1 Table of Contents Figures Pages Experimental 2 Synthesis 2 Crystallography 8 References 8 Table S1 9 APPENDIX 10
I) NMR spectra of starting azo-dye 1 S1-S6 10-12
II) NMR spectra of 1-OMe S7-S12 13-15
III) NMR spectra of 1-NMe S13-S18 16-18
IV) Comparison between 1H and 13C NMR spectra of compounds 1,
1-OMe and 1-NMe
S19,S20 19
V) NMR spectra of compound 2 S21-S26 20-22
VI) NMR spectra of compound 3 S27-S32 23-25
VII) NMR spectra of compound 4 S33,S34 26
VIII) NMR spectra of compound 5a S35-S56 27-37
IX) NMR spectra of compound 5b S57 38
X) NMR spectra of compound 5c S58,S59 38,39
XI) NMR spectra of compound 5d S60 39
XII) NMR spectra of compound 5e S61-S64 40,41
XIII) NMR spectra of compound 7 S65-S69 42-44
XIV) NMR spectra of compound 6 S70-S74 44-46
2 Experimental
Synthesis
All reagents were purchased from Aldrich, Merck and Fluka and were used without any further purification. The deuterated solvents were purchased from Deutero GmbH. Fluka silica gel (TLC-cards 60778 with fluorescent indicator 254 nm) were used for TLC chromatography and Rf-values determination. Merck Silica gel 60 (0.040-0.063 mm) was used for flash
chromatography purification of the products. The melting points were determined in capillary tubes on SRS MPA100 OptiMelt (Sunnyvale, CA, USA) automated melting point system. UV spectra were measured on Varian Cary 100 spectrophotometer in acetone. The NMR spectra were recorded on a Bruker Avance II+ 600 spectrometer (Rheinstetten, Germany), 1H at 600 MHz and 13C at 151 MHz, in CDCl3 or DMSO-d6; the chemical shifts were quoted in ppm in
δ-values against tetramethylsilane (TMS) as an internal standard and the coupling constants were calculated in Hz. The spectra were processed with Topspin 2.1 program. For simplicity, naphthyl ring nuclei are depicted as Ar, azo-dye pyridyl unit as Py, NH-connected pyridyl moiety as Py’, aryl or heteroaryl substituent at methyne bridge as Ar’, and methylene or methyne bridge as CH2 and CH, respectively. In some cases the spectra of slightly impure
samples were recorded and analysed due to the much lower solubility of the pure products. The compounds purities after final recrystallization were proved by TLC and melting points determination. The turbo spray mass spectra were taken on API 150EX (AB/MAS Sciex).
Starting azo-dye 1
To 3-amino pyridine (10 mmol) ice, conc. HCl (3 ml), and NaNO2 (11 mmol) were
subsequently added and the mixture was stirred at 0oC for 5 min to form a diazonium salt. To a solution of naphthalene-1-ol (10 mmol) in 10 % aq. NaOH (10 ml) the solution of the diazonium salt was added at 0oC. The residue formed was filtered off, washed with water and dried in air to give 4-(3-pyridyldiazenyl)naphthalen-1-ol 1, which was further used without purification. A sample for analyses was purified by flash chromatography on silica gel by using mobile phase with a gradient of polarity from CH2Cl2 to 2 % MeOH in CH2Cl2 (2 %
MeOH/CH2Cl2): m. p. 209.5-210 °C; Rf 0.38 (5 % MeOH/DCM); UV λmax 415 nm; 1H NMR
(DMSO-d6) 7.046 (d, 1H, J 8.4, CH-2 Ar), 7.613 (m, 2H, CH-5 Py and CH-7 Ar), 7.721 (ddd,
1H, J 8.4, 6.8, 1.3, CH-6 Ar), 7.935 (d, 1H, J 8.4, CH-3 Ar), 8.248 (d, 1H, J 8.3, CH-8 Ar), 8.292 (ddd, 1H, J 8.2, 1.9, 1.1, CH-6 Py), 8.699 (dd, 1H, J 4.3, 1.1, CH-4 Py), 8.920 (d, 1H, J 8.4, CH-5 Ar), 9.162 (d, 1H, J 1.9, CH-2 Py), 11.339 (s, 1H, OH); 13C NMR 108.93 (CH-2 Ar), 114.87 (CH-3 Ar), 122.90 (CH-8 Ar), 123.07 (CH-5 Ar), 124.78 (Cquat-8a Ar), 125.05 (CH-5
3 Py), 126.08 (CH-7 Ar), 127.25 (CH-6 Py), 128.56 (CH-6 Ar), 133.27 (Cquat-4a Ar), 140.13
(Cquat-4 Ar), 146.70 (CH-2 Py), 148.60 (Cquat-1 Py), 151.48 (CH-4 Py), 158.99 (Cquat-1 Ar).
Methylation of 1
To a solution of crude 1 (1 mmol) in THF (5 ml) KOH (2 mmol) and after 15 min CH3I (5
mmol) was added and the suspension was stirred at room temperature for 2 h. The solid phase was filtered off and washed with THF. The solvent was removed in vacuo and the residue formed was purified by flash chromatography on silica gel by using mobile phase with a gradient of polarity from CH2Cl2 to 15 % acetone/CH2Cl2 to give: compound 1-OMe: Rf 0.70
(5 % MeOH/DCM); 15 % yield; m. p. 126.5-127 °C; UV λmax 401 nm; 1H NMR (CDCl3) 4.098
(s, 3H, CH3), 6.921 (d, 1H, J 8.5, CH-2 Ar), 7.484 (dd, 1H, J 8.1, 4.8, CH-5 Py), 7.593 (ddd,
