Publisher’s version / Version de l'éditeur:
Cement and Concrete Research, 3, 3, pp. 233-245, 1973-05
READ THESE TERMS AND CONDITIONS CAREFULLY BEFORE USING THIS WEBSITE. https://nrc-publications.canada.ca/eng/copyright
Vous avez des questions? Nous pouvons vous aider. Pour communiquer directement avec un auteur, consultez la première page de la revue dans laquelle son article a été publié afin de trouver ses coordonnées. Si vous n’arrivez pas à les repérer, communiquez avec nous à PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca.
Questions? Contact the NRC Publications Archive team at
PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca. If you wish to email the authors directly, please see the first page of the publication for their contact information.
NRC Publications Archive
Archives des publications du CNRC
This publication could be one of several versions: author’s original, accepted manuscript or the publisher’s version. / La version de cette publication peut être l’une des suivantes : la version prépublication de l’auteur, la version acceptée du manuscrit ou la version de l’éditeur.
For the publisher’s version, please access the DOI link below./ Pour consulter la version de l’éditeur, utilisez le lien DOI ci-dessous.
https://doi.org/10.1016/0008-8846(73)90028-8
Access and use of this website and the material on it are subject to the Terms and Conditions set forth at
Alkali-aggregate reaction in Nova Scotia. III. Laboratory studies of
volume change
Duncan, M. A. G.; Swenson, E. G.; Gillott, J. E.
https://publications-cnrc.canada.ca/fra/droits
L’accès à ce site Web et l’utilisation de son contenu sont assujettis aux conditions présentées dans le site LISEZ CES CONDITIONS ATTENTIVEMENT AVANT D’UTILISER CE SITE WEB.
NRC Publications Record / Notice d'Archives des publications de CNRC:
https://nrc-publications.canada.ca/eng/view/object/?id=428ff7cd-2507-453a-9d1d-b80a51f404c5 https://publications-cnrc.canada.ca/fra/voir/objet/?id=428ff7cd-2507-453a-9d1d-b80a51f404c5CEMENT and CONCRETE RESEARCH. Vol. 3 , pp. 233-245, 1973. Pergamon P r e s s , I n c . P r i n t e d i n t h e United S t a t e s .
ALKALI-AGGREGATE REACTION IN NOVA SC OTIA
LII. LABORATORY STUDIES O F VOLUME CHANGE
M.A.G. Duncan, Atlantic Lndustrial R e s e a r c h I n s t i t u t e , Halifax, now with M i n i s t r y of R o a d s , S a l i s b u r y , R h o d e s i a ,
E . G. Swenson, Div. of Building R e s e a r c h , Nat. R e s . Council of Canada, and,
J.E. Gillott, Div. of Building R e s e a r c h , Nat. R e s . Council of Canada, now with Univ. of C a l g a r y , C a l g a r y , A l b e r t a .
(Communicated by G .
M .
Idorn)ABSTRACT
Studies of alkali-expansivity of Nova Scotia r o c k t y p e s included extensive length change t e s t s of m o r t a r b a r s and c o n c r e t e p r i s m s , and a l s o r o c k c y l i n d e r s i m m e r s e d i n a l k a l i solution. The e f f e c t s of s u c h v a r i a b l e s a s a l k a l i content, type of a l k a l i , t e m p e r a t u r e , wet and d r y cycling, a n i s o t r o p y , optimum p r o - portion of r o c k type, and t h e influence of pozzolan and f l y a s h w e r e studied. R e s u l t s obtained w e r e c o r r e l a t e d with p e t r o - g r a p h i c s t u d i e s so t h a t potentially expansive r o c k t y p e s in t h e province m a y be recognized. P r e v e n t i v e m e a s u r e s applicable t o field u s e w e r e d e t e r m i n e d f r o m a n a l y s e s of expansion data.
SOMMAIRE
L e s Ctudes d e l a dilatation due a u x a l c a l i s de c e r t a i n s g e n r e s de r o c h e s en Nouvelle - E c o s s e c o m p r e n n e n t d e n o m b r e u x e s s a i s de changement d e longueur s u r d e s b a r r e s de m o r t i e r e t d e s p r i s m e s de biiton, e t a u s s i s u r d e s c y l i n d r e s d e roche plongCs d a n s une solution alcaline. On Ctudie iigalement l e r61e d e v a r i - a b l e s c o m m e l a t e n e u r e n a l c a l i s , l e s g e n r e s d ' a l c a l i s , l a t e m - p g r a t u r e , un cycle humide ou s e c , l ' a n i s o t r o p i e , l a p r o p o r t i o n o p t i m a l e d e s g e n r e s d e r o c h e s e t l'influence de l a pouzzolane ou d e l a c e n d r e volante. L e s r i i s u l t a t s obtenus sont m i s e n corrClation a v e c l e s Ctudes ~ C t r o g r a p h i q u e s afin que l ' o n p u i s s e r e c o n n a i t r e , d a n s l a province, l e s g e n r e s de r o c h e s
s u s c e p t i b l e s d e s u b i r une dilatation. L ' a n a l y s e d e s donnges de l a dilatation a p e r m i s d r C t a b l i r d e s m e s u r e s p r g v e n t i v e s a p p l i - c a b l e s i n situ.
