O
pen
A
rchive
T
OULOUSE
A
rchive
O
uverte (
OATAO
)
OATAO is an open access repository that collects the work of Toulouse researchers and
makes it freely available over the web where possible.
This is an author-deposited version published in :
http://oatao.univ-toulouse.fr/
Eprints ID : 18213
To link to this article
: DOI: 10.1016/j.colsurfb.2016.12.007
URL :
http://dx.doi.org/10.1016/j.colsurfb.2016.12.007
To cite this version
: Conzatti, Guillaume and Cavalier, Sandrine and
Combes, Christèle and Torrisani, J and Carrerre, N and Tourrette,
Audrey PNIPAM grafted surfaces through ATRP and RAFT
polymerization: Chemistry and bioadhesion. (2017) Colloids and
Surfaces B Biointerfaces, vol. 151. pp. 143-155. ISSN 0927-7765
Any correspondence concerning this service should be sent to the repository
PNIPAM
grafted
surfaces
through
ATRP
and
RAFT
polymerization:
Chemistry
and
bioadhesion
G.
Conzatti
a,
S.
Cavalie
a,
C.
Combes
b,
J.
Torrisani
c,
N.
Carrere
c,d,
A.
Tourrette
a,∗aCIRIMAT,UniversityofToulouse,CNRS,INPT,UPS,UniversitéPaulSabatier,FacultédePharmacie,35ChemindesMaraichers,31062Toulousecedex9, France
bCIRIMAT,UniversitédeToulouse,CNRS,INPT,UPS,ENSIACET,4alléeEmileMonso,CS44362,31030Toulousecedex4,France
cUniversitéFédéraleMidi-Pyrénées,UniversitédeToulouseIIIPaulSabatier,InsermU1037,CRCTdeToulouse,2avenueHubertCurienCS5371731037 ToulouseCedex1,France
dGastrointestinalSurgeryDepartment,PurpanHospital–CHUdeToulouse,PlaceduDrBaylac,31059Toulouse,France
Keywords:
Surfacefunctionalization
Reversibleadditionfragmentationchain transferpolymerization
Atomtransferradicalpolymerization Poly(N-isopropylacrylamide) Bioadhesion
a
b
s
t
r
a
c
t
Biomaterialssurfacedesigniscriticalforthecontrolofmaterialsandbiologicalsysteminteractions. Beingregulatedbyalayerofmoleculardimensions,bioadhesioncouldbeeffectivelytailoredbypolymer surfacegrafting.Basically,thissurfacemodificationcanbecontrolledbyradicalpolymerization,which isausefultoolforthispurpose.Theaimofthisreviewistoprovideacomprehensiveoverviewofthe roleofsurfacecharacteristicsonbioadhesionproperties.Weplaceaparticularfocusonbiomaterials functionalizedwithabrushsurface,onpresentationofgraftingtechniquesfor“graftingto”and “graft-ingfrom”strategiesandonbrushcharacterizationmethods.Sinceatomtransferradicalpolymerization (ATRP)andreversibleaddition-fragmentationchaintransfer(RAFT)polymerizationarethemost fre-quentlyusedgraftingtechniques,theirmaincharacteristicswillbeexplained.Throughtheexampleof poly(N-isopropylacrylamide)(PNIPAM)whichisawidelyusedpolymerallowingtuneablecelladhesion, smartsurfacesinvolvingPNIPAMwillbepresentedwiththeirmainmodernapplications.
Contents
1. Introduction...143
2. Bioadhesion...144
2.1. Bioadhesionmechanisms...144
2.2. Surfacesandbioadhesion...144
3. Surfacemodification ... 147
3.1. Brushstructure ... 147
3.2. Graftingtechniques...147
3.2.1. Atomtransferradicalpolymerization(ATRP)...147
3.2.2. Reversibleaddition-fragmentationchaintransfer(RAFT)radicalpolymerization...148
3.3. Characterizationofbrushes ... 148
4. Smartbio-surfaces:exampleofpoly(N-isopropylacrylamide)thermosensitivesurfaces...150
5. Conclusion...152
Acknowledgement...152
References...152
1. Introduction
Current developments in medicine have led to a need for newbiomaterialsinapplicationsinvolvinginnovativestrategies
∗ Correspondingauthor.
E-mailaddress:audrey.tourrette@univ-tlse3.fr(A.Tourrette).
for disease treatment. These biomaterialshave to fulfilvarious requirementsdependingontheapplicationbutinallcases bio-compatibilityisacrucialpointthathastobeconsideredinearly processingstages.Biocompatibilityisdefinedastheabilitytoact inalivingsystemwithoutanytoxicityorrejection,whether phys-iological or immunological. Although biocompatibility includes “non-toxicity”,thesetwoconceptsstillremaindifferentiated.