1H, J 8.4, 6.8, 1.4, CH-7 Ar), 7.694 (ddd, 1H, J 8.3, 6.8, 1.4, CH-6 Ar), 7.976 (d, 1H, J 8.5, CH-3), 8.259 (ddd, 1H, J 8.2, 2.2, 1.6, CH-6 Py), 8.329 (ddd, 1H, J 8.3, 1.4, 0.7, CH-8 Ar), 8.689 (d, 1H, J 4.2, CH-4 Py), 8.943 (ddd, 1H, J 8.4, 1.4, 0.7, CH-5 Ar), 9.258 (bs, 1H, CH-2 Py); 13C NMR 55.98 (CH3), 103.73 (CH-2 Ar), 113.59 (CH-3 Ar), 122.24 (CH-8 Ar), 122.96
(CH-5 Ar), 124.12 (CH-5 Py), 125.59 (Cquat-8a Ar), 126.03 (CH-7 Ar), 127.38 (CH-6 Py),
127.90 (CH-6 Ar), 132.79 (Cquat-4a Ar), 141.67 (Cquat-4 Ar), 146.86 (CH-2 Py), 148.65 (Cquat-1
Py), 150.56 (CH-4 Py), 159.25 (Cquat-1 Ar); ESI (TIS)-Q m/z 264.16 [M+1]+, C16H13N3O; and
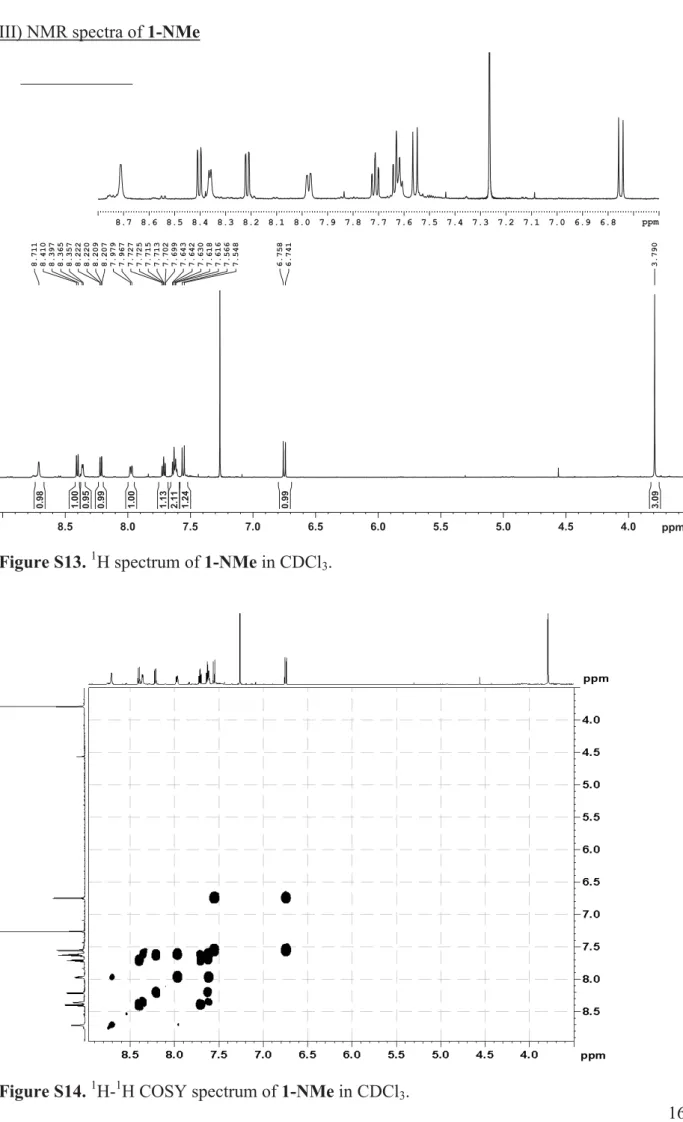
compound 1-NMe: Rf 0.51 (5 % MeOH/DCM); 4 % yield; UV λmax 443 nm; 1H NMR
(CDCl3) 3.790 (s, 3H, CH3), 6.749 (d, 1H, J 10.4, CH-2 Ar), 7.557 (d, 1H, J 10.4, CH-3 Ar),
7.615 (overlapped signals, CH-5 Py), 7.630 (ddd, 1H, J 7.9, 7.3, 0.8, CH-7 Ar), 7.714 (ddd, 1H, J 8.0, 7.3, 1.1, CH-6 Ar), 7.973 (dd, 1H, J 8.4, 1.3, CH-6 Py), 8.215 (dd, 1H, J 7.9, 1.1, CH-8 Ar), 8.362 (d, 1H, J 4.8, CH-4 Py), 8.404 (dd, 1H, J 8.0, 0.8, CH-5 Ar), 8.711 (bs, 1H, CH-2 Py); 13C NMR 45.62 (CH3), 124.21 (CH-5 Ar), 125.28 (CH -5 Py), 126.40 (CH-8 Ar),
127.41 (CH-6 Py), 128.67 (CH-3 Ar), 130.09 (CH-7 Ar), 130.82 (Cquat-8a Ar), 131.05 (CH-2
Ar), 132.46 (CH-2 Py), 132.89 (CH-6 Ar), 133.76 (Cquat-4a Ar), 137.61 (CH-4 Py), 144.89
(Cquat-4 Ar), 146.91 (Cquat-1 Py), 184.54 (Cquat-1 Ar); ESI (TIS)-Q m/z 264.22 [M+1]+,
C16H13N3O.
Compounds with methylene bridged side chain (Mannich reaction)
General procedure: To a solution of piperidine or 3-aminopyridine (1.5 mmol) in benzene (5-6
ml) paraformaldehyde (1.5 mmol), p-toluenesulfonic acid (10 mg), and then crude 1 (1 mmol) were added and the mixture was refluxed with stirring for 3 h. The products were purified by flash chromatography on silica gel.
Compound 2: 70 % overall yield; Rf 0.33 (5 % MeOH/DCM);m. p. 114-115 °C; UV λmax 422
4 CH2-2 and CH2-6 piperidine), 3.063 (bs, 2H, ½ of CH2-2 and CH2-6 piperidine), 3.910 (s, 2H,
CH2), 7.434 (dd, 1H, J 8.1, 4.6, CH-5 Py), 7.550 (ddd, 1H, J 8.3, 6.8, 1.0, CH-7 Ar), 7.640
(ddd, 1H, J 8.4, 6.8, 1.2, CH-6 Ar), 7.762 (s, 1H, CH-3 Ar), 8.207 (ddd, 1H, J 8.1, 2.0, 1.6, CH-6 Py), 8.309 (d, 1H, J 8.3, CH-8 Ar), 8.646 (dd, 1H, J 4.6, 1.3, CH-4 Py), 8.911 (d, 1H, J 8.4, CH-5 Ar), 9.220 (d, 1H, J 2.0, CH-2 Py), 10.737 (bs, 1H, OH); 13C NMR 23.79 (CH2-4
piperidine), 25.72 (CH2-3 and CH2-5 piperidine), 53.88 (CH2-2 and CH2-6 piperidine), 62.01
(CH2), 113.88 (CH-3 Ar), 114.06 (Cq-2 Ar), 122.44 (CH-8 Ar), 122.71 (CH-5 Ar), 123.99
(CH-5 Py), 125.05 (Cquat-8a Ar), 125.51 (CH-7 Ar), 126.67 (CH-6 Py), 127.65 (CH-6 Ar),
132.79 (Cquat-4a Ar), 139.76 (Cquat-4 Ar), 147.15 (CH-2 Py), 148.66 (Cquat-1 Py), 150.51 (CH-4
Py), 160.08 (Cquat-1 Ar); ESI (TIS)-Q m/z 347.31 [M+1]+ C21H22N4O.
Compound 3: 36 % overall yield; Rf 0.36 (5 % MeOH/DCM + 0.5% NH4OH); m. p.
161-162 °C (at 140.5-141 °C became very dark); UV λmax 451 nm; The product is not enough
soluble in DMSO-d6 to record proper 13C spectrum at reasonable time-scale; a part of the
signals were extracted from HSQC and HMBC experiments, another are missing. The extra pyridyl unit is depicted as Py”; 1H NMR (DMSO-d6) 3.40 (bs, NH-Cq-1 Py’, overlapped with
H2O in DMSO-d6), 4.259 (bs, 2H, CH2), 5.092 (s, 2H, N-CH2-NH), 6.271 (bs, 1H, NH-Cq-1
Py”), 6.949 (dd, 1H, J 8.1, 1.3, CH-6 Py’), 7.084 (dd, 1H, J 8.1, 4.5, CH-5 Py’), 7.143 (dd, 1H, J 8.3, 4.5, CH-5 Py”), 7.415 (bs, 1H, CH-5 Py), 7.481 (dd, 1H, J 8.3, 1.6, CH-6 Py”), 7.560 (t, 1H, J 7.4, CH-7 Ar), 7.715 (t, 1H, J 7.4, CH-6 Ar), 7.787 (d, 1H, J 4.0, CH-4 Py’), 7.918 (bs, 1H, CH-4 Py), 7.978 (d, 1H, J 4.3, CH-4 Py”), 8.057 (d, 1H, J 1.9, CH-2 Py’), 8.112 (bs, 1H, CH-8 Ar), 8.216 (bs, 1H, CH-3 Ar), 8.271 (bs, 1H, CH-6 Py), 8.422 (d, 1H, J 2.3, CH-2 Py”), 8.496 (bs, 1H, CH-5 Ar), 8.772 (bs, 1H, CH-2 Py); 13C NMR 42.36 (CH2-NH-Cq-1 Py’), 66.34
(CH2- NH-Cq-1 Py”), 117.98 (CH-6 Py’), 122.90 (CH-5 Ar), 123.46 (CH-5 Py”), 123.79 (CH-5
Py’), 123.87 (CH-6 Py”), 124.31 (CH-5 Py), 125.64 (CH-8 Ar), 127.75 (CH-7 Ar), 129.63 (Cquat-8a Ar), 131.97 (CH-6 Ar), 135.40 (Cquat-4a Ar), 135.70 (CH-2 Py’), 136.24 (CH-4 Py),
137.28 (CH-4 Py’), 139.33 (CH-2 Py”), 141.14 (CH-4 Py”), 144.13 (Cquat-1 Py”), 144.83
(Cquat-1 Py’), 144.97 (Cquat-1 Py); ESI (TIS)-Q m/z 462.29 [M+1]+, C27H23N7O.