V o l . 3 , No. 3
A L K A L I AGGREGATE REACTION, LABORATORY, EXPANSION
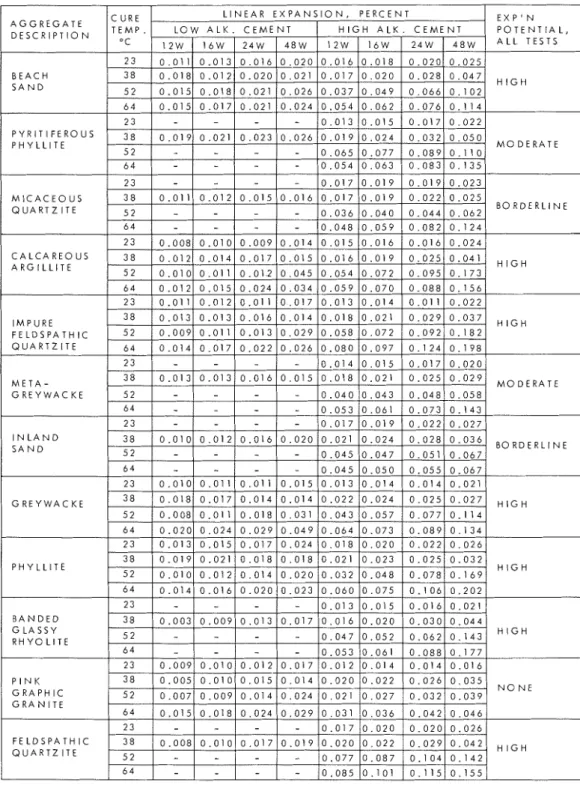
T h i s i s t h e t h i r d i n a s e r i e s of p a p e r s d e s c r i b i n g a f i v e - y e a r s t u d y of a l k a l i - a g g r e g a t e r e a c t i o n i n Nova S c o t i a . T h e f i r s t p a p e r ( 1 ) d e s c r i b e d t h e g e n e r a l i n v e s t i g a t i o n , c o n c l u s i o n s , and r e c o m m e n d a t i o n s , t h e s e c o n d ( 2 ) d e a l t with f i e l d s t u d i e s and p e t r o g r a p h i c e x a m i n a t i o n s ; t h e p r e s e n t p a p e r p r o v i d e s a d d i t i o n a l r e s u l t s of v a r i o u s l a b o r a t o r y t e s t s . A s f o r o t h e r a l k a l i - a g g r e g a t e r e a c t i o n s , t h e i d e n t i f i c a t i o n of a l k a l i - e x p a n s i v e r o c k s i n Nova S c o t i a n e c e s s i t a t e d r e p r o d u c t i o n of e x p a n s i o n s i n t h e l a b o r a t o r y a n d p e t r o g r a p h i c a n a l y s e s . B e c a u s e t h e r e a c t i o n w a s e x t r e m e l y s l o w , a s d e m o n s t r a t e d u n d e r n o r m a l c o n d i t i o n s of t e s t , a c c e l e r a t e d t e s t m e t h o d s h a d t o b e u s e d . In e x t r e m e c a s e s t h e s e t e n d e d t o r e d u c e , t o s o m e d e g r e e , t h e r e l i a b i l i t y of r e s u l t s o b t a i n e d f r o m l e n g t h change t e s t s . E x t e n s i v e t e s t i n g w a s t h u s n e c e s s a r y t o d e v e l o p t e s t c o m b i n a t i o n s t h a t could be a p p l i e d with c o n f i d e n c e i n e v a l u a t i n g a g g r e g a t e s o u r c e s i n t h e a r e a . T h e c r i t i c a l v a l u e s of t i m e v e r s u s e x p a n s i o n t h a t a r e i n t e n d e d t o d i s t i n g u i s h s a f e f r o m d e l e t e r i o u s a g g r e g a t e w e r e b a s e d on c o r r e l a t i o n of a l l t e s t d a t a f r o m m o r t a r b a r s , c o n c r e t e p r i s m s , a n d r o c k c y l i n d e r s , a s w e l l a s on f i e l d e x p e r i e n c e . T e s t s w e r e c a r r i e d out i n t r i p l i c a t e w i t h s o m e e x c e p t i o n s , i n which c a s e d u p l i c a t e s w e r e u s e d . M o r t a r B a r s P r o c e d u r e s f o r t e s t i n g m o r t a r b a r s f o r l e n g t h c h a n g e h a v e b e e n d e s c r i b e d ( 1 ) . M o d i f i c a t i o n s i n ASTM t e s t m e t h o d C227 u s e d i n t h e p r e s e n t s t u d i e s a r e n o t e d i n e a c h s e t of d a t a p r e s e n t e d .
T a b l e I s h o w s length change r e s u l t s of m o r t a r b a r s m a d e with t h e low and t h e h i g h a l k a l i c e m e n t s a t f o u r t e m p e r a t u r e s . A l a r g e i n c r e a s e i n t h e r a t e of e x p a n s i o n w a s r e g i s t e r e d b e t w e e n 1 0 0 ° F ( 3 8 ° C ) a n d 1 2 5 ° F ( 5 2 ° C ) f o r n e a r l y a l l c a s e s of a l k a l i - e x p a n s i v e r o c k s . With low a l k a l i c e m e n t t h e e x p a n s i o n s w e r e not e x c e s s i v e a t t h e h i g h e r t e m p e r a t u r e s a l t h o u g h s o m e i n c r e a s e did o c c u r . S t u d i e s of t h i s t y p e a i d e d i n t h e d e v e l o p - m e n t of a c c e l e r a t e d t e s t p r o c e d u r e s . T a b l e 11 g i v e s t y p i c a l e x p a n s i o n s u n d e r c o n d i t i o n s of t e m p e r a t u r e , a l k a l i n i t y a n d t i m e s e l e c t e d f o r e v a l u a t i n g Nova S c o t i a a g g r e g a t e s . Ln g e n e r a l t h e c o r r e l a t i o n , a n d t h e r e f o r e t h e c o n f i d e n c e , d e c r e a s e s w i t h i n - c r e a s i n g a c c e l e r a t i o n of e x p a n s i o n .