Sur-facefunctionalizationofamaterialissometimesnecessarytoavoid stronginflammatoryresponsesandimprovebiocompatibility.
Ontheotherhand,surfacesnotonlyactaspassiveinterfaces betweenthebody(immunesystem,blood,cells)andthe bioma-terialbutalsotakeanactivepartincellspreading,proliferation, differentiationandmigration:allofthesephenomenaare inten-sively linked to surface/cell interactions. Thus, surfaces play a crucialrole in thefunction ofthebiomaterialand canbeused, forexample,asactivatorsofcellsfortissuereconstruction(tissue engineering)[1].Inthisfield,celladhesiontothesurfaceisakey factorthatmustbecarefullyconsidered.Bioadhesionisdefinedas theadhesionbetweenabiologicalentity,e.g.cellsortissues,anda surface.Itisacomplexphenomenonthatinvolvesmany parame-ters.Foralongtime,itwasdifficultforscientiststoclearlyidentify themechanismsunderlyingcelladhesion,buttheyarebecoming increasinglywellunderstood.Theinterestofscientistsfor bioad-hesionis notonly theoreticalbut isalsoessential for thewide spectrumofapplicationsdependingoncell/surfaceaffinity.
Incellsheetengineering,surfacepropertiesareusedtocontrol celladhesion,sothelivingsheetscanbestrippedoffeasily.Itwas shownthatcellsheetsintegratewellintotissues[2]andare promis-ingtoolsfortissuereconstruction.Smartsurfacestunablebetween “on”(adhesive)and“off”(non-adhesive)statesarethenfeasible. Naturally,thestudyofthermosensitivematerialshasbeenwidely reportedintheliterature.Poly(N-isopropylacrylamide)(PNIPAM) isapromisingpolymerasachangeofitshydrophilicinteractions, andindirectlybioadhesivness,takesplacebetweentheroom tem-peratureandbodytemperature.Moreprecisely,itslowercritical solutiontemperature(LCST)occursat32◦C.Whilethemonomeris cytotoxic,thepolymershowsnotoxicityforthevariouscelltypes
[3]andconstitutesagoodexampletohelpunderstandbioadhesion mechanisms.Itscharacteristicsmakethispolymeroneofthemost intensivelystudiedintheliteraturewhethergraftedorcoatedonto materialsurfaces.
Plasmatreatmentis probablythemostcommonprocess for surface modification, both for the introduction of functional groupsorforcoatingthesurfacewithpolymer[4].In the pres-ence of air, oxygenated surfaces will be produced under the plasma, leading to a change in hydrophilicity [5]. This modifi-cation can involve an increase in cell adhesion, as is the case for polystyrene (PS) [6]. In addition, the plasma can etch the surface, enhancingits roughness.This modificationof topogra-phy, as observed in plasma-treated poly(methyl methacrylate) (PMMA),tendstoincreasethecellaffinitytothesesurfaces[7]. Theintroductionof reactivegroups alsopermits compoundsor chemical functionsto begrafted onto thematerial surfaces. In thepresence of oxygen, surfacesactivated viatheintroduction ofhydroxylgroups,canbegraftedwithmonomersorpolymers, such as NIPAM/PNIPAM though N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide (EDC)/N-hydroxysuccinimide(NHS) amide coupling[8].Underargonplasma,radicalswillbeproduced.These radicalscanbeuseddirectlyor,afterexpositiontoair,create per-oxidesthatcaninitiatepolymerization[9].
Whileplasmatreatmentpresentstheadvantageofbeingeasy toperformonmaterials,onlya lowlevel ofsurfacestructuring isobtained.Inaddition,itwasshownthatplasma-deposited PNI-PAMshowsgreatercelladhesionthane-beamcoatedPNIPAM[10]. Theseresultsemphasizedtheimportanceofthemethodusedfor surfacemodification.Controlofthesurfacestructurealsoseems tobeacriticalpoint.Abrushstructure,i.e.aself-assembled close-packedmonolayerofpolymerchains,providesprecisecontrolof thesurfacemorphologicalproperties.Morespecifically,itbecomes possibletocontrolthethicknessandthedensityofthegraftedlayer. Asaresult,thesestructuresarenowextensivelyused,especiallyfor biomedicalapplications,anddisplaygoodperformanceintermsof bioadhesion[11].
