Publisher’s version / Version de l'éditeur:
Proceedings of the 238th ACS Fall National Meeting and Exposition, 2009,
2009-08-16
READ THESE TERMS AND CONDITIONS CAREFULLY BEFORE USING THIS WEBSITE. https://nrc-publications.canada.ca/eng/copyright
Vous avez des questions? Nous pouvons vous aider. Pour communiquer directement avec un auteur, consultez la
première page de la revue dans laquelle son article a été publié afin de trouver ses coordonnées. Si vous n’arrivez pas à les repérer, communiquez avec nous à PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca.
Questions? Contact the NRC Publications Archive team at
PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca. If you wish to email the authors directly, please see the first page of the publication for their contact information.
NRC Publications Archive
Archives des publications du CNRC
This publication could be one of several versions: author’s original, accepted manuscript or the publisher’s version. / La version de cette publication peut être l’une des suivantes : la version prépublication de l’auteur, la version acceptée du manuscrit ou la version de l’éditeur.
Access and use of this website and the material on it are subject to the Terms and Conditions set forth at
Study of Clay Dispersion and Particle Size on Properties of Composite
Polymer Electrolytes
Siu, Ana; Mokrini, Asmae; Robitaille, Lucie
https://publications-cnrc.canada.ca/fra/droits
L’accès à ce site Web et l’utilisation de son contenu sont assujettis aux conditions présentées dans le site LISEZ CES CONDITIONS ATTENTIVEMENT AVANT D’UTILISER CE SITE WEB.
NRC Publications Record / Notice d'Archives des publications de CNRC:
https://nrc-publications.canada.ca/eng/view/object/?id=5a1e1bd2-4bad-46a6-8ff0-f67d2fd79ae3
https://publications-cnrc.canada.ca/fra/voir/objet/?id=5a1e1bd2-4bad-46a6-8ff0-f67d2fd79ae3
Prepr. Pap.-Am. Chem. Soc., Div. Fuel Chem. 2009, 54 (2), xxxx
Study of Clay Dispersion and Particle Size on Properties of Composite Polymer Electrolytes
Ana Siu, Asmae Mokrini and Lucie Robitaille
National Research Council – Industrial Materials Institute, 75 de Mortagne Blvd., Boucherville, Quebec, Canada J4B 6Y4. Introduction
The study of clay-reinforced polymer composites has been one of the most rapidly growing areas in polymer and materials science, since its discovery in the mid 1980s1. This rapid growth is primarily attributed to the significant changes it brings to a material when only a few weight percent of the clay filler is added. These changes include better flame retardancy, permeability, mechanical strength and modulus, dimensional, thermal and chemical stability, electrical conductivity and optical clarity in comparison to conventional fiber-filled polymer matrices1, 2.
For this work, we used clay polymer composites to mitigate challenges, such as material handling, physical stability and barrier capacity of thin (< 30 – 40 microns) solid electrolytes for fuel cell applications. It has already been demonstrated in the literature that the ability of membranes to reject methanol had improved by as much as 30% when clay was incorporated into Nafion® membranes. This property subsequently led to a slight improvement in the performance of direct methanol fuel cells3. Another study also noted an improvement in the thermooxidative stability of polybenzimidazole (PBI) membranes when organically-modified montmorillonite was incorporated4. In addition to the 30% decrease of the composite’s coefficient of thermal expansion in comparison to pure PBI, the mechanical and methanol barrier properties also improved. Although the benefits of clays have been well described, selecting the appropriate clay type has been commonly a trial and error process. In this paper, a series of clays with different aspect ratios will be incorporated into Nafion® matrix. The effect of their dispersion and
sizes on polymer electrolytes properties related to fuel cell applications will be presented.
Experimental
Composite Fabrication. Clays with different aspect ratios (Table 1) (Sumecton-SA from Kunimie, Cloisite-Na+ from Southern Clays, Somasif Me-100 from Unicoop and Na-Ts-S from Topy) (0.030g) were added to a vial containing 5 mL Millipore water and stirred at room temperature for at least 2 hours. The mixture was sonicated for 8 hours at 60 oC prior to adding to Nafion® solution (6.12 g, 5 wt % resin from Aldrich) to make a 10 wt % composite film. The entire clay / polymer solution was sonicated for another 20 min., to ensure adequate mixing then another 10 min. after adding 15 mL dimethylacetamide. The content was emptied onto a glass casting plate and inserted into an oven. The solvent was evaporated from the mixture by heating it at 50 oC overnight. The film was heated to 100 oC for 2 hours and then at 130 oC for another 2 hours. They were released from the glass substrate by immersion in water.
