand, u s i n g a f l a t - b o t t o m l o o s e - f i t t i n g m e t a l c y l i n d e r , i n a f l a t - b o t t o m p o l y e t h y l e n e b o t t l e , 31mm
i n d i a m e t e r and 85 mm h i g h (equipped w i t h p r e s s f i t l i d ) . A mass of a b o u t 0.5 t o 1 g of foam was used f o r e a c h specimen. When packed i n t o t a r e d b o t t l e s , t h e s e specimens c a n be u s e d t o d e t e r m i n e t h e amount of w a t e r a d s o r b e d , a s w e l l a s t h e pH.3.
R e g u l a r foam u s e d a s specimens-
a p i e c e of UFFI foam h a v i n g a t l e a s t one u n i f o r m s u r f a c e and of a s i z e a l l o w i n g a t l e a s t t h r e e s e p a r a t e measurements t o be made, c a n s e r v e a s a specimen. Because t h e pH o f foam can be d i f f e r e n t a t t h e o u t s i d e s u r f a c e and i n t h e c o r e , i ti s
n e c e s s a r y t o p r o d u c e specimens r e p r e s e n t i n g b o t h o u t s i d e , i n s i d e ( c o r e ) and r e v e r s e s i d e . I f a p i e c e of foam of normal t h i c k n e s s i s t a k e n from a w a l l , t h e n i t s t h i c k n e s s c u t i n h a l f , one h a s s u r f a c e s t h a t c o r r e s p o n d t o o u t s i d e and i n s i d e f a c e s as w e l l a s t h e core.E x p e r i e n c e w i t h t h i s t e s t series h a s shown t h a t specimens of t h e t y p e d e s c r i b e d i n
( 3 )
a r e most u s e f u l , w h i l e t h o s e d e s c r i b e d i n ( 2 ) a r e u s e f u l f o r p r o v i d i n g more r e p r e s e n t a t i v e pH v a l u e s and a d e t e r m i n a t i o n of t h e m o i s t u r e c o n t e n t . The compacted specimens (Type 1 ) a r e l e a s t u s e f u l and i n most c a s e s n o t j u s t i f i e d .TEST PROCEDURE
Three t e s t methods
were
found f e a s i b l e :1. The w e t e l e c t r o d e method on r e l a t i v e l y d r y m a t e r i a l
--
u s i n g u n d i s t u r b e d specimens (Type 3). The pH e l e c t r o d e i s w e t t e d by p l a c i n g a d r o p of d i s t i l l e d w a t e r on t h e s u r f a c e o r by d i p p i n g t h e e l e c t r o d e i n t od i s t i l l e d w a t e r and s h a k i n g o f f t h e e x c e s s . It i s t h e n p r e s s e d i n t o t h e s u r f a c e of t h e foam, making a d e p r e s s i o n of 2-4 mm, t h u s l o c a l l y
compacting t h e foam. The s t a b l e pH v a l u e i s r e c o r d e d . T h i s o p e r a t i o n i s r e p e a t e d i n t h e immediate v i c i n i t y t o g i v e a t l e a s t t h r e e v a l u e s . More v a l u e s s h o u l d be t a k e n i f t h e v a r i a t i o n between i n d i v i d u a l r e a d i n g s i s t o o g r e a t .