Toobtaincontrolledbrushes,awell-definedgraftedpolymeris necessary.Controlledradicalpolymerizationcanbeusedtocontrol polymergraftingusingvarioustechniques.Themostpopularones arering-openingpolymerization,nitroxide-mediated polymeriza-tion,reversibleadditionfragmentationchaintransfer(RAFT)and atomtransferradicalpolymerization(ATRP).Areviewoffunctional polymerbrushesproduced bycontrolledradicalpolymerization waspublishedbyOlivieretal.[12].RAFTpolymerizationandATRP arewidelyusedastheyareversatileandreducepoly-dispersities. Theyrelyontheequilibriumbetweendormantandactivespecies, soaresometimescalled“living”polymerizations.
It is necessary to understand cell adhesion mechanisms to designabiomaterialwithtunablebioadhesionproperties.Theaim ofthisreviewisnottobeexhaustiveonsurfacemodification pro-cessingbuttogiveacomprehensivemultidisciplinaryoverviewof thecell/surfaceadhesionmechanismsaswellasthechemical engi-neeringofsurfaces.Hence,anintroductiontocellularbiologyand bioadhesionwillbemade.Asecondpartwilldealwithbrushesand thechemicalroutesusedtoobtainsuchstructures.Moreprecisely, ATRPandRAFTpolymerizationmethodswillbereviewed along withthewaysinwhichtheyhavebeencharacterized.Finally, engi-neeringoftunablesurfacesusingPNIPAMwillbepresentedwith itsmainandmostrecentapplications.
2. Bioadhesion
2.1. Bioadhesionmechanisms
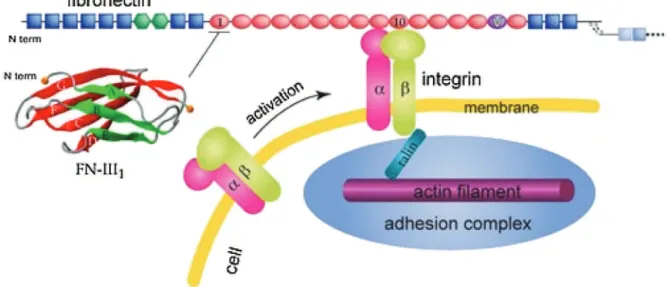
Withasizefrom1to100mm,cellsarecomposedofvarious enti-ties.Somearededicatedtoitsstructure:aphospholipidbilayer, thecellmembrane,maintainstheseparationbetweenthe cyto-sol(internalliquidphase)andtheextra-cellularmatrix(ECM)and thecytoskeleton,composedmainlyofmicrotubulesandactin fil-amentnetworks, control therigidity of the structure [13]. The ECMcompositionvariesdependingonthetissueconcerned.It con-tainsanumberofproteins:fibronectin,collagen,laminin,butalso growthfactorsandalltheproteinsneededfor cellsupportand inter-cellcommunication.Actinfilamentsassembletoconstitute thecytoskeleton.Theyareconnectedtointegrins(transmembrane glycoproteins) throughvinculin andtalin(Fig.1)[14,15].These integrinsspecificallybindtoECMproteinssuchasfibronectin(or vitronectin)througharginylglycylasparticacid(contractionof L-arginine,glycine,andL-asparticacid,abbreviatedRGD)coupling. Theseintegrin/ECM proteininteractions areresponsiblefor cell adhesiontosurfaces(intercellularcohesionisregulatedbyother mechanisms,e.g. cadherin-mediated homotypic junctions).This adhesioniscommonlydividedintothedifferentphasesdescribed in Fig. 2a [16]. The first seconds of contact are characterized by the formation of non-specific interactions. Then, biological interactions occur,including adhesionprotein/fibronectin inter-actions.Thissecondstepleadstoa cascadeofactionsincluding thereorganizationofthecytoskeletonandclusteringofintegrin receptors.Consequently,cellscontracttheircytoskeletonto main-tainamechanicalstateoftension,alsocalledprestress.Later,cells produceECMtoreinforcetheirintegrationandmaintaina propi-tiousenvironment.Itisthusobviousthattheadsorptionofadhesive proteinsisakeypointforcelladhesion.
2.2. Surfacesandbioadhesion
Twomainparameterswilldeterminethebehaviorofimplanted biomaterials:(1)theirbulkproperties,especiallytherigidity,plays aroleinthequalityoftheimplantationintotissues,and(2)their surface propertiescontrol theimmune systemresponse (called immunogenicity),thedestructionofcellintegrity,andthe
bioad-Fig.1.Theadhesionstructureofacellinamatrix.Thematrixislinkedtothecytoskeletonthroughintegrinsandtalins.ReproducedfromRef.[14]withthepermissionof TheRoyalSocietyofChemistry.