Composite Film Characterization. Transmission electron microscope (TEM) images, X-Ray Diffraction data (Bruker 40 kV, 40 mA, 1 mm slit, 2Ө = 3.2 – 40) and differential scanning calorimetry (DSC) data (Q200 TA Instrument, nitrogen atmosphere, -80 – 250 o
C at 10 oC min-1) were collected from dried non-acidified films.
Proton conductivity, water uptake, dimensional swelling and mechanical tests were measured for acidified membranes. The films were converted to acidic form by equilibration in 2 M H2SO4
overnight. Excess acid was removed by soaking the protonated films
in Millipore water overnight. Water uptake and dimensional swelling were calculated using the equations shown below:
Dog-bone shaped samples were cut and dried at room temperature in a vacuum oven prior to testing in an Instron 1123 mounted with an extension meter. The samples were stretched at a rate of 2 mm min-1; Young modulus and yield strength from the samples were obtained from respective stress-strain curves.
In-plane proton conductivities were measured using a Solartron 1260. A strip of membrane (approximately 0.5 x 2 cm) was set between two platinum (Pt) electrodes and an alternating current was passed through the plane of the sample. The samples were placed in an ESPEC-SH261 environmental chamber for humidity and temperature control. In the case of room temperature and liquid water conditions, the samples were immersed in Millipore water. The temperature in the room was 23 2 oC. Nyquist plots between 5
MHz to 10 Hz were collected and membrane resistances were extrapolated by fitting the semi-circle part of the data to Randles equivalent circuit. Proton conductivities were calculated using the following equation:
where is proton conductivity, L is the distance between the Pt electrodes, R is membrane resistance and A is the cross-sectional area of the strip. The samples were allowed to equilibrate in the chamber with monitoring. The strip normally took 30min to 1 hour to equilibrate.
Results and Discussion
Composite Properties. Sumecton and Cloisite composite films appear transparent but those of Somasif-ME-100 and Na-Ts-S appear slightly opaque. TEM images of all four composites are illustrated in Figure 1. Clays appear as dark lines in the grey polymer matrix. Good clay dispersion is observed for Sumecton and Cloisite composite films. Lots of exfoliated sheets and only a few short stacks of clay agglomerates were visible from them. XRD data in Table 1 show an absence of the clay peak for Sumecton composite, but a broad peak centered at 6.38 (2Ө) is observed for Cloisite composite. The downward shift of 2Ө is indicative of increasing clay basal spacing and therefore an expansion of the clay galleries. These results are consistent with the clarity of the film.
However, clay dispersion appears to be poor for composites containing higher clay aspect ratios (i.e. Somasif-ME-100 and Na-Ts-S). TEM images show some exfoliation, but much of the clays appear as bundle of fibers in these composite systems. The downward shift in 2Ө values in the XRD data, however, allude to some polymer intercalation within the clay galleries. The poorer clay dispersion could simply be a consequence of: (1) the nature of the clay, or (2) the increase likelihood of neighbor contact.
Glass transition temperature of the composites appears to be indifferent to the different clay aspect ratios as they all exhibit similar Tg value as dry Nafion® ~ 160 oC5 (see Table 1). Exceptions are
observed for re-cast Nafion® and Cloisite composite where no Tg
could be detected. The density of the composite appears to exhibit a downward trend with increasing aspect ratio which may infer to lower crystalline content or more void space within the polymer.
Prepr. Pap.-Am. Chem. Soc., Div. Fuel Chem. 2009, 54 (2), xxxx
Table 1. Properties of Nanocomposites
Sample Clay Aspect Ratio* Appearance Neat Clay Basal Peak (2Ө) Clay Peak in Composite (2Ө) Tg (o C) Density of Composite (g cm-3 )**
Recast-Nafion None Clear film none none none 1.90
Nafion +
Sumecton 50 Clear film 6.56 none 153.6 1.80
Nafion + Cloisite
280 Clear,
yellowish film 7.56 6.38 none 1.70
Nafion + Na-Ts-S 5000 White opaque film 9.1 6.78 151.4 1.55 Nafion + Somasif ME 100 6000 Slightly opaque film 9.1 6.42 151.5 1.26 * From reference6.
** Density of membrane after drying in vacuum oven at 80 oC overnight.
Sumecton Cloisite
Somasif Me-100 Na-Ts-S
Figure 1. TEM images of clay composites.