Compound 4: A solution of 3-aminopyridine (1.5 mmol) and paraformaldehyde (1.5 mmol) in
DMSO (1 ml) was stirred at 85 °C for 1.5 h. The mixture was cooled to room temperature, the crude 1 (1 mmol) was added, and the solution was kept at room temperature for 3 h. The reaction mixture was poured in water and the solid phase formed was filtered off. The product was purified by flash chromatography on silica gel to give 4: 56 % overall yield; Rf 0.17 (5 %
MeOH/DCM); m. p. 168-169 °C; UV λmax 450 nm; 1H NMR (DMSO-d6) 4.258 (s, 2H, CH2),
6.273 (bs, 1H, NH-CH2), 6.948 (ddd, 1H, J 8.3, 2.9, 1.4, CH-6 Py’), 7.083 (ddd, 1H, J 8.3, 4.6,
5 (ddd, 1H, J 8.3, 7.1, 1.4, CH-6 Ar), 7.788 (dd, 1H, J 4.6, 1.2, CH-4 Py’), 7.919 (bs, 1H, CH-4 Py), 8.059 (d, 1H, J 2.7, CH-2 Py’), 8.108 (bs, 1H, CH-8 Ar), 8.224 (bs, 1H, CH-3 Ar), 8.269 (bs, 1H, CH-6 Py), 8.496 (bs, 1H, CH-5 Ar), 8.779 (bs, 1H, CH-2 Py), 11.702 (bs, 1H, NH-N); ESI (TIS)-Q m/z 356.18 [M+1]+, C21H17N5O.
Compounds with aryl-methyne bridged side chain (Betti condensation)
General procedure: A solution of crude 1 (1 mmol), 3-aminopyridine (1.5 mmol) and aldehyde
(1.5 mmol) in DMSO (1 ml) was kept at room temperature for 4-6 days. The reaction mixture was poured in water and the solid phase formed was filtered off. The product was purified by flash chromatography on silica gel.
Compound 5a: Reaction time: 4 days; 54 % overall yield; Rf 0.28 (5 % MeOH/DCM);m. p.
176-177 °C; UV λmax 453 nm; Compound 5a is not enough soluble even in DMSO-d6 to record
proper 13C spectrum at reasonable time-scale; a part of the signals were extracted from HSQC and HMBC experiments, another are missing; 1H NMR (DMSO-d6, 343K) 5.957 (bs, 1H, CH),
6.300 (bs, 1H, NH-CH), 6.964 (ddd, 1H, J 8.3, 2.8, 1.4, CH-6 Py’), 7.052 (ddd, 1H, J 8.3, 4.6, 0.4, CH-5 Py’), 7.261 (tt, 1H, J 1.3, 7.3, CH-4 Ar’), 7.356 (ddt, 2H, J 7.7, 7.3, 1.5, CH-3 and CH-5 Ar’), 7.436 (dd, 1H, J 8.3, 4.7, CH-5 Py’), 7.538 (dt, 2H, J 7.7, 1.3, CH-2 and CH-6 Ar’), 7.559 (dd, 1H, J 8.3, 1.2, CH-7 Ar), 7.702 (ddd, 1H, J 8.3, 7.1, 1.4, CH-6 Ar), 7.810 (dd, 1H, J 4.6, 1.4, CH-4 Py’), 7.953 (dd, 1H, J 8.3, 2.1, CH-4 Py), 8.112 (d, 1H, J 2.8, CH-2 Py’), 8.141 (bd, 1H, J 8.1, CH-8 Ar), 8.321 (bs, 1H, CH-3 Ar), 8.351 (bd, 1H, J 4.5, CH-6 Py), 8.546 (bd, 1H, J 7.9, CH-5 Ar), 8.838 (d, 1H, J 1.9, CH-2 Py), 11.449 (bs, 1H, NH-N); 13C NMR 55.68 (CH), 118.63 (CH-6 Py’), 119.63 (CH-6 Py), 122.19 (CH-5 Ar), 122.41 (CH-2 Py), 123.08 (CH-5 Py’), 123.74 (CH-5 Py), 124.50 (CH-8 Ar), 126.86 (CH-4 Ar’), 126.99 (CH-7 Ar), 127.16 (CH-2 and CH-6 Ar’), 128.08 (CH-3 and CH-5 Ar’), 130.87 (CH-6 Ar), 134.07 (Cquat
-8a Ar), 135.03 (Cquat-4a Ar), 136.10 (CH-2 Py’), 136.68 (CH-4 Py), 137.54 (CH-4 Py’), 140.98
(Cquat-1 Ar’), 143.47 (Cquat-1 Py’), 146.93 (Cquat-1 Py). The signals for CH-3 Ar and CH-6 Py
changing their places at 302K: 8.302 (bm, 1H, CH-6 Py), 8.368 (bs, 1H, CH-3 Ar); CH-3 Ar shows NOESY cross peaks with CH-2 and CH-6 Ar’ and with CH-6 Py’; ESI (TIS)-Q m/z 432.23 [M+1]+, C27H21N5O.
Compound 5b: Reaction time: 4 days; 53 % overall yield; Rf 0.26 (5 % MeOH/DCM);m. p.
156-157 °C; UV λmax 455 nm; 1H NMR (DMSO-d6) 5.981 (bs, 1H, CH), 6.543 (bs, 1H,
NH-CH), 6.894 (bd, 1H, J 7.9, CH-6 Py’), 7.051 (dd, 1H, J 8.3, 4.6, CH-5 Py’), 7.233 (d, 1H, J 4.4, CH-4 Ar’), 7.440 (m, 1H, CH-5 Py), 7.466 (bs, 1H, CH-2 Ar’), 7.561 (dd, 1H, J 5.0, 2.9, CH-5 Ar’), 7.553 (t, 1H, J 7.6, CH-7 Ar), 7.704 (td, 1H, J 7.6, 1.0, CH-6 Ar), 7.778 (dd, 1H, J 4.4, 0.9, CH-4 Py’), 7.958 (bs, 1H, CH-4 Py), 8.069 (d, 1H, J 2.8, CH-2 Py’), 8.131 (bs, 1H, CH-8
6 Ar), 8.329 (bs, 1H, CH-3 Ar), 8.362 (bs, 1H, CH-6 Py), 8.541 (bs, 1H, CH-5 Ar), 8.817 (bs, 1H, CH-2 Py), 11.756 (bs, 1H, NH-N); ESI (TIS)-Q m/z 438.19 [M+1]+, C25H19N5OS.