V o l . 3, N o . 3
A L K A L I AGGREGATE REACTION, LABORATORY, EXPANSION
Table 1 L I N E A R E X P A N S I O N S O F M O I S T C U R E D M O R T A R BARS M A D E W I T H L O W A N D H I G H A L K A L I C E M E N T S A T F O U R T E M P E R A T U R E S L A = 0 . 3 4 % o n d H A 0 . 8 8 % A L K . , Calculated o r N o 2 0 W = T I M E , W E E K S P Y R I T I F E R O U S P H Y L L I T E M O D E R A T E B O R D E R L I N E A R G l L L l T E I M P U R E M O D E R A T E I N L A N D S A N D G R E Y W A C K E P H Y L L I T E B A N D E D F E L D S P A T H I C Q U A R T Z I T E 2 3 3 8 5 2 6 4 0 . 0 0 8 0 . 0 1 0 0 . 0 1 7 - 0 . 0 1 9 - - 0 . 0 1 7 0 . 0 2 0 0 . 0 7 7 0 . 0 8 5 0 . 0 2 0 0 . 0 2 2 0 . 0 8 7 0 , 1 0 1 0 . 0 2 0 0 . 0 2 9 0 . 1 0 4 0 . 1 1 5 0 . 0 2 6 0 . 0 4 2 0 . 1 4 2 0 . 1 5 5
Vol.
3, No. 3 A L K A L I AGGREGATE R E A C T I O N , LABORATORY, E X P A N S I O NT A B L E I1
T y p i c a l M o r t a r B a r E x p a n s i o n Data using the ASTM C227 Modified T e s t f o r E v a l u a t i o n of Nova S c o t i a A g g r e g a t e
*
c a l c u l a t e d a s Na 02
F i g u r e 1 shows t h e e f f e c t of t o t a l a l k a l i n i t y of c e m e n t p a s t e on 48-week e x p a n s i o n s of m o r t a r b a r s m a d e with a g g r e g a t e s of v a r y i n g type and r e -
Rock Type o r Sand Gr eywacke M e t a - g r e y - wacke C a l c a r e o u s a r g i l l i t e P h y l l i t e I m p u r e f e l d s - pathic q u a r t z i t e M i c a c e o u s q u a r t z i t e P i n k g r a p h i c g r a n i t e a c t i v i t y . F o r t h e s a m e a g g r e g a t e s , s e l e c t e d a s t y p i c a l e x a m p l e s , F i g u r e 2 shows t h e v a r i a t i o n of p e r c e n t e x p a n s i o n with a m o u n t of t e s t a g g r e g a t e r e l a t i v e t o n e u t r a l s a n d p r e s e n t . F o r t h e conditions 1 0 0 ° F ( 3 8 ° C ) 100 p e r - c e n t r e l a t i v e h u m i d i t y , t o t a l a l k a l i n i t y of 1 . 0 0 p e r c e n t c a l c u l a t e d a s N a 2 0 , and a 48 -week d u r a t i o n , t h e p r o p o r t i o n s of a l k a l i - e x p a n s i v e r o c k t y p e s f o r m a x i m u m e x p a n s i o n o c c u r r e d a t f r o m 35 to 100 p e r c e n t c o n c e n t r a - t i o n , i n 26 s a m p l e s t e s t e d . T h e one e x c e p t i o n w a s t h e c h e r t y a g a t e that showed m a x i m u m e x p a n s i o n a t about 4 p e r c e n t c o n c e n t r a t i o n . Six a l k a l i - e x p a n s i v e a g g r e g a t e s , a s d e t e r m i n e d by c o r r e l a t i o n of a l l t e s t d a t a , w e r e u s e d with t h e low a l k a l i c e m e n t with o r without added
E x p ' n P o t e n t i a l , A l l T e s t s High M o d e r a t e High High High B o r d e r l i n e None P e r c e n t L i n e a r E x p a n s i o n a t d i f f e r e n t A l k a l i " C 3 8
70
Alk* 0. 88 Weeks 144.
036.
052 . 0 7 8 - 0 5 5.
051 . 0 3 5 --
64 0. 88 16.
073 . 0 6 1.
070 . 0 7 5.
U97.
059.
018 c o n t e n t s 3 8 1. 04 72.
069 . 0 3 8.
84 . 0 7 5.
102 .042 . 0 3 8 t e m p e r a t u r e s 52 0. 88 24.
077 . 0 4 8.
095.
078.
092 . 0 4 4.
014 and 3 8 1.42 36.
078.
125.
173.
045.
169 . 0 6 9 - - 64 0. 34 4 8.
049 - -.
024.
023.
b26- -
.
017V o l . 3, No. 3
ALKALI AGGREGATE REACTION, LABORATORY, EXPANSION
0 CALCAREOUS
/
1
ARGILLITE PHYLLITE GREYWACKE1s
A L K A L I N I T Y O F C E M E N T P A S T E A S N a 2 0 , PER C E N T FIG. 1The effect of c e m e n t p a s t e a l k a l i n i t y on t h e 48 week p e r c e n t a g e e x p a n s i o n s of m o r t a r b a r s p r e p a r e d f r o m v a r i o u s a g g r e g a t e s
and m o i s t c u r e d a t 1 0 0 ° F ( 3 8 ° C ) in s e a l e d c o n t a i n e r s .
