Active surfaces: Ferrofluid-impregnated surfaces for active manipulation of droplets
Texte intégral
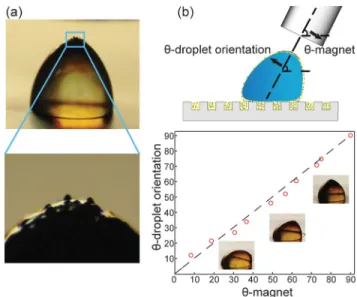
Figure

Documents relatifs
Active correction of the tilt angle of the surface plane with respect to the rotation axis during azimuthal scan.. Kalashnyk,
Our Sea of Nodes representation semantics is not a sequential one: non- ϕ data nodes do not depend on a particular region node, and are evaluated with a denotational semantics.
Based on a recently introduced spatial relation called enlacement, which generalizes the idea of surrounding for arbitrary objects, we propose a fuzzy landscape model that allows
We have developed an underwater mobile robot localization algorithm that allows un- derwater robots to act as mobile sensors in the sensor network by using ranging information..
Il convient dès lors de confirmer l’hypothèse que la diversité des ressources est corrélée avec les différents statuts des connaissances et catégories cognitives
(ii) To non-specialized butchers on the left side (young butchers, selling other types of red meat beside camel meat, illegal slaughter of camel, marketing of carcasses of
81 National Research University Higher School of Economics, Moscow, Russia, associated to 42 82 National University of Science and Technology “MISIS”, Moscow, Russia, associated to
Mauricette Fournier et Annick Stoehr-Monjou, « « Cartographie géo-littéraire et géo-historique de la mobilité aristocratique au V e siècle d’après la correspondance de