See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/244644838
5,5'-Bithiazole and 2,5'-Bithiazole
Article in Acta Crystallographica Section C Crystal Structure Communications · January 1995 DOI: 10.1107/S0108270194002969 CITATION 1 READS 27 4 authors, including:
Some of the authors of this publication are also working on these related projects:
Crystal engenneringView project
Intermolecular interactions in proton transfer compoundsView project Nourredine Benali-Cherif
Académie des Sciences et Technologies d'Algérie
160PUBLICATIONS 489CITATIONS
SEE PROFILE
All content following this page was uploaded by Nourredine Benali-Cherif on 09 November 2016.
76
ORGANIC C O M P O U N D S
Acta Cryst. (1995). C51, 7 6 - 7 75,5'-Bithiazole and 2,5'-Bithiazole
NOURREDINE BENALI-CHERIF AND MARCEL PIERROT Laboratoire de Bio-Inorganique Structurale, Faeultd des Sciences et Techniques de Saint-Jerdme, Case C12, 13397 Marseille CEDEX 20, France CHRISTOPHE BAUDRION AND JEAN PIERRE AUNE ENSSPICAM, Facultd des Sciences et Techniques de Saint-Jerdme, Case C12, 13397 Marseille CEDEX 20, France
(Received 9 August 1993; accepted 16 March 1994)
Abstract
B i t h i a z o l e s are k n o w n to be p o t e n t i a l a n t i b a c t e r i a l , a n t i f u n g a l a n d a n t i - i n f l a m m a t o r y a g e n t s a n d t h e i r c o n f o r m a t i o n s are o f p a r t i c u l a r interest. O f the six possible i s o m e r s o n l y t h r e e s t r u c t u r e s h a v e so far b e e n studied: 2 , 2 ' - b i t h i a z o l e [Bolognesi, C a t e l l i n i , D e s t r i & P o r z i o (1987). Acta Cryst. C43, 1171-1173], 4 , 4 ' - b i t h i a z o l e [ R a t z i m b a z a f y (1987). Th~se de d o c t e u r es sciences, U n i v . A i x - M a r s e i l l e III, F r a n c e ] a n d 2,4"-bithiazole [ B e n a l i - C h e r i f , P i e r r o t , B a u d r i o n
& A u n e (1995). Acta Cryst. C51, 72-75]. As a
c o n t i n u a t i o n o f this series, we describe here the c r y s t a l s t r u c t u r e s o f t w o o t h e r isomers, n a m e l y , 5 , 5 ' - b i t h i a z o l e , C6H4N2S2, a n d 2 , 5 ' - b i t h i a z o l e , C6H4N2S2. In b o t h s t r u c t u r e s the b i t h i a z o l e m o l - ecules are p l a n a r . C o m m e n t A c o m p a r i s o n o f the 5 , 5 ' - b i t h i a z o l e (I) a n d 2,5"- b i t h i a z o l e (II) w i t h k n o w n b i t h i a z o l e s d o e s n o t reveal HI /
,s d
"%<\,
II "c5--c{, II II c_--c,s: II H2 / m / H H4' (I) (II) a n y differences e i t h e r in b o n d d i s t a n c e s or b o n d angles as s h o w n in T a b l e 2. M o r e o v e r , in b o t h f o r m s the t h i a z o l e rings are c o p l a n a r as has been f o u n d in the p r e v i o u s l y d e t e r m i n e d i s o m e r s ( G r a i g , G o o d w y n , O n g g o & R a e , 1988; B o l o g n e s i , C a t e l l i n i , Destri & P o r z i o , 1987).© 1995 International Union of Crystallography Printed in Great Britain - all rights reserved
I
H1 S" N .~C4 ( ~ • ~ . , N " H2 C4' (O) S AH4" ~ , , 5 5 , / \ ~ ' ~ . . ~ - , 'Y
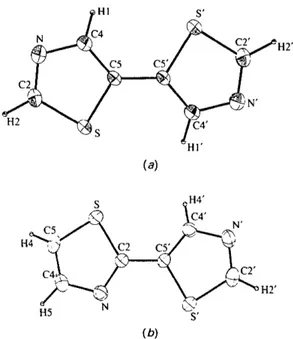
C4~'~ f --~ P" C2' H5 S' (b)Fig. 1. ORTEP plots (Johnson, 1976) of the title compounds with the atomic numbering schemes: (a) (I) and (b) (II). Dis- placement ellipsoids are at the 50% probability level.