Compound 5c: Reaction time: 4 days; 56 % overall yield; Rf 0.15 (5 % MeOH/DCM);m. p.
136-137 °C; UV λmax 459 nm; 1H NMR (DMSO-d6, 373K) 6.029 (bs, 1H, CH), 6.958 (bd, 1H,
J 8.2, CH-5 Py’), 7.025 (bd, 1H, J 8.2, CH-6 Py’), 7.345 (dd, 1H, J 7.7, 4.6, CH-5 Py or Ar’), 7.437 (dd, 1H, J 7.8, 4.6, CH-5 Py or Ar’), 7.549 (dd, 1H, J 8.1, 0.9, CH-7 Ar), 7.702 (dd, 1H, J 8.2, 1.2, CH-6 Ar), 7.805 (dt, 1H, J 7.9, 1.6, CH-4 Py or Py’), 7.884 (dt, 1H, J 7.9, 1.6, CH-4 Py or Py’), 7.950 (bd, 1H, J 7.6, CH-4 Ar’), 8.075 (bs, 1H, CH-2 Py’), 8.153 (bd, 1H, J 8.0, CH-8 Ar), 8.311 (bs, 1H, CH-6 Py), 8.371 (bs, 1H, CH-3 Ar), 8.468 (bs, 2H, J 1.3, 7.7, CH-6 Ar’), 8.554 (bd, 1H, J 8.2, CH-5 Ar), 8.766 (bs, 1H, CH-2 Py or Ar’), 8.850 (bs, 1H, CH-2 Py or Ar’); 13C NMR 54.88 (CH), 119.81 (CH-6 Py’), 120.72 (CH-5 Py’), 121.31 (CH-6 Py), 123.24 (CH-4 Ar’), 123.31 (CH-5 Ar), 124.08 (CH-5 Py or Ar’), 124.62 (CH-5 Py or Ar’), 125.28 (CH-2 Py’), 125.32 (CH-8 Ar), 127.76 (CH-7 Ar), 131.66 (CH-6 Ar), 134.65 (CH-4 Py or Py’), 135.46 (CH-4 Py or Py’), 148.84 (CH-6 Ar’), 149.51 (CH-2 Py or Ar’), 130.69 (Cquat
-8a Ar), 137.08 (Cquat-4a Ar), 138.79 (Cquat-1 Ar’), 141.28 (Cquat-1 Py’), 145.16 (Cquat-1 Py);
ESI (TIS)-Q m/z 433.22 [M+1]+, C26H20N6O.
Compound 5d: Reaction time: 4 days; 35 % overall yield; Rf 0.30 (5 % MeOH/DCM);m. p.
214-215 °C; UV λmax 455 nm; 1H NMR (DMSO-d6) 2.286 (s, 3H, CH3), 5.776 (bs, 1H, CH),
6.483 (bs, 1H, NH-CH), 6.087 (bd, 1H, J 7.7, CH-6 Py’), 7.056 (dd, 1H, J 8.3, 4.6, CH-5 Py’), 7.080 (d, 1H, J 7.5, CH-4 Ar’), 7.240 (t, 1H, J 7.6, CH-5 Ar’), 7.321 (bd, 1H, J 7.5, CH-6 Ar’), 7.344 (bs, 1H, CH-2 Ar’), 7.431 (bs, 1H, CH-5 Py), 7.546 (bt, 1H, J 7.2, CH-7 Ar), 7.706 (ddd, 1H, J 8.2, 7.2, 1.3, CH-6 Ar), 7.784 (dd, 1H, J 4.6, 0.9, CH-4 Py’), 7.937 (bs, 1H, CH-4 Py), 8.059 (d, 1H, J 2.8, CH-2 Py’), 8.083 (bs, 1H, CH-8 Ar), 8.282 (bs, 1H, CH-6 Py), 8.400 (bs, 1H, CH-3 Ar), 8.482 (bs, 1H, CH-5 Ar), 8.794 (bs, 1H, CH-2 Py), 11.819 (bs, 1H, NH-N); 13C NMR 21.18 (CH3), 56.50 (CH), 118.70 (CH), 119.97 (CH), 122.90 (CH), 123.69 (CH), 123.78
(Cquat), 124.35 (CH), 124.90 (CH), 128.15 (CH), 128.20 (CH), 128.31 (CH), 128.51 (CH),
136.10 (Cquat), 136.28 (CH), 136.71 (Cquat), 137.37 (Cquat), 137.63 (CH), 137.73 (Cquat), 143.93
(Cquat); ESI (TIS)-Q m/z 446.32 [M+1]+, C28H23N5O.
Compound 5e: Reaction time: 6 days; 17 % overall yield; Rf 0.29 (5 % MeOH/DCM);m. p.
212-213 °C; UV λmax 456 nm; 1H NMR (DMSO-d6) 3.707 (s, 3H, OCH3), 3.749 (s, 3H,
OCH3), 5.782 (bs, 1H, CH), 6.446 (bs, 1H, NH-CH), 6.870 (bd, 1H, J 7.2, CH-6 Py’), 6.915 (d,
1H, J 8.5, CH-5 Ar’), 7.008 (bm, 1H, CH-6 Ar’), 7.055 (dd, 1H, J 8.3, 4.6, CH-5 Py’), 7.141 (d, 1H, J 1.9, CH-2 Ar’), 7.426 (bs, 1H, CH-5 Py), 7.550 (bt, 1H, J 7.2, CH-7 Ar), 7.709 (ddd, 1H, J 8.2, 7.2, 1.3, CH-6 Ar), 7.783 (d, 1H, J 4.2, CH-4 Py’), 7.919 (bs, 1H, CH-4 Py), 8.051 (d, 1H, J 2.7, CH-2 Py’), 8.081 (bs, 1H, CH-8 Ar), 8.267 (bs, 1H, CH-6 Py), 8.375 (bs, 1H,
7 CH-3 Ar), 8.476 (bs, 1H, CH-5 Ar), 8.784 (bs, 1H, CH-2 Py), 11.801 (bs, 1H, NH-N); 13C NMR 54.98 (CH), 55.59 (OCH3), 55.70 (OCH3), 111.79 (CH-5 Ar’), 111.78 (CH-2 Ar’),
118.73 (CH-6 Py’), 119.82 (CH-6 Ar’), 119.88 (Cquat), 122.90 (CH-5 Ar), 123.66 (CH-5 Py’),
123.74 (CH-6 Py), 124.35 (CH-5 Py), 125.62 (CH-8 Ar), 128.05 (CH-7 Ar), 132.45 (CH-6 Ar), 136.23 (CH-4 Py), 136.348 (CH-2 Py’), 136.34 (Cquat), 137.64 (CH-4 Py’), 143.98 (Cquat),
148.24 (Cquat), 148.80 (Cquat); ESI (TIS)-Q m/z 492.28 [M+1]+, C29H25N5O3. Reaction between 5b and acetaldehyde
Version 1. To a solution of 5b (0.2 mmol) in DMF (6 ml; 3x10-2 M) acetaldehyde (1 mmol)
was added and was kept at room temperature for 6 days. The reaction mixture was poured in water and the solid phase formed was filtered off. The product was purified by flash chromatography on silica gel.
