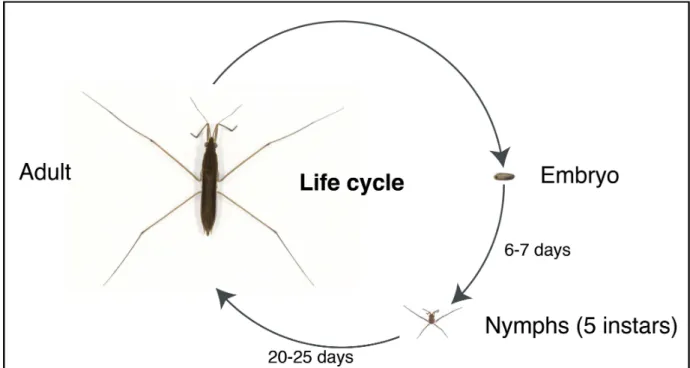
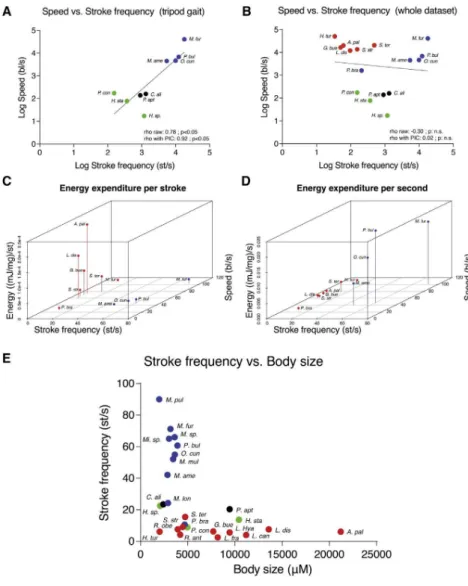
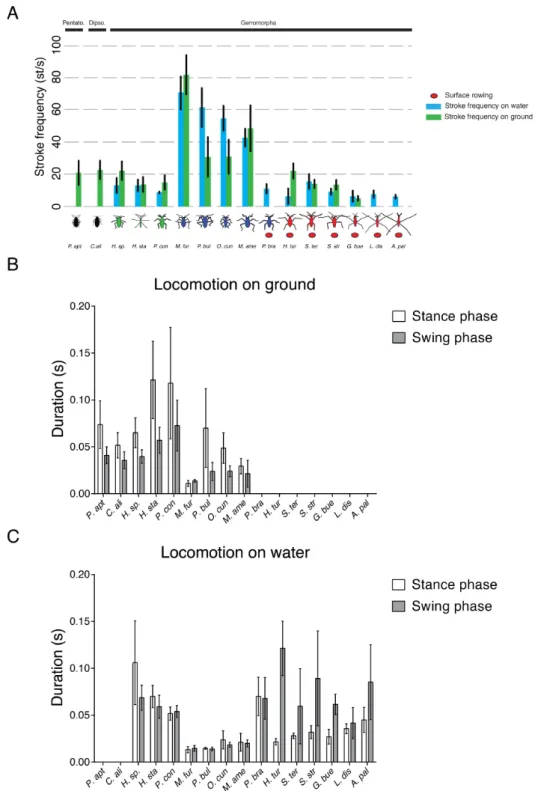
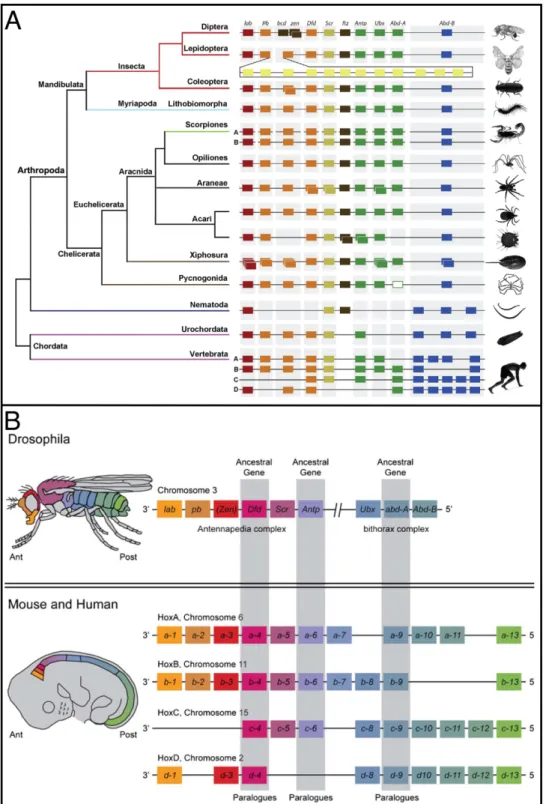
Developmental mechanisms of adaptive phenotypes and associated ecological relevance in the semiaquatic bugs
Texte intégral
Figure
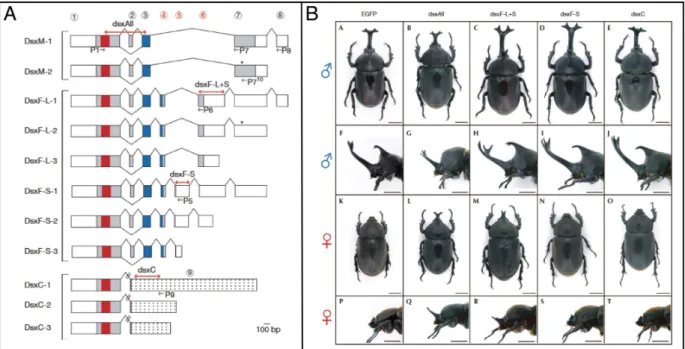
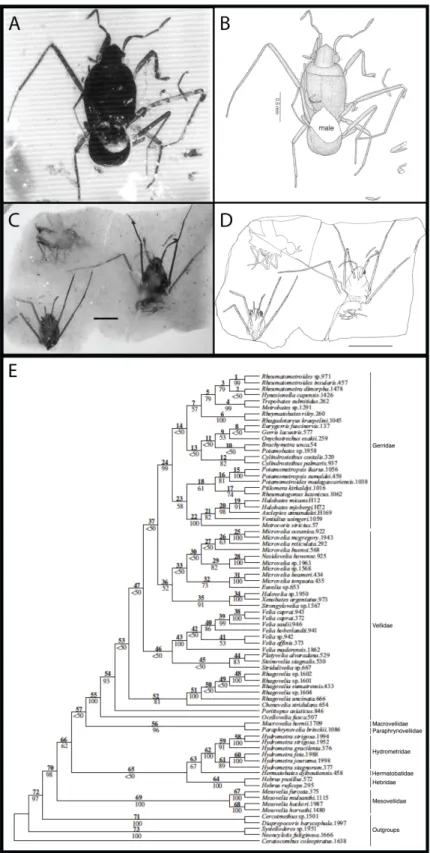
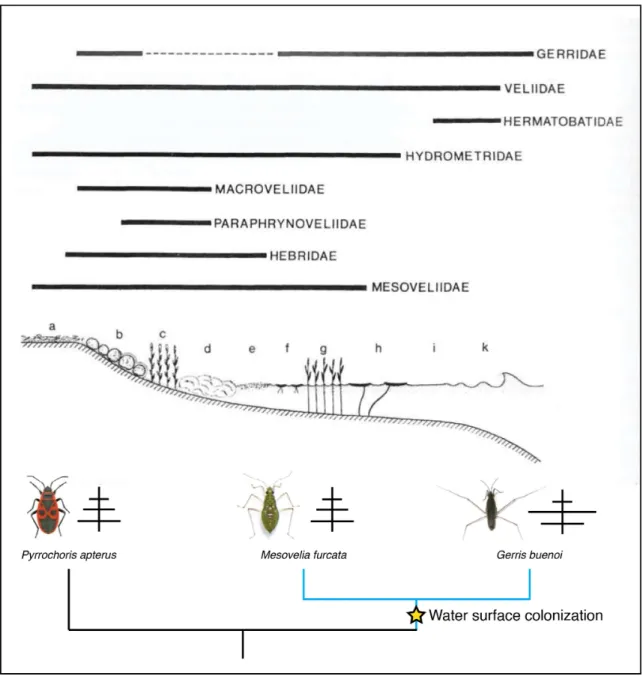
Documents relatifs
Two types of software were used to analyse the microarray data; Genespring was used for statistical analysis and visualisation of transcriptomic data and Cytoscape was used
setulose setae on idiosoma and legs; ctenidial or palmate (not unambiguously detectable) setae 1a, b; taeniform setae on coxae II and III; leg tarsi, with thin, elongate empodium and
In addition to water vapor, a small contribution to downward LWR at the surface is attributed to midlevel clouds (in agreement with Bouniol et al. At the interannual time scale,
Les projets présentés précédemment ont permis d’apporter des réponses aux enjeux de l’uti- lisation des GNSS dans le domaine ferroviaire. Dans les architectures et les
Dans ce contexte, l’oxyde de graphène réduit (rGO) est un très bon candidat comme support pour ce type de matériau catalytique en raison de sa stabilité vis-à-vis
Hierarchical clustering of gene family and functional domain assignments, and phylogenetic analyses revealed lineage specific losses of carbohydrate-active enzymes (CAZymes)
(Acari: Tarsonemidae), is described based on phoretic females collected under the hemielytra of the aradid bug Aradus betulae (Linnaeus, 1758) (Heteroptera:.. Aradidae) in
The insects studied were mainly observed on the waterfront of the mangrove between the roots of Rhizophora mangle or mangrove hinterland, in the water holes between