E x p e r i m e n t a l
5,5'-Bithiazole (I) was synthesized by the same method used to obtain 2,2'-bithiazole (Graig, Goodwyn. Onggo & Rae, 1988), a homocoupling reaction of 5-bromothiazole using nickel as catalyst. After liquid chromotographic purifica- tion the product was crystallized from an EtOH/H20 solu- tion over 2 weeks. 2.5'-Bithiazole (II) was synthesized by a cross-coupling reaction between 5-bromothiazole and 2- trimethyltinthiazole (Dondoni, Fogagnolo, Medici & Negrini, 1987). It was purified by liquid chromatography and co'stal- lized after 1 year at 277 K in EtOH/H20 solution.
5,5 '-Bithiazole Crystal data
CoH4NzSz Mo Ka radiation
M, = 168.24 A = 0.71069 ~,
Monoclinic Cell parameters from 25
P21/n reflections a = 8.670 (3) A 0 = 14-16 ° b = 3.902 (2) A. /.L = 0.641 mm- c = 10.463 (1) ,~ T = 293 K 3 = 95.32 (2) ° Rectangular V = 352.5 (4) , ~ 0.4 × 0.3 × 0.3 mm Z = 2 Colorless D, = 1.585 Mg m -3 Data collection Enraf-Nonius CAD-4 R,nt = ().024 diffractometer Om~, = 24 ~
Acta Crvstallographica Section C ISSN 0108-2701 ~ 1995
B E N A L I - C H E R I F , P I E R R O T , B A U D R I O N A N D A U N E 7 7 0/20 scans Absorption correction: none 1340 measured reflections 663 independent reflections 655 observed reflections [1 > 3a(l)]
Refinement
Refinement on F R = 0.031wR
= 0.038 S = 0.508 655 reflections 54 parameters All H-atom parametersrefined 2,5 '-Bithiazole
Crystal data
C 6 H 4 N 2 5 2Mr
= 168.24 Orthorhombic P212121 a = 5.770 (3) ,~, b = 5.923 (2) ,~ c = 21.056 (1) ,~ v - - 719.6 (3) ,~3 z = 4 Dx = 1.555 Mg m - 3Data collection
E n r a f - N o n i u s C A D - 4 diffractometer 0/20 scans Absorption correction: none 790 measured reflections 771 independent reflections 712 observed reflections [1 > 3o"(I)1 h = 0 ~ 10 k = - 4 - - ~ 4 1 = - 1 2 ---, 12 2 standard reflections frequency: 60 min intensity variation: < 1% w = 1/cr2(F) (A/Or)max = O.OlApm,~
= 0.259 e ,~-3 Apm,n = - 0 . 2 7 2 e ,~-3 Atomic scattering factorsfrom
International Tables
for X-ray Crystallography
(1974, Vol. IV)
Mo K a radiation A = 0.71069,4,
Cell parameters from 25 reflections 0 = 1 4 - 1 6 ° # = 0.627 m m - I T = 293 K Prism 0.5 x 0.4 × 0.3 m m Yellow O m a x = 24 ° h = 0 ---, 6 k = - 4 ---~ 7 l = - 1 2 - - , 2 5 2 standard reflections frequency: 60 rain intensity variation: < 1% Refinement R e f i n e m e n t o n F R = 0.041
wR
= 0.054 S = 2.3 712 reflections 91 parameters All H-atom parametersrefined
w = I/o'2(F) (A/a)max = 0.02 Apm.x = 0.35 e ,~-3 Apmin = - 0 . 2 9 e ,~-3 Atomic scattering factors
from
International Tables
for X-ray Co, stallography
(1974, Vol. IV)
T a b l e 1.
Fractional atomic coordinates and equivalent
isotropic displacement parameters (~,~-)
Beq = (4/3)Ei~fl3i)ai.a). x V z Beq 5.5 CBithiazole S ().65708 (8) 0.3036 (2) 0.86501 (6) 3.28 (1) N 0.8262 (3) 0.2622 (8) 1.0773 (2) 4.16 (6) C4 0.6848 (3) 0.3941 (9) 1.1031 (3) 3.49 (6) C5 0.5785 (3) 0.4368 (7) 1.0022 (2) 2.37 (5) C2 0.8271 (3) 0.2079 (9) 0.9550 (3) 3.69 (6) 2.5 '-Bithiazole S 1.0429 (2) 0.5702 (2) 0.86101 (4) 4.05 (2) S' 0.4611 (2) 0.1078 (2) 0.88846 (4) 3.85 (2) N 0.6328 (4) 0.5300 (5) 0.8164 (I) 3.44 (5) N' 0.7446 (6) -0.0335 (5) 0.9727 (I) 4.12 (6) C2 0.7754 (5) 0.4423 (5) 0.8581 (1) 2.71 (5) C2' 0.5448 (7) -0.0746 (5) 0.9475 (2) 4.20 (8) C4 0.7376 (8) 0.7107 (6) 0.7861 (2) 3.84 (7) C4' 0.8435 (5) 0.1508 (6) 0.9438 (2) 3.51 (7) C5 0.9544 (7) 0.7551 (5) 0.8038 (2) 4.10 (7) C5' 0.7179 (6) 0.2502 (5) 0.8975 (I) 2.80 (5) T a b l e 2.