NaOH o r KOH in t h e m i x i n g w a t e r t o obtain v a r i o u s t o t a l a l k a l i n i t i e s c a l c u - l a t e d a s N a 2 0 . Ln the m o r t a r b a r e x p a n s i o n d a t a shown i n T a b l e 111 i t i s s e e n t h a t v e r y l i t t l e d i f f e r e n c e , if any, e x i s t s between the r e l a t i v e a g g r e s - s i v e n e s s of t h e s e two a l k a l i s u s u a l l y p r e s e n t in p o r t l a n d c e m e n t . T h i s con- f l i c t s with r e s u l t s of a l k a l i - e x p a n s i v e r o c k c y l i n d e r s i m m e r s e d in a l k a l i s , w h e r e t h e NaOH w a s m u c h m o r e a g g r e s s i v e than t h e KOH.
SAME AS FIGURE 1 2 I 0 I I I I I I I I I I I I I 0 2 0 4 0 6 0 8 0 1 0 0 1 2 0 1 4 0 TEST A G G R E G A T E P R E S E N T , PER C E N T FIG. 2 V a r i a t i o n of p e r c e n t a g e l i n e a r e x p a n s i o n with t h e amount of v a r i o u s a g g r e g a t e s p r e s e n t f o r 48 -week-old m o r t a r b a r s m o i s t c u r e d in s e a l e d c o n t a i n e r s a t 1 0 0 ° F ( 3 8 ° C ) . (Alkalinity of c e m e n t p a s t e 1 . 0 0 % a s Na 0). 2
V o l . 3 , No. 3 A L K A L I AGGREGATE REACTION, LABORATORY, EXPANSION
T A B L E I11
EXPANSIONS O F MORTAR BARS MADE WITH ADDED NaOH OR KOH. l @ O ° F ( 3 8 ° C ) IN S E A L E D CONTAINERS.
S E L E C T I O N O F 6 A L K A L I - E X P A N S I V E AGGREGATES. L . A . C e m e n t = 0. 14% N a 2 0
+
0. 20% K 2 0 c a l c u l a t e d a s N a 2 0 H . A. C e m e n t = 0. 2.7% N a L O+
0. 61% K 2 0 c a l c u l a t e d a s N a 2 0 B e a c h s a n d A g - g r e - g a t e C a l c a r e o u s a r g i l l i t e Equiv. N a 2 0 Yo + F e l d s p a t h i c q u a r t z i t e G r e y w a c k e P h y l l i t e G r e y w a c k e C o n c r e t e P r i s m s P e r c e n t l i n e a r e x p a n s i o n a t 48 w e e k s C o n c r e t e Low A l k a l i c=-! P r i s m s 96 w e e k s a t T h e c o ~ l c r e t e m i x w a s of n o m i n a l 3500 p s i d e s i g n a n d t y p i c a l of job m i x e s e x c e p t t h a t n o c h e m i c a l a d m i x t u r e w a s u s e d . F u r t h e r d e s c r i p t i o n i s g i v e n i n t h e f i r s t p a p e r ( 1 ) w h i c h a l s o g i v e s c h a r a c t e r i s t i c e x p a n s i o i lc u r v e s f o r t h e 3- by 3- by 11-inch prisms used (75- by 75- by 275-mm).
1
0 . 3 4+
KOH/
t o 0 . 7 1.
0221
.
033 I.
020.
041 I i 0 2 2/
.
043.
02.0.
029.
022.
032 1.
025.
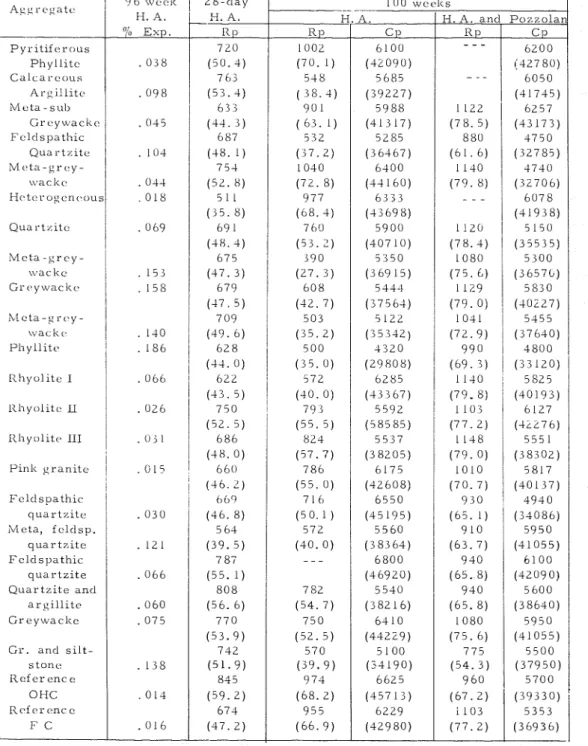
027 S o m e r e l a t i o n s h i p s b e t w e e n e x p a n s i o n of c o n c r e t e , m o d u l u s of r u p t u r e , R p , (ASTM C 2 9 3 - 6 8 ) , a n d t h e m o d i f i e d c u b e s t r e n g t h , C p , (ASTM C 11 6 - 6 8 ) a f t e r n e a r l y two y e a r s m o i s t c u r i n g a t 1 0 0 ° F ( 3 8 " C ) , a r e s h o w n i n T a b l e IV. I n n e a r l y a l l c a s e s when e x p a n s i o n b e c o m e s e x c e s s i v e t h e m o d u - l u s d r o p s f r o m i t s 2 8 - d a y v a l u e . When a p o z z o l a n i s s u b s t i t u t e d f o r 2 5 p e r - c e n t of t h e c e m e n t , b y w e i g h t , a n d s t i l l u s i n g a h i g h a l k a l i c e m e n t , t h e m o d u l u s g e n e r a l l y c o n t i n u e s t o i n c r e a s e . T h i s i s e x p e c t e d b e c a u s e of t h e e f f e c t of t h e p o z z o l a n i n d e c r e a s i n g e x p a n s i o n ( T a b l e V). C o m p r e s s i v e s t r e n g t h s a t 100 w e e k s ( a t 1 0 0 ° F , ( 3 8 ° C ) ) d i d not s h o w a s good c o r r e l a t i o n w i t h t h e e x p a n s i o n . t KOH t o 1 . 0 0.