Compound 7: 51 % yield; Rf 0.51 (5 % MeOH/DCM);m. p. 116-117 °C decomp.; UV λmax 408
nm; 1H NMR (CDCl3) 2.349 (ddd, 1H, J 13.4, 8.4, 2.4, ½ CH2), 2.421 (ddd, 1H, J 13.4, 11.0,
5.6, ½ CH2), 4.633 (dd, 1H, J 8.3, 5.8, CH-Ar’), 5.849 (dd, 1H, J 5.1, 2.3, CH-OH), 6.941 (dd,
1H, J 5.0, 1.3, CH-4 Ar’), 6.996 (ddd, 1H, J 2.9, 1.3, 0.6, CH-2 Ar’), 7.305 (dd, 1H, J 5.0, 3.0, CH-5 Ar’), 7.413 (ddd, 1H, J 8.1, 4.7, 0.7, CH-5 Py), 7.510 (ddd, 1H, J 8.1, 6.8, 1.2, CH-7 Ar), 7.588-7.616 (ddd, 1H, J 1., CH-6 Ar, overlapped with CH-3 Ar), 7.602 (d, 1H, J 0.5, CH-3 Ar), 8.156 (ddd, 1H, J 8.2, 2.3, 1.6, CH-4 Py), 8.281 (dt, 1H, J 8.1, 0.8, CH-8 Ar), 8.622 (dd, 1H, J 4.7, 1.6, CH-6 Py), 8.851 (dt, 1H, J 8.2, 0.6, CH-5 Ar), 9.126 (d, 1H, J 2.0, CH-2 Py); 13C NMR 33.80 (CH-Ar’), 36.02 (CH-OH), 114.51 (CH-3 Ar), 118.47 (Cquat-2 Ar), 122.02 (CH-8
Ar), 122.70 (CH-2 Ar’), 122.98 (CH-5 Ar), 124.09 (CH-5 Py), 125.33 (Cquat-4 Ar), 126.13
(CH-7 Ar), 126.51 (CH-5 Ar’), 127.22 (CH-4 Ar’), 127.46 (CH-6 Ar), 127.73 (CH-4 Py), 131.95 (Cquat-4a Ar), 141.30 (Cquat-8a Ar), 144.66 (Cquat-1 Ar’), 146.44 (CH-2 Py), 148.53
(Cquat-1 Py), 150.36 (CH-6 Py), 151.68 (Cquat-1 Ar); ESI (TIS)-Q m/z 388.58 [M+1]+,
C22H17N3O2S.
Version 2. To a suspension of 5b (0.1 mmol) in ethanol (20 ml) acetaldehyde (1 mmol) was
added and the mixture was stirred at room temperature for 4 days. Complete dissolution was reached at the end of the reaction; followed by TLC. The solvent was evaporated and the product was purified by flash chromatography on silica gel followed by preparative TLC.
Compound 6: 67 % yield; Rf 0.49 (5 % MeOH/DCM);m. p. 188-189 °C; UV λmax 450 nm; 1H
NMR (CDCl3) 1.377 (t, 3H, J 7.0, CH3), 3.762 (q, 2H, J 7.0, CH2), 5.826 (bs, 1H, CH-Ar’),
7.109 (dd, 1H, J 5.1, 1.0, CH-4 Ar’), 7.208 (dd, 1H, J 2.6, 0.8, CH-2 Ar’), 7.29 (dd, 1H, J 5.1, 3.1, CH-5 Ar’), 7.470 (dd, 1H, J 8.1, 4.7, CH-5 Py), 7.606 (ddd, 1H, J 8.2, 6.9, 1.3, CH-7 Ar), 7.693 (ddd, 1H, J 8.4, 6.8, 1.3, CH-6 Ar), 7.737 (s, 1H, CH-3 Ar), 8.226 (ddd, 1H, J 8.1, 2.0,
8 1.8, CH-4 Py), 8.366 (dd, 1H, J 8.2, 0.8, CH-8 Ar), 8.679 (dd, 1H, J 4.6, 1.3, CH-6 Py), 8.925 (dd, 1H, J 8.4, 0.8, CH-5 Ar), 9.223 (d, 1H, J 1.9, CH-2 Py), 9.676 (bs, 0.8, NH); 13C NMR 15.18 (CH3), 65.76 (CH2), 81.53 (CH-Ar’), 113.23 (CH-3 Ar), 117.92 (Cquat-2 Ar), 122.55
(CH-8 Ar), 122.78 (CH-5 Ar), 123.26 (CH-2 Ar’), 124.06 (CH-5 Py), 125.80 (Cquat-4 Ar),
126.03 (CH-7 Ar), 126.53 (CH-4 Ar’), 126.79 (CH-5 Ar’), 127.05 (CH-4 Py), 128.05 (CH-6 Ar), 132.66 (Cquat-4a Ar), 140.67 (Cquat-8a Ar), 141.16 (Cquat-1 Ar’), 147.05 (CH-2 Py), 148.54
(Cquat-1 Py), 150.72 (CH-6 Py), 155.89 (Cquat-1 Ar); ESI (TIS)-Q m/z 389.54 [M+1]+,
C22H20N4OS.
Crystallography
The crystals of 4 perchlorate, 6 and 7 were mounted on a glass capillary and all geometric and intensity data were taken from these crystals. Diffraction data were taken on an Agilent SupernovaDual diffractometer equipped with an Atlas CCD detector using micro-focus Mo Kα radiation (λ = 0.71073 Å) at room temperature. The determination of the unit cell parameters, data collection and reduction were performed with Crysalispro software [S1]. The structures were solved by direct methods and refined by the full-matrix least-squares method on F2 with
ShelxS and ShelxL 2018/1 programs [S2]. All non-hydrogen atoms, including solvent molecules, were located successfully from Fourier maps and were refined anisotropically. The H atoms were placed in idealized positions (C—H = 0.86 to 0.93 Å) and were constrained to ride on their parent atoms, with Uiso(H) = 1.2Ueq(C) or 1.5Ueq(Cmethyl). The most important
crystallographic and refinement indicators are listed on Table S1.
References
[S1] Rigaku Oxford Diffraction, CrysAlisPro Software system, version 1.171.37.35, Rigaku Corporation, Oxford, UK 2018.
9 Table S1. Crystal data and the most important structure refinement indicators for compounds 4 as perchlorate, 6 and 7.
Identification code Compound 4
perchlorate Compound 6 Compound 7 Empirical formula C23H22 Cl2N6O9 C22 H20 N4 O S, H2 O C22 H17 N3 O2 S, 0.5(C6 H6) Formula weight 597.36 406.5 426.50 Temperature/K 290 290 290
Crystal system Triclinic Monoclinic Monoclinic
Space group P-1 P21/c P21/c a/Å 8.6004(13) 15.582(2) 5.0282(8) b/Å 10.5677(13) 5.5480(9) 19.643(3) c/Å 14.8352(17) 23.037(3) 22.121(3) α/° 107.404(11) 90 90 β/° 95.633(11) 90.669 91.341(13) γ/° 92.707(11) 90 90
Unit cell volume /Å3 1276.2(3) 1991.3(5) 2151.3(6)
Z 2 4 4
ρcalcg/cm3 1.555 1.356 1.317
μ/mm-1 0.320 0.189 0.178
F(000) 616.0 856 892
Crystal size/mm3 0.25 × 0.15 × 0.10 0.25 × 0.15 × 0.15 0.3 × 0.25 × 0.25
Radiation MoKα (λ = 0.71073) MoKα (λ = 0.71073) MoKα (λ = 0.71073)
Θ range for data collection/° 2.9 to 28.4 3.1 to 27.1 2.8 to 29.1
Index ranges -7 ≤ h ≤ 10, -12 ≤ k ≤ 14, -17 ≤ l ≤ 18 -18 ≤ h ≤ 16, -6 ≤ k ≤ 7, -29 ≤ l ≤ 28 -6 ≤ h ≤ 5, -25 ≤ k ≤ 25, -20 ≤ l ≤ 30 Reflections collected/independent/ I > 2σ(I) 8992 / 5212/ 2556 9766 /3585/ 1324 11756/4981/ 1470 Rint / Rsigma 0.063 / 0.0328 0.095 / 0.1909 0.1005 / 0.133 Data/restraints/parameters 5212/48/391 3585/0/286 4981/ 14/ 300 Goodness-of-fit on F2 1.032 1.030 0.948
R1, wR2 indexes, I>=2σ (I) 0.01029, 0.2525 0.095/0.150 0.0907 /0.2416
R1, wR2 indexes, all data 0.1954, 0. 3107 0.259/0.209 0.2472 / 0.3569
Largest diff. peak/hole / e Å-3 0.0.8/-0.6 0.26/-0.30 0.27 / -0.27
10 I) NMR spectra of starting azo-dye 1
Figure S1. 1H spectrum of 1 in DMSO-d6.