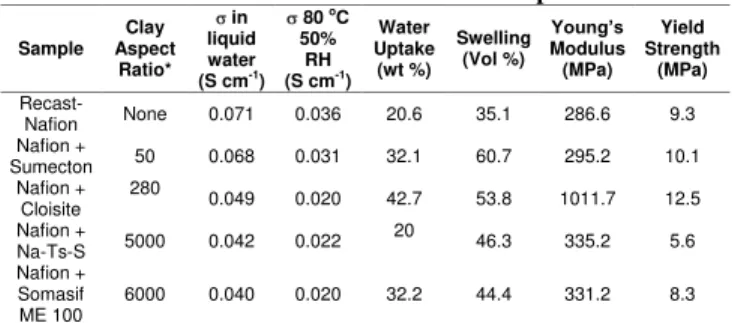
Composite Film Characterization. The increase in the clay’s aspect ratio appears to have a negative impact on the composites’ proton conductivity (Table 2), both, in the wet state at room temperature and at reduced humidity at 80 oC. The values drop by approximately 30 and 40 % from the neat film, respectively, and stabilize at about an aspect ratio of 280. Water uptake and membrane swelling also increase with inclusion of clays with higher aspect ratio. The increase reaches a maximum at around an aspect ratio of 280. A similar bell shaped trend is observed in the material’s Young’s modulus and yield strength. It is not immediately clear whether the lack of complete clay exfoliation in systems with high clay aspect ratio (e.g. Somasif Me-100 and Na-Ts-S) induce a plateau effect in the conductivity and a drop in mechanical strength and swelling. Further experiments will be needed in order to verify whether more exfoliation (perhaps a lower loading for clays with higher aspect ratio) gives the same results.
It is however clear from well exfoliated Sumecton and Cloisite systems that proton conductivity decreases with increasing aspect ratio, whereas, water uptake, membrane swelling and mechanical strength all increase. This behaviour is expected for mechanical strength tests because high aspect ratio clays increase the contact
between the filler and the matrix and hence it has a reinforcing effect on the strength7. Having said that one would also expect that
swelling and water uptake behavior should follow the same trend – that is decrease with increasing aspect ratio. The opposite behavior suggests that perhaps the increased surface area also increases the number of exposed ionic sites in the clay thereby attracting more water. Perhaps one way to mitigate this issue is to modify clay surfaces in order to improve the interaction between filler and matrix interaction and consequently leading to a reduction in swelling from the composites.
Table 2. Characterization Results of Composites
Sample Clay Aspect Ratio* in liquid water (S cm-1) 80 oC 50% RH (S cm-1) Water Uptake (wt %) Swelling (Vol %) Young’s Modulus (MPa) Yield Strength (MPa) Recast-Nafion None 0.071 0.036 20.6 35.1 286.6 9.3 Nafion + Sumecton 50 0.068 0.031 32.1 60.7 295.2 10.1 Nafion + Cloisite 280 0.049 0.020 42.7 53.8 1011.7 12.5 Nafion + Na-Ts-S 5000 0.042 0.022 20 46.3 335.2 5.6 Nafion + Somasif ME 100 6000 0.040 0.020 32.2 44.4 331.2 8.3 * From reference6. Conclusions
Clays of different aspect ratios have successfully been incorporated into re-cast Nafion®. Dispersion and exfoliation of Sumecton and Cloisite clays appear to be good. Somasif Me-100 and Na-Ts-S systems show some exfoliation mostly as bundled clay platelets with some intercalation as indicated by increased distance between clay galleries. Increasing the aspect ratio of clay platelets in the composite appears to lower their ability of the composite to transport protons but increases their ability to swell and absorb water in well exfoliated systems. They also become less elastic but attain higher yield strength and therefore should resist more to deformation under compression (i.e. under fuel cell operation).
Acknowledgement. The authors would like to thank Nicole Coté for DSC, Florence Perrin for TEM, Karine Theberge for XRD, Martin Bureau and Manon Plourde for mechanical tests.
References
(1) Okada, A.; Usuki, A. Macromolecular Materials and Engineering 2006, 291, 1449-1476.
(2) Gao, F. Materials Today 2004, 7, (11), 50-55.
(3) Lee, W.; Kim, H.; Kim, T. K.; Chang, H., Journal of Membrane Science
2007, 292, (1/2), 29-34.
(4) Chuang, S. W.; Hsu, S. L.-C.; Hsu, C. L. Journal of Power Sources 2007, 168, (1), 172-177.
(5) Cho, K. Y.; Jung, H. Y.; Sung, K. A.; Kim, W. K.; Sung, S. J.; Park, J. K.; Choi, J. H.; Sung, Y. E. Journal of Power Sources 2006, 159, , 524-528. (6) Utracki, L. A.; Sepehr, M.; Boccaleri, E. Polymers for Advanced
Technologies 2007, 18, (1), 1-37.
(7) Ray, S. S.; Okamoto, M. Progress in Polymer Science 2003, 28, (11), 1539-1641.