055.
070 . 0 7 5 0 3 6 . 0 3 8.
042 t NaOH t o 0 . 7 1.
038.
068.
042 . 0 3 0 . 0 2 4.
028 H . A . t H . A .+
N ~ O H I NaOH t NaOH.
052.
0 9 4.
063.
053 . 0 3 9.
0 46I
.
068.
077.
088 . 0 5 1.
075.
069.
098.
1 0 4.
157.
186 . 0 7 5V o l . 3 , N o . 3 239
A L K A L I AGGREGATE REACTION, LABORATORY, EXPANSION
Table IV E x p a n s i o n , Moclulus of R u p t u r c ( l t p ) , ancl C o ~ n p r c s s i v c S t r e n g t h ( C p ) of 3 x 3 x I 1 i n . ( 7 5 s 7 5 s 2 7 5 l n m ) C o n c r e t e P r i s r r ~ s C u r e d a t 3 8 ° C f o r N e a r l y T w o Y e a r s . l t p : A S T M C 2 0 3 - 6 8 ; C p A S T M C 1 1 6 - 6 8 . H. A. = H i g h Allcali C e m e n t . l t p i n p s i ( a n d k g f / c m 2 ) a n d C p i n p s i ( a n d k ~ / r n ~ ) P h y l l i t c C a l c a r e o u s Argillitcs M e t a - s u b G r c ~ y w a c i i c F c l c l s p a t h i c Quartzite Mrta-grey- \vat lees H c t c r o g c n c s o u s I P i n k g r a n i t e F c l d s p a t h i c
i
q u a r t z i t e M e t a , f c l d s p . q u a r t z i t e F c l c l s p a t h i c q u a r t z i t e j Q ~ l a r t z i t e a n d a r g i l l i t c G r e y w a c k e G r . a n d s i l t - s t o n c R e f c r c n c c O H C I\ s H. A. ancl P o z ~ o l a I 427 8 0 ) ( 4 1 7 4 5 ) I 1 2 2 6 2 5 7 ( 7 8 . 5 ) ( 4 3 1 7 3 ) 8 80 4 7 5 0 ( 0 1 6 ) ( 3 2 7 8 5 ) 1 1 4 0 4 7 4 0 ( 7 9 . 8 ) ( 3 ~ 7 0 6 ) - . - 6 0 7 8 ( 4 1 9 3 8 ) 1 1 2 0/
5 1 5 0 ( 7 8 . 4 )1
( 3 5 5 3 5 ) 5 3 0 0 5 8 3 0 1 0 4 1 ( 3 7 6 4 0 ) ( 7 L ' 9 )1
4 8 0 0 9 9 0 ( 6 9 . 3 )/
( 3 3 1 2 0 ) 1 1 4 0,
5 8 2 5 ( 7 9 - 8 );
( 4 0 1 9 3 ) 1 1 0 7 1 6 1 2 7 ( )1
( 4 ~ ~ 7 6 ) 5 5 5 1 ( 7 9 . 0 )/
( 3 8 3 0 2 ) 1 0 1 0/
5 8 1 7 ( 7 0 . 7 )/
( 4 0 1 3 7 ) 9 3 0 ' 4 9 4 0 ( 6 5 . I )/
( 3 4 0 8 6 ) 9 1 0 5 9 5 0 ( 6 3 . 7 ) ( 4 1 0 5 5 ) 9 4 0 61 0 0 ( 6 5 . 8 ) ( 4 2 0 9 0 ) 9 4 0 5 6 0 0 ( 6 5 . 8 ) ( 3 8 6 4 0 ) 1 0 8 0 5 9 5 0 ( 7 5 . 6 ) ( 4 1 0 5 5 ) 7 7 5 5 5 0 0 ( 5 4 . 3 ) ( 3 7 9 5 0 ) 9 6 0 5 7 0 0 ( 6 7 . 2 ) ( 3 9 3 3 0 ) 1 0 3 5 3 5 3 ( 7 7 . 2 ) ( 3 6 9 3 6 )V o l . 3 , N o . 3
A L K A L I AGGREGATE REACTION, LABORATORY, EXPANSION
T A B L E V P e r c e n t a g e L i n e a r E x p a n s i o n of C o n c r e t e P r i s m s a f t e r 9 6 W e e k s M o i s t C u r - ing i n S e a l e d C o n t a i n e r s a t 1 0 0 ° F ( 3 8 ° C ) . T h e E f f e c t of P a r t i a l S u b s t i t u t i o n of C e m e n t by P o z z o l a n o r F l y A s h A l k a l i n i t y of H. A. c e m e n t 1. 007'0 ( a s Na 0) e x c e p t w h e r e e x t r a A l k a l i a d - d e d , a s shown, t o c o m p e n s a t e f o r recfuced c e m e n t c o n t e n t . L . A . C e m e n t 0.347'0 ( a s Na 0 ) 2 D e s c r i p t i o n C a l c a r e o u s A r g i l l i t e F e l d s p a t h i c Q u a r t z i t e M e t a - G r e y w a c k e M e t a - G r e y w a c k e G r e y w a c k e P h y l l i t e B a n d e d , G l a s s y R h y o l i t e F e l d s p a t h i c Q u a r t z i t e M e t a - F e l d s p a t h i c Q u a r t z i t e Q u a r t z i t e a n d A r g i l l i t e P i n k G r a p h i c G r a n i t e S i l i c e o u s B a l l a s t C e m e n t
1
-- -- - T a b l e V g i v e s m o r e d e t a i l e d r e s u l t s of t h e e f f e c t of p o z z o l a n a n d f l y a s h i n s u p p r e s s i n g e x p a n s i o n of c o n c r e t e p r i s m s m a d e w i t h a l k a l i - e x p a n s i v e a g g r e g a t e a n d h i g h a l k a l i c e m e n t . T h e p o z z o l a n i s a c a l c i n e d v o l c a n i c tuff w i t h a good p e r f o r m a n c e r e c o r d . D e t r o i t - E d i s o n f l y a s h w a s a l s o u s e d . In o r d e r t h a t t h e e f f e c t of t h e m i n e r a l a d m i x t u r e s h o u l d not be a t t r i b u t e d m e r e l y t o t h e r e d u c t i o n i n t o t a l a l k a l i r e s u l t i n g f r o m d e c r e a s e i n c e m e n t c o n t e n t , a c o m p a r a t i v e s e r i e s of t e s t s w a s c a r r i e d out i n w h i c hVol. 3 ,
No.