Figure S2. 1H-1H COSY spectrum of 1 in DMSO-d6.
7.0 7.5 8.0 8.5 9.0 9.5 10.0 10.5 11.0 11.5 ppm 7.458 7.472 7.481 7.542 7.553 7.595 7.608 7.616 7.621 7.629 7.707 7.709 7.718 7.721 7.723 7.732 7.735 7.852 7.867 7.891 7.903 7.927 7.941 8.017 8.029 8.061 8.074 8.103 8.118 8.241 8.255 8.285 8.299 8.447 8.459 8.694 8.696 8.702 8.751 8.790 8.913 8.928 9.160 9.163 11.339 11.686 0.21 1.00 0.23 2.25 1.24 1.20 0.20 2.34 0.21 1.14 0.98 0.90 1.02 0.21
11 Figure S3. 1H-1H NOESY spectrum of 1 in DMSO-d6.
Figure S4. 13C (down) and DEPT (up) spectra of 1 in DMSO-d6.
105 110 115 120 125 130 135 140 145 150 155 160 ppm 108.93 114.87 122.90 123.07 124.78 125.05 126.08 127.25 128.56 133.27 140.13 146.70 148.60 151.48 158.99
12 Figure S5. 1H-13C HSQC spectrum of 1 in DMSO-d6.
13 II) NMR spectra of 1-OMe
Figure S7. 1H spectrum of 1-OMe in CDCl3.
Figure S8. 1H-1H COSY spectrum of 1-OMe in CDCl3.
4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 ppm 4.098 6.914 6.928 7.473 7.481 7.487 7.495 7.580 7.581 7.591 7.593 7.595 7.605 7.607 7.681 7.683 7.692 7.694 7.697 7.706 7.708 7.969 7.983 8.249 8.251 8.255 8.262 8.265 8.268 8.321 8.335 8.686 8.693 8.936 8.950 9.258 3.17 1.04 1.11 1.18 1.05 1.06 1.03 0.99 1.10 1.00 1.07 7.0 7.2 7.4 7.6 7.8 8.0 8.2 8.4 8.6 8.8 9.0 9.2 ppm
14 Figure S9. 1H-1H NOESY spectrum of 1-OMe in CDCl3.
Figure S10. 13C (down) and DEPT (up) spectra of 1-OMe in CDCl3.
60 70 80 90 100 110 120 130 140 150 160 ppm 55.98 103.73 113.59 122.24 122.96 124.12 125.59 126.03 127.38 127.90 132.79 141.67 146.86 148.65 150.56 159.25 60 70 80 90 100 110 120 130 140 150 160 ppm
15 Figure S11. 1H-13C HSQC spectrum of 1-OMe in CDCl3.
16 III) NMR spectra of 1-NMe
Figure S13. 1H spectrum of 1-NMe in CDCl3.
Figure S14. 1H-1H COSY spectrum of 1-NMe in CDCl3.
4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 ppm 3.790 6.741 6.758 7.548 7.566 7.616 7.618 7.630 7.642 7.643 7.699 7.702 7.713 7.715 7.725 7.727 7.967 7.979 8.207 8.209 8.220 8.222 8.357 8.365 8.397 8.410 8.711 3.09 0.99 1.24 2.11 1.13 1.00 0.99 0.95 1.00 0.98 6.8 6.9 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 7.8 7.9 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 ppm
17 Figure S15. 1H-1H NOESY spectrum of 1-NMe in CDCl3.
Figure S16. 13C (down) and DEPT (up) spectra of 1-NMe in CDCl3.
50 60 70 80 90 100 110 120 130 140 150 160 170 180 190 ppm 45.62 124.21 125.28 126.40 128.67 130.09 130.82 131.05 132.89 184.54 50 60 70 80 90 100 110 120 130 140 150 160 170 180 190 ppm
18 Figure S17. 1H-13C HSQC spectrum of 1-NMe in CDCl3.
19
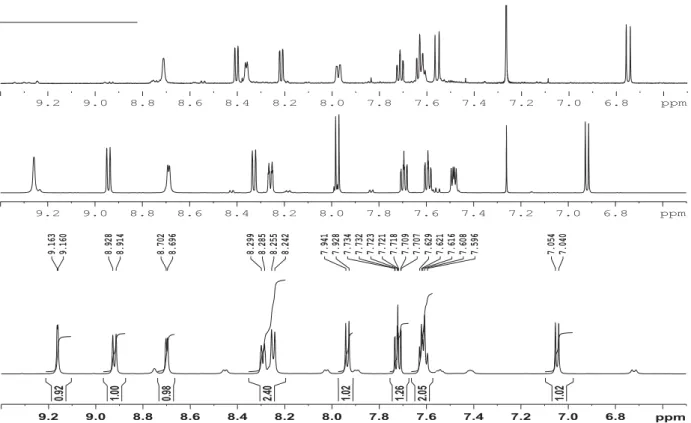
IV) Comparison between 1H and 13C NMR spectra of compounds 1, 1-OMe and 1-NMe
Figure S19. 1H NMR spectra of 4-(pyridin-3-yldiazenyl)naphthalen-1-ol (1) in DMSO-d6
(down), 1-OMe in CDCl3 (middle), and 1-NMe CDCl3 (up).
Figure S20. 13C NMR spectra of 4-(pyridin-3-yldiazenyl)naphthalen-1-ol (1) in DMSO-d6
(down), 1-OMe in CDCl3 (middle), and 1-NMe CDCl3 (up).
6.8 7.0 7.2 7.4 7.6 7.8 8.0 8.2 8.4 8.6 8.8 9.0 9.2 ppm 7.040 7.054 7.596 7.608 7.616 7.621 7.629 7.707 7.709 7.718 7.721 7.723 7.732 7.734 7.928 7.941 8.242 8.255 8.285 8.299 8.696 8.702 8.914 8.928 9.160 9.163 1.02 2.05 1.26 1.02 2.40 0.98 1.00 0.92 6.8 7.0 7.2 7.4 7.6 7.8 8.0 8.2 8.4 8.6 8.8 9.0 9.2 ppm 6.8 7.0 7.2 7.4 7.6 7.8 8.0 8.2 8.4 8.6 8.8 9.0 9.2 ppm 110 115 120 125 130 135 140 145 150 155 160 165 170 175 180 185 ppm 108.93 114.87 122.90 123.07 124.78 125.05 126.08 127.25 128.56 133.27 140.13 146.70 148.60 151.48 158.99 103.734 113.588 122.241 122.958 124.123 125.591 126.028 127.380 127.900 132.788 141.672 146.861 148.651 150.557 159.253 124.213 125.260 125.277 126.396 128.674 130.093 130.818 131.049 132.888 184.541
20 V) NMR spectra of compound 2
Figure S21. 1H spectrum of 2 in CDCl3.