2. Dry e l e c t r o d e on h u n d d i f i e d m a t e r i a l
--
u s i n g s h r e d d e d (Type 2 ) o r u n d i s t u r b e d (Type 3) which have been exposed t o a b o u t 95%RH
c o n d i t i o n s i n a d e s i c c a t o r o r o t h e r s u i t a b l e c o n d i t i o n i n g chambers. It d o e s n o t a p p e a r n e c e s s a r y t o w a i t w i t h s u c h c o n d i t i o n i n g beyond 1 o r 2 d a y s b u t t h e optimum t i m eseems
t o v a r y f o r d i f f e r e n t samples. There i s more d i s c u s s i o n of t h i s i n t h e s e c t i o n on c o n d i t i o n i n g .3. Measurement of pH of l e a c h i n g s o l u t i o n
--
a 0.1-g specimen of s h r e d d e d UFFIi s
d i s p e r s e d i n 10 mL of d i s t i l l e d w a t e r f o r 24 h o u r s . The pH of t h e s o l u t i o n i s t h e n measured by immersing t h e e l e c t r o d e .CONDITIONING OF SAMPLES
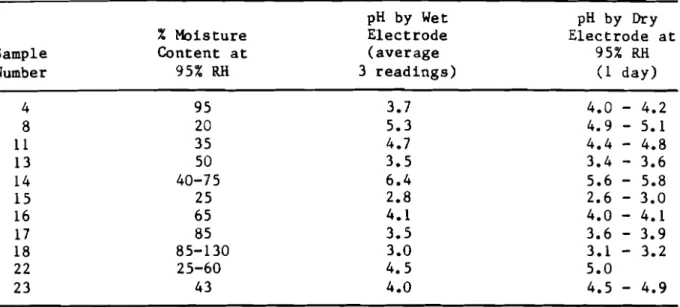
Some s a m p l e s t e s t e d had v e r y low pH v a l u e s ( a b o u t 2) by w e t e l e c t r o d e o r by d r y e l e c t r o d e a f t e r a v e r y s h o r t e x p o s u r e t o h i g h h u m i d i t y , b u t t h e y i n c r e a s e d t o v a l u e s above 3 o r 4 when a l l o w e d t o i n c r e a s e i n m o i s t u r e from e x p o s u r e t o h i g h h u m i d i t y f o r 14 days. T h i s c a n b e n o t e d w i t h samples 4,
15, 17 and 1 8 , T a b l e 1. O t h e r s a m p l e s d i d n o t r e s p o n d i n t h e same way a s c a n be s e e n i n T a b l e 1.
There h a v e been c a s e s where t h e w e t e l e c t r o d e method gave nuch lower pH v a l u e s t h a n t h e d r y e l e c t r o d e a s i n T a b l e 2, sample CAN-INT and CAN-EXT. The t h i r d sample, RAPCO, showed good agreement between v a l u e s o b t a i n e d by b o t h methods. These o b s e r v a t i o n s would l e a d t o t h e c o n c l u s i o n t h a t t h e c o n d i t i o n i n g r e q u i r e d t o o b t a i n t h e l o w e s t pH may w e l l b e d i f f e r e n t f o r e a c h sample. T h i s may n o t be s u r p r i s i n g when one c o n s i d e r s t h e g r e a t d i f f e r e n c e s i n t h e amount of w a t e r a d s o r b e d by samples a t 95%
RH,
T a b l e 1.REPRODUCIBILITY OF RESULTS
It was assumed t h a t t h e pH of a s u r f a c e of UFFI o v e r a n a r e a of a b o u t 25 c m 2 would b e uniform. To e s t a b l i s h t h e r e p r o d u c i b i l i t y o f t h e pH
measurement, t e n r e a d i n g s on t h r e e d i f f e r e n t s a m p l e s were t a k e n f i r s t by t h e w e t e l e c t r o d e method, t h e n r e p e a t e d o n t h e same samples w i t h a d r y e l e c t r o d e a s t h e s a m p l e s were exposed t o 95%
RH
f o r o n e , two, t h r e e , e i g h t and t e n days. T a b l e 2 shows t h e r e s u l t s . The measurements were made i n l a b o r a t o r y c o n d i t i o n s which were 48 t o 58%RH
and 25 t o 30°C. The t i m e r e q u i r e d t o g e t a s t a b l e pH v a l u e r a n g e d f r o m a b o u t 5 t o 15 s e c o n d s . It w i l l b e n o t e d t h a t t h e s t a n d a r d d e v i a t i o n v a r i e d from a h i g h of 0.47 f o r sample CAN-EXT by w e t e l e c t r o d e t o a low of 0.07 f o r t h e RAPCO sample a f t e r 10 d a y s c o n d i t i o n i n g .It may be t h a t t h e a s s u m p t i o n t h a t t h e sample was u n i f o r m i s n o t v a l i d , i n which c a s e t h e o b s e r v e d v a r i a t i o n i n measurement i s d u e t o b o t h t h e
v a r i a b i l i t y i n t h e sample and i n t h e measurement.