3ALKALI AGGREGATE
R E A C T I O N , LABORATORY,
EXPANSIONt o t a l a l k a l i n i t y w a s m a i n t a i n e d a t t h e high a l k a l i c e m e n t l e v e l . E v e n i n t h i s c a s e , a s shown in T a b l e V , t h e m i n e r a l a d m i x t u r e s w e r e both v e r y effective i n r e d u c i n g e x p a n s i o n t o v a l u e s c o n s i d e r e d a c c e p t a b l e . Use of a pozzolan o r f l y a s h would r e q u i r e c o n s i d e r a t i o n of d i s a d v a n t a g e s a s w e l l a s advantage s
.
Rock C v l i n d e r s An a d a p t a t i o n of ASTM C586 w a s u s e d in d e t e r m i n i n g a l k a l i - e x p a n s i v i t y by i m m e r s i n g r o c k s a m p l e s in a l k a l i solution, (NaOH u n l e s s o t h e r w i s e s t a t e d ) . C y l i n d e r s w e r e c o r e d m e a s u r i n g 0. 375 in. ( 9 . 6 m m ) in diarn. by 1 . 3 8+
0.02 in. ( 3 5 + 5 m m ) long. T h e y w e r e d r i l l e d f r o m t h r e e m u t u a l l y orthogonal f a c e s of e a c h r o c k s a m p l e e x c e p t i n a few c a s e s w h e r e t h i s w a s not p o s s i b l e . A f t e r grinding t h e e n d f a c e s p a r a l l e l , t h e c y l i n d e r s w e r e c l e a n e d in o r g a n i c solvent and t h e n d r i e d t o constant weight. S e v e r a l s a t u r a t i o n t e c h n i q u e s w e r e u s e d d u r i n g t h i s t e s t s e r i e s . In s o m e s t u d i e s t h e y w e r e p r e s o a k e d i n w a t e r . In a l l t e s t s reference s p e c i m e n s w e r e continuously i m m e r s e d in w a t e r a t t h e r e q u i s i t e c u r i n g t e m p e r a t u r e . T h e expansioii due t o t h e w a t e r i m m e r s i o n w a s s u b t r a c t e d f r o m t h e r e s u l t s of a l k a l i i m m e r s i o n and t h e c o r r e c t e d value r e p o r t e d . The s p e c i a l c o m p a r - a t o r u s e d t o m e a s u r e length c h a n g e s o v e r long p e r i o d s of t i m e w a s s e l e c t e d t o e l i m i n a t e w e a r on t h e c y l i n d e r , e n s u r e a constant m e a s u r i n g p r e s s u r e , and p e r m i t r a p i d m e a s u r e m e n t t o r e d u c e s h r i n k a g e e f f e c t s . FIG. 3 T i m e - l i n e a r e x p a n s i o n r e l a t i o n s h i p s of c y l i n d e r s t a k e n f r o m a r g i l l i t e , showing e x p a n s i o n i n 3 m u t u a l l y orthogonal d i r e c t i o n s , A, B and C. C u r e d in 2. 67 M NaOH a t 1 0 0 ° F ( 3 8 ° C ) T E S T D U R A T I O N , W E E K S 0 . 2 0 + z w 0 . 1 6 U ai w a.
0 . 1 2 Z9
I I I I I I I I I I I I- -i
- MEAN - - V17
VOLUMETRIC Z2
0 . 0 8/"
7
X w ai - - - 0 I I I I I I I I I I I I I 0 8 16 24 32 40 48 56A L K A L I AGGREGATE REACTION, LABORATORY, EXPANSION V o l . 3 , No. 3
T h e a n i s o t r o p y exhibited b y m o s t of t h e r o c k s i s d e m o n s t r a t e d i n F i g u r e 3 f o r one of the a r g i l l i t e s s t u d i e d . A f o r m u l a w a s u s e d t o obtain f r o m t h e s e t h r e e length change r e s u l t s a p e r c e n t m e a n v o l u m e t r i c e x p a n - sion which would, f o r a given s a m p l e , a l w a y s be the s a m e r e g a r d l e s s of the r e l a t i v e i n c l i n a t i o n s of c o r e s t o t h e bedding plane. Using t h i s m e t h o d a n d a d e r i v e d c r i t i c a l v o l u m e t r i c e x p a n s i o n of 0.056 p e r c e n t , a good c o r r e l a - tion w a s obtained between t h e r o c k c y l i n d e r t e s t s and t h o s e of t h e m o r t a r b a r s and c o n c r e t e p r i s m s in distinguishing r o c k s t h a t expanded e x c e s s i v e l y i n t h e p r e s e n c e of alkali.