Figure S22. 1H-1H COSY spectrum of 2 in CDCl3.
2 3 4 5 6 7 8 9 10 11 ppm 1.699 3.063 3.910 7.424 7.431 7.437 7.445 7.537 7.539 7.548 7.550 7.553 7.562 7.564 7.627 7.628 7.638 7.640 7.643 7.652 7.654 7.762 8.197 8.200 8.203 8.210 8.213 8.217 8.302 8.316 8.642 8.644 8.649 8.652 8.904 8.918 9.219 9.222 10.737 6.31 4.97 2.24 2.30 1.17 1.14 1.15 1.06 1.08 1.09 1.05 1.06 1.00 1.04
21 Figure S23. 1H-1H NOESY spectrum of 2 in CDCl3.
Figure S24. 13C (down) and DEPT (up) spectra of 2 in CDCl3.
30 40 50 60 70 80 90 100 110 120 130 140 150 160 ppm 23.79 25.72 29.73 30.97 53.86 62.01 113.88 114.06 122.44 122.71 123.99 125.05 125.51 126.67 127.65 132.79 139.76 147.15 148.66 150.51 160.08 30 40 50 60 70 80 90 100 110 120 130 140 150 160 ppm
22 Figure S25. 1H-13C HSQC spectrum of 2 in CDCl3.
23 VI) NMR spectra of compound 3
Figure S27. 1H spectrum of 3 in DMSO-d6.
Figure S28. 1H-1H COSY spectrum of 3 in DMSO-d6.
4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 ppm 4.259 5.092 6.271 6.943 6.954 6.956 7.073 7.081 7.087 7.094 7.132 7.139 7.146 7.153 7.415 7.472 7.475 7.486 7.489 7.547 7.559 7.572 7.703 7.715 7.727 7.784 7.790 7.918 7.975 7.982 8.058 8.112 8.216 8.271 8.420 8.424 8.496 8.773 2.12 2.17 0.85 1.00 1.05 1.09 1.09 1.15 1.11 1.26 1.09 1.24 1.17 1.26 1.27 0.95 1.30 1.02 1.00 7.0 7.2 7.4 7.6 7.8 8.0 8.2 8.4 8.6 8.8 ppm
24 Figure S29. 1H-1H ROESY spectrum of 3 in DMSO-d6.
Figure S30. 13C (down) and DEPT (up) spectra of 3 in DMSO-d6.
60 70 80 90 100 110 120 130 140 150 160 170 180 ppm 42.36 54.98 66.34 117.98 119.84 122.90 123.46 123.72 123.78 123.87 124.31 124.51 124.74 124.95 125.13 125.40 127.75 131.97 132.13 135.40 135.70 136.11 136.24 136.81 137.28 139.33 141.14 144.13 144.83 144.97 153.33
25 Figure S31. 1H-13C HSQC spectrum of 3 in DMSO-d6.
26 VII) NMR spectra of compound 4
Figure S33. 1H spectrum of 4 in CDCl3.
Figure 34. 1H spectra of 4 at 300K (down), 328K (middle) and 358K (up) in DMSO-d6.
5 6 7 8 9 10 11 12 ppm 4.258 6.274 6.938 6.940 6.942 6.945 6.952 6.954 6.956 6.959 7.071 7.072 7.079 7.080 7.085 7.093 7.418 7.547 7.549 7.561 7.572 7.574 7.702 7.704 7.714 7.718 7.727 7.730 7.783 7.785 7.791 7.793 7.917 7.921 8.057 8.061 8.224 8.269 8.496 8.779 11.702 2.00 0.38 0.28 0.91 0.93 1.01 1.12 1.24 1.29 0.92 0.33 1.15 1.10 1.28 1.06 1.00 1.17 0.28 0.87 1.10 7.0 7.2 7.4 7.6 7.8 8.0 8.2 8.4 8.6 8.8 ppm 7.4 7.5 7.6 7.7 7.8 7.9 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 8.8 8.9 ppm 7.4 7.5 7.6 7.7 7.8 7.9 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 8.8 8.9 ppm 7.4 7.5 7.6 7.7 7.8 7.9 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 8.8 8.9 ppm
27 VIII) NMR spectra of compound 5a
Figure S35. 1H spectrum of 5a in DMSO-d6 at 343K (down) and at 302K (up)
Figure S36. 1H spectrum of 5a in DMSO-d6 at 343K.
7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 7.8 7.9 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 8.8 8.9 ppm 6.5 7.0 7.5 8.0 8.5 9.0 9.5 10.0 10.5 11.0 11.5 12.0 ppm 7.057 7.068 7.243 7.256 7.261 7.266 7.277 7.280 7.283 7.332 7.337 7.341 7.353 7.356 7.370 7.375 7.420 7.431 7.440 7.452 7.529 7.542 7.547 7.550 7.567 7.570 7.682 7.685 7.699 7.702 7.706 7.720 7.723 7.803 7.806 7.814 7.818 7.942 7.961 7.966 8.108 8.115 8.131 8.151 8.321 8.347 8.355 8.537 8.556 8.835 8.840 11.449 1.00 0.75 1.12 1.02 1.14 2.16 1.27 3.09 1.26 1.00 1.16 2.09 2.04 1.04 0.94 0.79 7.0 7.2 7.4 7.6 7.8 8.0 8.2 8.4 8.6 8.8 ppm
28 Figure S37. 1H-1H COSY spectrum of 5a in DMSO-d6 at 343K.
Figure S38. 1H-1H NOESY spectrum of 5a in DMSO-d6 at 343K.
ppm 6 7 8 9 10 11 12 ppm 4 5 6 7 8 9 10 11
29 Figure S39. 1H-13C HSQC spectrum of 5a in DMSO-d6 at 343K.
30 selective NOE experiments at 298K
Figure S41. Irradiation at 5.86 ppm.
31 Figure S43. Irradiation at 6.90 ppm.
32 Figure S45. Irradiation at 7.26 ppm.
33 Figure S47. Irradiation at 7.43 ppm.
34 Figure S49. Irradiation at 7.70 ppm.
35 Figure S51. Irradiation at 7.94 ppm.
36 Figure S53. Irradiation at 8.30 ppm.
37 Figure S55. Irradiation at 8.51 ppm.
38 IX) NMR spectra of compound 5b
Figure S57. 1H spectrum of 5b in DMSO-d6.
X) NMR spectra of compound 5c
Figure S58. 1H spectrum of 5c in DMSO-d6 at 373K.