Comparison o f bond distances ( A ) and angles
(o)with
2,2 ,_and
4 , 4 ' - b i t h i a z o l e2.2'- 4,4'- 5,5'- 2,5'-
Bithiazole* Bithiazolet Bithiazole Bithiazole~ S--C2 1.717 (2) 1.710 (4) 1.715 (3) 1,716(3), 1.720(4) S---C5 1.706 (2) 1.704 (4) 1.725 (3) 1.707(4), 1.716(3) N---C2 1.3(16 (3) 1.300 (5) 1.297 (4) 1.310(4), 1.292(5) N---C4 1.373 (3) 1.383 (3) 1.380 (4) 1.385(4), 1.374(5) C4---C5 1.345 (4) 1.360(5) 1.364(4) 1.331(6), 1.349(4) C--C' 1.449 (3) 1.469 (7) 1.445 (3) 1.448 (4) S---C2--N 115.2(2) 115.5(3) 1 1 5 . ( 1 ( 3 ) 114.4(2), 115.5(3) S---C5--C4 109.9 (2) 109.8 (3) 108.7 (2) 110.6(3), 109.3(2) C2--N---C4 109.1 (2) 109.8(3) 110.0(2) 109.9(3), 109.8(3) N--C4---C5 116.4(2) 115.3 (3) 116,6(2) 115.7(3), 116.3(3) C2--S---C5 89.0 ( I ) 89.6 (2) 89.7 ( 1 ) 89.3(2), 89.2(2)
* Bolognesi, Catellini, Destri & Porzio (1987). t Ratzimbazafy (1987).
:~ 2,5 t-Bithiazole has no molecular symmetry. The two values are for corresponding but non-symmetrical distances and angles.
Absences for the
P2t/n
cell arehOl, h + 1
odd, and 0k0, k odd, and for the P2~2~2m cell are h00, h odd, 0k0, k odd and 001, l odd. All non-H atoms were found by direct methods (Frenz, 1978) and the structures were refined successfully with a full- matrix least-squares procedure using anisotropic displacement parameters for the non-H atoms. For both c o m p o u n d s , H atoms were located on a difference Fourier synthesis and included in the refinement. The weighting scheme used was derived fromor(I,,)
= [cr-(lo) + (0.041o)21 I/2.Lists of structure factors, anisotropic displacement parameters, H- atom coordinates and complete geometry have been deposited with the IUCr (Reference: PAl078). Copies may be obtained through The Managing Editor, International Union of Crystallography, 5 Abbey Square, Chester CHI 2HU, England.
...
References
Benali-Cherif, N., Pierrot, M., B a u d r i o n , C. & A u n e , J. P. (1995). Acta Cryst. C51, 72-75.
Bolognesi, A., Catellini, M., Destri, S. & Porzio, W. (1987). Acta Cryst. C43, 1171-1173.
D o n d o n i , A., F o g a g n o l o , M., Medici, A. & Negrini, E. (1987). Synthesis, 185-186.
Frenz, B. A. (1978). The Enraf-Nonius CAD-4 SDP - a Real-Time System for Concurrent X-ray Data Collection and Crystal Struc- ture Solution. Computing in Crystallography, edited by H. Schenk, R. Olthof-Hazekamp, H. van Koningsveld & G. C. Bassi, pp. 64-71. Delft Univ. Press.
Graig, D. C., Goodwyn, H. A., Onggo, D. & Rae, A. D. (1988).
Aust. J. Chem.
41, 1625-1628.Johnson, C. K. (1965).
ORTEP.
Report ORNL-3794. Oak Ridge National Laboratory, Tennessee, USA.Ratzimbazafy, A. (1987). Th+se de docteur es sciences, Univ. Aix-Marseille III, France.
View publication stats View publication stats