T y p i c a l v o l u m e t r i c e x p a n s i o n r e s u l t s on r o c k c y l i n d e r s i m m e r s e d in 1 . 0 m o l a r NaOH a t 7 3 ° F ( 2 3 ° C ) a r e p r e s e n t e d i n T a b l e VI. T h e r o c k t y p e s
shown by t h e m o r t a r b a r and concrete prism t e s t s t o be alkali-expansive were T A B L E VI
T Y P I C A L VOLUMETRIC EXPANSION WITH TIME O F MAINLY
ALKALI-EXPANSIVE ROCK CYLINDERS IN 1. 0 MOLAR NaOH
AT 7 3 ° F ( 2 3 ° C ) R o c k T y p e M e t a - g r e y w a c k e M e t a - g r e y w a c k e Q u a r t z i t e g r e y w a c k e Q u a r t z i t e g r e l w a c k e C a l c a r e o u s a r g i l l i t e C a l c a r e o u s a r g i l l i t e P h y l l i t e P y r i t i f e r o u s p h y l l i t e F e l d s p a t h i c q u a r t z i t e F e l d s p a t h i c q u a r t z i t e M i c a c e o u s q u a r t z i t e M i c a c e o u s s c h i s t C h a l c e d o n y ( R i v e r s d a l e ) A g a t e ( B r a z i l ) F l i n t ( E n g l a n d ) P e r c e n t E x p a n s i o n a t W e e k s S h o w n 4
.
002.
0 0 4.
0 0 2.
0 0 2 . 0 1 1 . 0 0 1 . 0 3 6 . 0 0 4.
0 0 5-.
0 0 6 . 0 0 4 . 0 0 1 . 0 1 3 . 0 3 6.
007 7 2.
1 0 0.
0 9 5.
0 9 6 . 0 2 7 . 0 6 0 . 1 5 9 . 0 4 4.
0 6 2 . 0 2 4 . 0 6 4 1 L . 0 0 9.
007 . 0 0 5.
0 0 7 . 0 1 6 . 0 1 6 . 0 3 7.
0 0 8-.
0 0 1-.
0 0 4 . O O O . 0 0 2.
0 0 3.
0 4 3 . 0 0 2 9 6.
1 3 8.
1 6 2.
1 5 9.
0 5 8 . 0 6 7 . 2 3 9.
0 8 8 . 0 8 1 . 0 4 6 1 3 8 48 i . 0 5 9.
0 4 2 . 0 4 7 . 0 1 1.
051.
1 2 8 . l o 4 . 0 2 2 . 0 3 6.
0 0 8 . 0 1 4 . 0 5 1-.
0 4 3-.
0 3 6 . 0 3 5 1 2 0.
1 6 1 . 2 1 1 . 2 2 2.
07 3 . 0 7 4.
286 . 1 1 5.
070 . 0 5 9 . 2 0 0.
1 4 71
:
2 5 51
.
303 D i s i n t e g r a t i n g D i s i n t e g r a t i n g-.
237 . 0 2 2-.
0 1 3V o l . 3, N o . 3 243 A L K A L I AGGREGATE REACTION, LABORATORY, EXPANSION
g e n e r a l l y t h o s e t h a t r e a c h e d t h e c r i t i c a l v o l u m e t r i c e x p a n s i o n i n t h e r o c k c y l i n d e r t e s t . S i m i l a r good a g r e e m e n t w a s found f o r n o n - e x p a n s i v e r o c k s . I t i s of i n t e r e s t t h a t s o m e rock t y p e s known t o be a l k a l i - s i l i c a r e a c t i v e a p p e a r e d t o c o n t r a c t when s u b j e c t e d t o t h i s t e s t , p r o b a b l y t h r o u g h p a r t i a l d i s s o l u t i o n . F i g u r e 4 s h o w s t y p i c a l c u r v e s i n which r a t e of e x p a n s i o n of a phyllite i n c r e a s e s with c o n c e n t r a t i o n of NaOH. In F i g u r e 5 i s shown t h e i n c r e a s e i n
r a t e FIG. 4 T h e e f f e c t of NaOH c o n c e n t r a t i o n on the t i m e - m e a n v o l u m e t r i c expansion r e l a t i o n s h i p f o r phyllite a t 1 0 0 ° F ( 3 8 ° C ) . FIG. 5 T h e effect of t e m p e r a t u r e on t h e t i m e - m e a n v o l u m e t r i c expansion r e l a t i o n s h i p f o r g r eywacke c u r e d in 2. 67 M NaOH of expansion of a g r e y w a c k e Z 0 8 1 6 2 4 3 2 4 0 4 8 5 6 6 4 7 2 8 0 T E S T D U R A T I O N , W E E K S T E S T D U R A T I O N , W E E K S with i n c r e a s e in t e m p e r a t u r e . T h e s e r e s u l t s a r e in a g r e e m e n t with t h o s e f r o m m o r t a r b a r and c o n c r e t e p r i s m t e s t s . I t was found in s e p a r a t e s e r i e s of e x p e r i m e n t s t h a t s m a l l a x i a l l o a d s of 2
t h e o r d e r of 11 p s i (76 k N / m ) r e d u c e d t h e expansion r a t e . KOH and LiOH w e r e found to p r o d u c e s m a l l e x p a n s i o n s r e l a t i v e t o t h o s e of NaOH.