6.5 7.0 7.5 8.0 8.5 9.0 9.5 10.0 10.5 11.0 11.5 12.0 ppm 5.981 6.543 6.887 6.900 7.040 7.048 7.054 7.062 7.230 7.237 7.428 7.433 7.440 7.448 7.466 7.510 7.515 7.518 7.523 7.541 7.553 7.566 7.690 7.692 7.704 7.716 7.717 7.775 7.782 7.958 8.066 8.071 8.131 8.329 8.362 8.541 8.817 11.756 0.94 0.82 1.05 1.05 1.00 1.99 1.03 1.11 1.20 1.09 1.03 2.18 1.94 1.01 1.02 0.79 6.5 7.0 7.5 8.0 8.5 ppm 6.029 6.089 6.963 6.965 7.016 7.018 7.030 7.032 7.064 7.070 7.356 7.426 7.434 7.439 7.535 7.536 7.548 7.560 7.561 7.689 7.701 7.713 7.715 7.798 7.811 7.876 7.890 7.942 7.957 8.068 8.081 8.145 8.159 8.310 8.371 8.467 8.546 8.560 8.766 8.849 1.00 2.01 2.02 1.04 1.09 2.97 2.03 4.06 2.08
39 Figure S59. 1H-13C HSQC spectrum of 5c in DMSO-d6 at 373K.
XI) NMR spectra of compound 5d
Figure S60. 1H spectrum of 5d in DMSO-d6.
3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 9.5 10.0 10.5 11.0 11.5 12.0 ppm 2.293 2.448 2.464 2.477 2.487 2.500 2.708 2.711 2.713 2.716 2.720 3.387 3.397 3.448 3.539 3.623 5.966 6.695 7.080 7.093 7.114 7.116 7.118 7.121 7.127 7.130 7.132 7.134 7.220 7.221 7.228 7.229 7.259 7.266 7.273 7.280 7.287 7.300 7.411 7.424 7.436 7.441 7.454 7.466 7.483 7.528 7.540 7.557 7.643 7.723 7.747 7.759 7.771 7.895 7.897 7.906 7.908 7.918 7.920 7.922 7.931 7.933 7.938 7.940 7.994 8.001 8.132 8.136 8.219 8.270 8.274 8.352 8.499 8.543 8.554 8.613 8.692 9.005 3.00 0.83 0.84 1.05 2.18 1.40 2.12 1.19 1.28 1.29 1.06 1.33 2.14 1.18 0.88 6.0 6.5 7.0 7.5 8.0 8.5 9.0 ppm
40 XII) NMR spectra of compound 5e
Figure S61. 1H spectrum of 5e in DMSO-d6.
Figure S62. 1H-1H NOESY spectrum of 5e in DMSO-d6.
5 6 7 8 9 10 11 12 ppm 3.707 3.749 6.446 6.856 6.864 6.876 6.908 6.922 7.000 7.008 7.013 7.044 7.052 7.058 7.066 7.139 7.142 7.426 7.538 7.550 7.562 7.696 7.698 7.709 7.711 7.721 7.723 7.779 7.786 7.917 7.920 8.049 8.054 8.267 8.375 8.476 8.784 11.801 2.96 1.96 0.82 1.00 1.10 0.96 1.05 1.03 1.00 1.06 1.15 1.03 1.12 2.00 3.21 1.03 0.84 7.0 7.2 7.4 7.6 7.8 8.0 8.2 8.4 8.6 8.8 ppm
41 Figure S63. 13C spectrum of 5e in DMSO-d6.
Figure S64. 1H-13C HSQC spectrum of 5e in DMSO-d6.
60 70 80 90 100 110 120 130 140 150 160 ppm 55.59 55.70 111.78 118.73 119.82 119.88 122.90 123.66 124.35 136.34 137.64 143.98 148.24 148.80
42 XIII) NMR spectra of compound 7
Figure S65. 1H spectrum of 7 in CDCl3. Figure S66. 13C spectrum of 7 in CDCl3. 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 ppm 2.329 2.333 2.344 2.348 2.352 2.356 2.366 2.370 2.400 2.410 2.419 2.422 2.432 2.441 4.621 4.631 4.635 4.645 5.843 5.847 5.851 5.855 6.936 6.938 6.944 6.946 6.992 6.992 6.994 6.995 6.996 6.997 6.998 7.000 7.298 7.303 7.307 7.312 7.402 7.403 7.410 7.411 7.416 7.417 7.424 7.425 7.496 7.498 7.508 7.510 7.513 7.521 7.524 7.588 7.590 7.603 7.614 7.616 8.146 8.149 8.150 8.153 8.160 8.162 8.164 8.166 8.274 8.287 8.617 8.619 8.624 8.627 8.844 8.845 8.858 9.124 9.128 2.00 1.54 1.61 0.19 0.86 0.91 0.48 0.19 0.84 0.85 0.19 1.05 1.12 1.34 1.96 0.53 1.07 0.86 0.20 1.09 1.05 1.04 7.0 7.2 7.4 7.6 7.8 8.0 8.2 8.4 8.6 8.8 9.0 9.2 ppm 40 50 60 70 80 90 100 110 120 130 140 150 160 ppm 29.73 31.54 33.80 36.02 36.32 36.60 37.52 76.87 77.08 77.29 92.48 95.19 114.20 114.51 118.23 118.47 122.02 122.18 122.29 122.70 122.98 124.09 125.29 125.33 126.13 126.25 126.51 126.81 127.05 127.22 127.46 127.61 127.73 131.95 141.30 141.40 144.02 144.66 146.44 148.53 150.36 150.44 151.68 152.30 162.74
43 Figure S67. 1H-1H COSY spectrum of 7 in CDCl3.
Figure S68. 1H-13C HSQC spectrum of 7 in CDCl3. ppm 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 ppm 3 4 5 6 7 8 9 ppm 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 ppm 30 40 50 60 70 80 90 100 110 120 130 140 150
44 Figure S69. 1H-13C HMBC spectrum of 7 in CDCl3.
XIV) NMR spectra of compound 6
Figure S70. 1H spectrum of 6 in CDCl3. ppm 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 ppm 30 40 50 60 70 80 90 100 110 120 130 140 150 2.0 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 9.5 ppm 1.366 1.377 1.389 3.744 3.756 3.768 3.780 5.826 7.104 7.105 7.112 7.114 7.206 7.210 7.263 7.286 7.291 7.295 7.300 7.459 7.467 7.472 7.480 7.592 7.606 7.618 7.679 7.681 7.693 7.704 7.706 7.737 8.216 8.220 8.223 8.230 8.233 8.236 8.359 8.373 8.676 8.682 8.918 8.932 9.035 3.86 2.08 1.08 1.08 1.23 1.03 1.72 1.36 1.18 1.12 1.78 1.17 1.02 1.03 1.00 0.80 7.2 7.4 7.6 7.8 8.0 8.2 8.4 8.6 8.8 9.0 9.2 ppm
45 Figure S71. 13C spectrum of 6 in CDCl3.
Figure S72. 1H-1H NOESY spectrum of 6 in CDCl3.
20 30 40 50 60 70 80 90 100 110 120 130 140 150 160 ppm 15.18 65.76 81.53 113.23 117.92 122.55 122.78 123.26 124.06 125.51 125.80 126.03 126.53 126.71 126.79 127.05 128.05 132.36 132.66 140.67 141.16 147.05 148.54 150.72 155.89 ppm 1 2 3 4 5 6 7 8 9 ppm 2 3 4 5 6 7 8 9
46 Figure S73. 1H-13C HSQC spectrum of 6 in CDCl3. Figure S74. 1H-13C HMBC spectrum of 6 in CDCl3. ppm 1.5 2.0 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 9.5 ppm 20 40 60 80 100 120 140 ppm 1.5 2.0 2.5 3.0 3.5 4.0 4.5 5.0 5.5 6.0 6.5 7.0 7.5 8.0 8.5 9.0 9.5 ppm 20 40 60 80 100 120 140 160
47 XV) UV spectra
Figure S75. UV spectra of compounds 1, 1-OMe, 1-NMe, and 2 in acetone.
48 Figure S77. UV spectra of compounds 5a-5e in acetone.