Fig.2.(a)Stagesofcelladhesion:interactionsandkinetics.(b)SEMpictureofamyoblastcellonanartificialsurface,2001.Fig.2bisreprintedfromRef.[17],Copyright (2002),withthepermissionofElsevier.
hesion. As cell membranes are anionic, cationic surfaces have to be carefully used since they can damage cells and tissues, whereasnegativelychargedpolymerswillelectrostaticallyrepel cells[18–21].Theimportanceofsurface/cellinteractionshasbeen investigatedformanyyears,andaninterestingdiscussionwas pub-lishedin2001byCastneretal.[17].Inthisaim,syntheticmaterials andbioadhesionhavebeenstudiedandobservedbyscanning elec-tronmicroscopy(SEM,Fig.2b).
Animportantpointistorealizethatinalivingsystemsurfaces arespontaneouslycoveredbyproteins.Itisnowbetterunderstood
thatcelladhesionisbothruledby(i)specific(biological) interac-tions(receptor/ligand)and(ii)thephysico-chemicalpropertiesof thematerial.Hydrophilicity,mechanicalpropertiesand morphol-ogyareamongtheparametersinvolvedinthebioadhesionprocess. AsmentionedabovecelladhesionbetweentissuesandECMis possiblethroughthespecificbindingofintegrinwithfibronectin (Fig.1)[22].In2011,Peietal.observedtheimportanceof spe-cificinteractions(i.e.fibrinogenRGDcoupling)intheprocessof attachmentbetweencellsandtheirsubstrate[23].Theynotedthe number of human foreskin fibroblast(hFF) cells and how they
spread on poly(ethylene glycol) (PEG)brush surfaces (see Sec-tion3.1)graftedontoTiO2andcomparedpre-treatedhFF(blocked integrinbonding-sites)andnon-treatedhFF(freeintegrinbonding sites).
Thenumberofattachedcellsstronglydecreasedwhenthe inte-grinbondingsiteswereblocked.Nevertheless,weaknon-specific interactions occurred when there was a high level of affinity betweenthecellsandthebiomaterial.Thisstudyalsoprovides pre-ciousinformationontheinfluenceofthePEGbrushdensity.PEG isananti-foulingpolymer.Athighgraftingdensity,PEGchainsare collapsedina“brushregime”(L<2Rg,withLthedistancebetween twoneighboringchainsandRgtheradiusofgyrationofpolymer chains),whereasa“mushroomregime”isobservedatlower val-ues(L>2Rg).AgradientofPEGwasusedtoobserveitseffecton proteinadsorptionand celladhesion.For shortexperiments,as thedensityofPEGincreased,astrongdecreaseofthenumberof adheredcellsoccurredwhenthebrushregimewasreachedanda correlationestablishedwiththeproteinadsorptionprofile. Fibro-blastsaturationoccursinthe“brushregime”,meaningthatevena lowamountoffibrinogen(2.2±3.4ng/cm2)issufficienttoactivate adhesion.Heretheimportanceofprotein/cellspecificinteractions isobviouslycrucial.
Amodel involvinga proteinlayerbetweenbiomaterialsand cellswasestablishedintheearly2000’s[1,17,24].Accordingtothis model,cell/matrixadhesiondependsontheabilityofthesurfaceto adsorbproteinswithoutmodifyingtheirnativestructure. Denatu-rationisbroughtaboutbythewaterstructurenearthesurface,i.e. thehydrophilicityofthesuperficiallayeronthematerial.Areview, publishedin2011dealtwiththeconceptofnativeimmune sys-temresponse(cascadesystem)[25].Thearticleemphasizedthe importanceof thenon-denaturationof protein structure atthe material/bodyinterfacetoavoidactivationoftheprimaryimmune system.In1998Volgeretal.alreadyunderlinedtheimportance ofsurfacechemistryintermsofhydrophobic/hydrophilic proper-ties[26].Thisgroupshowedthatwater/surfaceinteractionsare ofinterest.Ascriteriaofhydrophilicity/hydrophobicitytheyused contactanglemeasurements.Astronglyboundwatertoasurface (i.e.hydrophilicsurface)cannotberemoved.Thiswillavoid inter-actionsdirectlybetweena biologicalentity(e.g.a protein), and thesurface,leadingtoloworabsenceofadsorption[27]. More-over,itiswidelyacceptedthatmoderatelyhydrophilicmaterials aresuitableforcelladhesion,withacontactanglearound70–80◦