VARIATION I N pH OF FOAM IN-SITU I N THE WALL
A series of w a l l e t t e s m e a s u r i n g 8 ' x 8 ' were foamed i n 1977 w i t h UFFI by t h r e e s u p p l i e r s . I n 1982 t h e s e were examined by removing t h e " i n t e r i o r " gypsum board and t h e pH was measured u s i n g t h e w e t e l e c t r o d e method a t t h e t o p , m i d d l e and bottom l o c a t i o n s of t h e w a l l s p a c e . I n e a c h l o c a t i o n t h r e e measurements were made, o n e i n t h e m i d d l e and o n e a t e a c h edge. The r e s u l t s shown i n T a b l e 3 i n d i c a t e t h a t o n l y t h e Borden foam had s i g n i f i c a n t
v a r i a t i o n i n pH between d i f f e r e n t l o c a t i o n s of t h e foam i n t h e w a l l s p a c e . It would a p p e a r t h a t pH measurement, b e i n g a q u i c k and s i m p l e t e s t , would b e u s e f u l a s a n i n d e x t o s u r v e y t h e v a r i a b i l i t y i n c h a r a c t e r i s t i c s of t h e foam i n a g i v e n house.
pH
MEASUREMENT OF LEACH SOLUTIONThe pH of l e a c h s o l u t i o n s of UFFI was compared t o t h e s u r f a c e pH (wet e l e c t r o d e ) t o d e t e r m i n e i f t h e two methods gave s i m l l a r r e s u l t s . T h i s would a l l o w pH measurements o b t a i n e d by e i t h e r method t o b e compared.
A number of UFFI s a m p l e s f o r which s u r f a c e pH measurements had b e e n completed were powdered by g r i n d i n g them w i t h a m o r t a r and p e s t l e f o r 60 s e c o n d s . A mass of 0.10 g of e a c h UFFI powder was t h e n weighed i n t o 25-L s c r e w t o p g l a s s v i a l s . Ten
mL
of d i s t i l l e d , d e i o n i z e d water were added t o t h e v i a l s which were t h e n t i g h t l y c l o s e d a n d s h a k e n v i g o r o u s l y byhand f o r a few s e c o n d s t o d i s p e r s e t h e powder. The v i a l was t h e n p l a c e d i n a r o t a t i n g r a c k which t u r n e d i t end-over-end 20 times p e r m i n u t e f o r
24 h o u r s . The pH o f t h e s l u r r y was t h e n measured u s i n g t h e same a p p a r a t u s a s was used f o r s u r f a c e pH measurements.
R e s u l t s of s u r f a c e a n d l e a c h pH measurements f o r a v a r i e t y o f s a m p l e s a r e t a b u l a t e d i n T a b l e 4. The two sets of v a l u e s a r e g e n e r a l l y i n c l o s e agreement ( a d i f f e r e n c e of l e s s t h a n 0.5 pH u n i t s ) f o r e a c h foam, a n d show t h e same t r e n d s . An e x c e p t i o n i s t h e B r i t i s h foam, which had a d i f f e r e n c e
of more t h a n 4 pH u n i t s . T h i s may be b e c a u s e B r i t i s h foam had b e e n t r e a t e d w i t h ammonia. I n g e n e r a l , t h e w e t e l e c t r o d e s u r f a c e pH measurements and w a t e r l e a c h a t e pH's a r e comparable.