V o l . 3 , N o . 3 A L K A L I AGGREGATE R E A C T I O N , LABORATORY, E X P A N S I O N
s i o n i n v a r i o u s c o n c e n t r a t i o n s of NaOH, t h e s a m p l e s w e r e t r a n s f e r r e d to d i s t i l l e d w a t e r . T h e e x p a n s i o n s continued at o n l y a s l i g h t l y s l o w e r r a t e .
A s p e c i a l r o c k c y l i n d e r study w a s c a r r i e d out on a n u m b e r of Nova S c o t i a r o c k t y p e s to d e t e r m i n e t h e r e l i a b i l i t y of a s e l e c t e d t e s t condition i n r e l a t i o n t o t e s t s with m o r t a r b a r s and c o n c r e t e p r i s m s m a d e with high a l k a l i c e m e n t . In t h i s c a s e t h e c y l i n d e r s w e r e v a c u u m s a t u r a t e d with d i s t i l l e d w a t e r and allowed t o e q u i l i b r a t e f o r 28 d a y s b e f o r e i n i t i a l m e a s u r e m e n t and i m m e r s i o n i n a l k a l i . T h e conditions w e r e 2.67 m o l a r NaOH a t 1 2 5 ° F (52 " C ) f o r 16 w e e k s , u s i n g 0.056 p e r c e n t v o l u m e t r i c
e x p a n s i o n a s the c r i t i c a l value. T h e d e g r e e of a g r e e m e n t with t e s t r e s u l t s f r o m m o r t a r b a r s and c o n c r e t e p r i s m s i n d e t e c t i n g r o c k s t h a t a r e p r o n e t o e x c e s s i v e e x p a n s i o n i s shown i n F i g u r e 6, ( a ) and ( b ) . T h e s e and o t h e r e x p e r i m e n t s showed t h a t t h e r o c k c y l i n d e r t e s t i s a good m e t h o d f o r the i n i t i a l e v a l u a t i o n of a n a g g r e g a t e s o u r c e . - L I N E A R E X P A N S I O N O F M O R T A R B A R S , P E R C E N T FIG. 6 ( a ) C o m p a r i s o n of e x p a n s i o n of 16-week-old r o c k c y l i n d e r s and 72- week-old m o r t a r b a r s (Nova S c o t i a r o c k t y p e s ) . PRISMS W I T H 1.00% C E M E N T A L K A L I (as NapO) L I N E A R E X P A N S I O N O F P R I S M S P E R C E N T FIG. 6 ( b ) C o m p a r i s o n of e x p a n s i o n s of 1 6 - w e e k - o l d r o c k c y l i n d e r s and 9 6 - week-old c o n c r e t e p r i s m s (Nova S c o t i a r o c k t y p e s ) .
V o l . 3, No. 3
ALKALI AGGREGATE REACTION, LABORATORY, EXPANSION
S u m m a r y
Good a g r e e m e n t w a s obtained between t h e v a r i o u s t e s t s with m o r t a r b a r s , c o n c r e t e p r i s m s , and r o c k c y l i n d e r s i n distinguishing potentially a l k a l i - expansive r o c k s f r o m non-expansive r o c k s . T o s o m e d e g r e e t h i s was r e m a r k a b l e c o n s i d e r i n g the h e t e r o g e n e i t y of r o c k t y p e s and m i n e r a l s i n any given s a m p l e . Some c o r r e l a t i o n d a t a and t h e r e c o m m e n d e d t e s t condi- t i o n s a r e outlined i n t h e f i r s t p a p e r (1). F u r t h e r and detailed t e s t d a t a are
t o be found i n t h e d o c t o r a l t h e s i s of t h e f i r s t a u t h o r ( 3 ) .
A s i n o t h e r t y p e s of a l k a l i - a g g r e g a t e r e a c t i o n s which have b e e n studied and r e p o r t e d , t h e m o r t a r b a r and c o n c r e t e p r i s m t e s t s continue t o be good d i r e c t m e t h o d s f o r evaluation. T h e r o c k c y l i n d e r t e s t , h e r e t o f o r e l i m i t e d t o t h e a l k a l i - c a r b o n a t e r o c k r e a c t i o n , h a s been shown t o be applicable t o evaluation of t h e r o c k s i n Nova Scota.
I n t e r p r e t a t i o n of t h e e x p e r i m e n t a l r e s u l t s given i n t h i s p a p e r , and a l s o i n t h e two p r e c e d i n g o n e s , w i l l be d e a l t with i n t h e next and l a s t p a p e r i n t h i s s e r i e s .
R e f e r e n c e s
1. M.A.G. Duncan, E . G . Swenson, J . E . G i l l o t t , and M . R . F o r a n . Cem. and C o n c r . R e s . , Vol. 3, NO. 1, p. 55 (1973).
2. M.A.G. Duncan, J . E . G i l l o t t , and E.G. Swenson. C e m . and C o n c r . R e s . , Vol. 3 , No. 2, p. 1 1 9 (1973).
3. M.A.G. Duncan. " C o r r e l a t i o n of F i e l d and L a b o r a t o r y Evidence of Alkali-Silica R e a c t i v i t y i n Nova Scotia C o n c r e t e f ' , P h . D T h e s i s , Nova Scotia T e c h n i c a l College, 1970, p. 1