Temperature, geometry, and bifurcations in the numerical modeling of the cardiac mechano-electric feedback.
Texte intégral
Figure
Documents relatifs
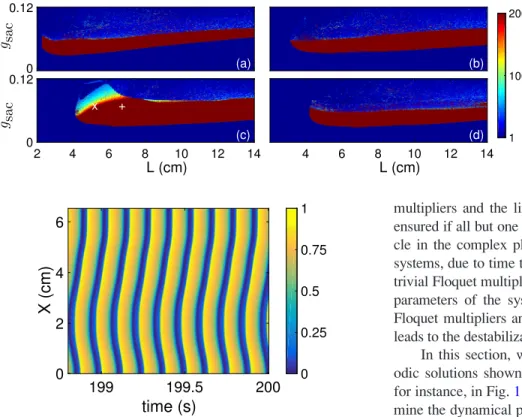
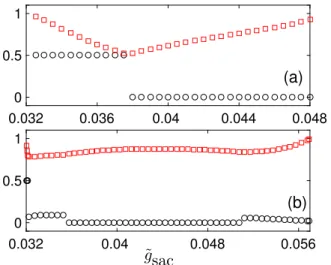
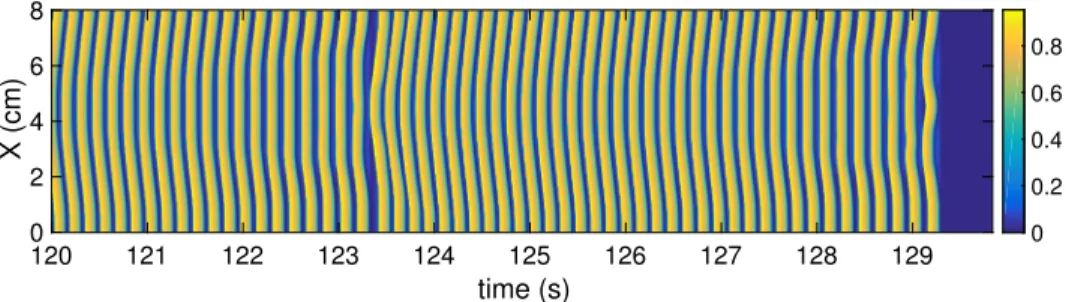
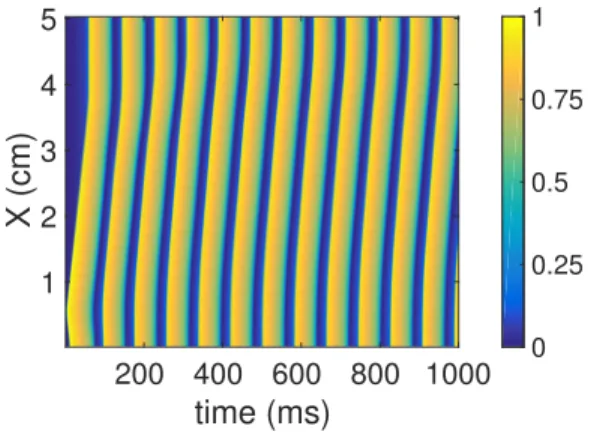
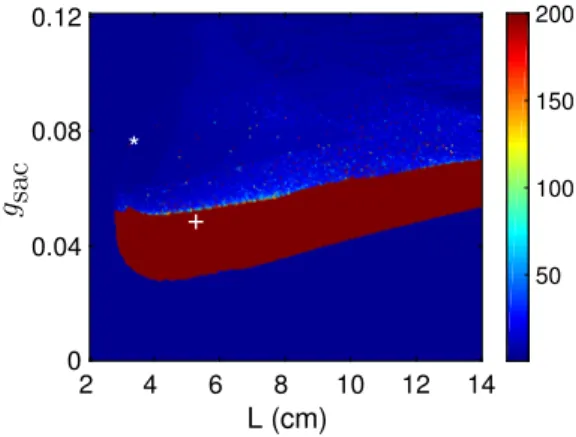
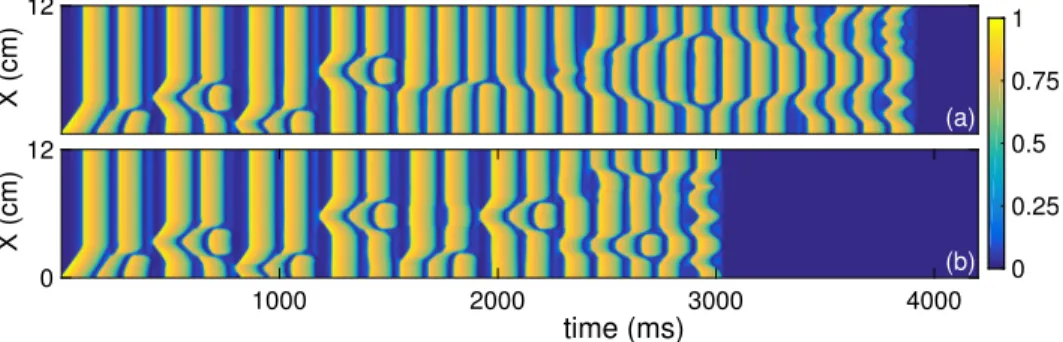
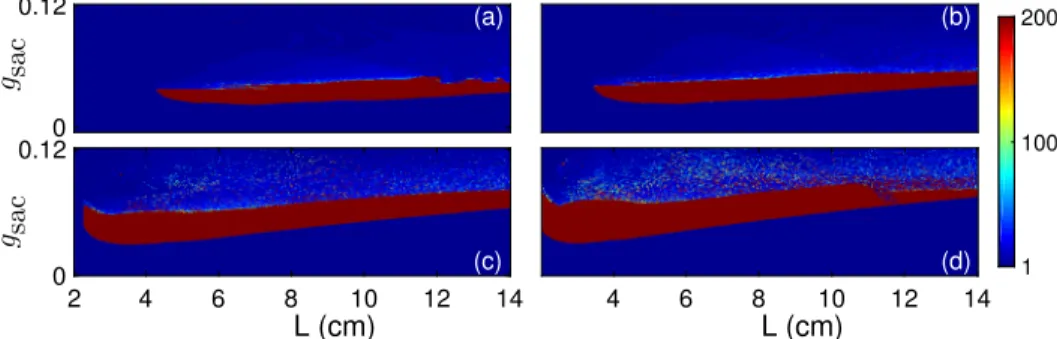
This behaviour is completely reversible in the simulated system (Figure 2). This parameter represents the extent of extracellular stimulation by an agonist. The
First introduced by Faddeev and Kashaev [7, 9], the quantum dilogarithm G b (x) and its variants S b (x) and g b (x) play a crucial role in the study of positive representations
A second scheme is associated with a decentered shock-capturing–type space discretization: the II scheme for the viscous linearized Euler–Poisson (LVEP) system (see section 3.3)..
(A few meters below the surface should be enough.) This is used as a boundary value to determine the temperature, independent of time, in the interior of the
Particularly, the effect of the initial wet curing time on the mechanical behavior such as the compressive strength and the durability of the SCCs (acid and sulfate attack) as well
A diagram of analytic spaces is an object of Dia(AnSpc) where AnSpc is the category of analytic spaces.. Cohomological motives and Artin motives. all finite X-schemes U ). an
Aware of the grave consequences of substance abuse, the United Nations system, including the World Health Organization, has been deeply involved in many aspects of prevention,
If I could have raised my hand despite the fact that determinism is true and I did not raise it, then indeed it is true both in the weak sense and in the strong sense that I