CONCLUSIONS
It h a s been d e m o n s t r a t e d t h a t t h e s u r f a c e pH of UFFI s a m p l e s i s e a s y t o measure, and a p p e a r s t o i n d i c a t e t h e amount of f r e e a c i d i n t h e foam.
The pH v a l u e d e p e n d s n o t o n l y o n t h e p a r t i c u l a r s a m p l e b u t a l s o o n i t s c o n d i t i o n i n g when a d r y e l e c t r o d e i s used. Values f o r some foams may change s i g n i f i c a n t l y a s t h e t i m e f o r c o n d i t i o n i n g a t h i g h h u m i d i t y i s i n c r e a s e d ( d i l u t i o n of s u r f a c e a c i d ) . T h i s e f f e c t is small i n t h e c a s e of o t h e r foams.
The s i m p l e s t method of t e s t , t h a t i n v o l v i n g t h e w e t e l e c t r o d e , a p p e a r s t o g i v e t h e l o w e s t pH v a l u e s and i s p e r h a p s t h e most r e l i a b l e . T h i s
measurement i s comparable t o pH a s measured by l e a c h i n g t h e W F I w i t h w a t e r and measuring t h e pH of t h e l e a c h a t e .
T a b l e 1
-
pH o f Shredded UFFI Packed i n P o l y e t h y l e n e B o t t l e s pH by Wet PH by Dry % M o i s t u r e E l e c t r o d e E l e c t r o d e a t Sample C o n t e n t a t ( a v e r a g e 95%RH
Number 9 5 %RH
3 r e a d i n g s ) ( 1 d a y ) 4 9 5 3.7 4.0-
4.2 8 2 0 5.3 4.9-
5.1 11 35 4.7 4.4-
4.8 1 3 5 0 3.5 3.4-
3.6 1 4 40-7 5 6.4 5.6-
5.8 1 5 2 5 2.8 2.6-
3.0 16 65 4.1 4.0-
4.1 17 8 5 3.5 3.6-
3.9 1 8 85-130 3.0 3.1-
3.2 22 25-60 4.5 5.0 2 3 43 4.0 4.5-
4.9Table 2
-
T e s t s t o Determine R e p r o d u c i b i l i t y o f pH Measurements Dry e l e c t r o d e-
sample exposed t o 95% RH c o n d i t i o n Wet Sample E l e c t r o d e 1 day 2 d a y s 3 d a y s 8 d a y s 10 d a y s pH S.D. pH S.D. pH S.D. pH S.D. pH S.D. pH SOD. CAN-INT 3.94 0.11 3.91 0.19 3.69 0.26 3.74 0.22 3.97 0.33 CAN-EXT
2.73 0.17 2.35 0.22 2.47 0.37 2.31 0.24 2.48 0.35 RAPCO 3 . 7 1 0 . 1 2 3 . 8 6 0 . 1 0 4 . 0 4 0 . 3 9 4 . 0 1 0 . 1 8 3 . 9 2 0 . 1 0 3 . 9 9 0 . 0 7Table 3
-
Survey o f t h e V a r i a b i l i t y of pH o f W F I i n The WallWall Space No. 1 No. 2 No. 3
Canada Foam TOP Middle Bottom Rapco Foam TOP Middle Bottom Borden Foam TOP Middle Bottom
T a b l e 4
-
Comparison of S u r f a c e and Leach pH f o r UFFK Samples UFF I S u r f a c e pH (wet e l e c t r o d e ) Leach pH - -Borden A BordenB
Rapco A RapcoB
Rapcoc
Canada A Canada B Day b l Day i / 2 Day 1 3 Day /I4 /I 2 11 3 1 4 23 Bishop Lavoie B r i t i s h Dee j a r d i n s Korwin 10 Barkerow
S t e e n b a k k e r Vandal S t e . Marie Code t o Sample I d e n t i f i c a t i o n-
T a b l e 1Sample No. Name of Family
Van Dudeir T a y l o r B a s t i e n P e l a e z Levesque Savard Nan R a a l t W e i n s t e i n Korwin Dupont Artacho