[28,29]. For example, endothelial cells show good attachment to polycaprolactone-grafted-poly(methyl methacrylate) (PCL-g-PMMA)surfacewitha contactanglearound70◦.Additionally,it isnowacceptedthatsurfaceswhicharetoohydrophobicleadto thedenaturationofproteins[1,28].Moreprecisely,hydrophobic andhydrophilic aminoacids,constitutiveof proteins,rearrange theirorganizationdependingonthesurroundingmediaandthus hydrophobicsurfacescanfavortheexternalisationofhydrophobic moieties,leadingtounfoldedproteins[30].Regardingthis assess-ment, post-treatments (e.g. plasma treatments) are sometimes usedtoincreasesurfacehydrophilicitythroughtheintroduction ofpolarfunctionalgroups.Incontrast,itisknownthatthe interac-tionsofhighlyhydrophilicsurfaceswithECMadhesionproteinsare weak[6].Keselowskyetal.characterizedvariousfunctionalgroups on the criteria of fibronectin adsorption (in increasing order): OH<COOH<CH3<NH2.Ontheotherhand,adhesionofosteoblasts increasesasfollows:CH3<NH2=COOH<OH[31].Thistrend inver-sion can beexplained by the geometrical deformation of ECM proteins,i.e.denaturation.Onbrushsurfaces,theoptimalcontact angledependsnotonlyonthenatureofthematrixbutalsoonthe methodofsurfacemodification.Thiscanbeduetotheinfluenceof chainlengthanddensityofthegraftedpolymeronthe conforma-tionoftheproteinadsorbedonthesurface.Forinstance,ithasbeen pointedoutthatwhenFe2+isusedasinitiatorforthegraft
polymer-izationofPMMAonpoly(L-lacticacid)(PLLA)surfaces,maximum chondrocyteattachmentisobtainedwhenthecontactangleis52◦, whereasUV-initiatedsurfacesareoptimalforacontactangleof76◦
[28].Theauthorssuggestthatthedifferenceofbiological proper-tiesbetweenthetwoPLLAgraftedPMMAcouldbeduenotonlyto surfacewettability(contactangle)butalsotothehigherdensity, uniformityandshorterchainsofiron-initiatedpolymerized sur-faces.Thewettabilitycriterionisobviouslystronglylimitedasthe surfacemechanicalpropertiesandstructure,celltypesandcharge densityareignored.Forexample,Bacakovaetal.showedthatsoft matricesare notfavorablefor celladhesion[6].Theyexplained thatECMdepositedonsuchsurfacesisnotabletoresisttheforces involvedduringcellfocalpointformation.
Indeed,specificinteractionsarenotenoughtodescribed adhe-sivephenomenaentirelyandphysico-chemicalpropertieshaveto beaddedtotheequation.Hence,theinternalorganizationofcellsis remodelledthroughouttheirlifeandisstronglyinfluencedbythe surroundingmediumnotonlyviachemicalstimulationbutalso bymechanosensing,untilamorphologicalequilibriumisreached
[13,32].Thereby,theshapeofcells, aswellastheirrigidityand motilitydependontheirsupport.Forexample,manycellshavethe abilitytosensethestiffness,byapplyingastress,oftheir exter-nalenvironment.Thesecellsincludebrain,muscle,neuronsand manyothercelltypes[33].Cellsprobethesurfacethroughmyosin andactin filamentcross-bridging anda stiffnesscontrolloop is setup:cytoskeletonand adhesionwilladaptdepending onthe feedback.Asaconsequence,stiffermatricesleadtoanincreasein theelasticityofthecells,and canbemeasuredbyatomicforce microscopy(AFM)[34]or,moregently,byindentationwith opti-caltweezers[35].However,rigidityscanningofthecellsubstrate isatime-consumingprocess,takingfromminutestohours[13], thusviscositycanbeconsidered,inthecaseofagelforexample. Sometimes,cross-linkingcanimprovecellactivityonagel[36].In addition,mechanosensingcanbeaninitiatorofcelldisplacement onsurfaces(mechanotaxis,discoveredbyLoetal.[37]),fromthe softtothestiff[38]andmotilitywereshowntobelinkedtofocal contactsandthusindirectlytocellularadhesion[39,40].
Moreover,dependingon theirtype, cells willnot behave in the same way depending on the elasticity of the biomaterial. Forinstance,onsoftmatrices,fibroblastsadhereinalabileway whereasonstiffermaterialstheymakestablefocalpoints (adhe-sion)andrigidifytheircytoskeletons.Consequently,themotilityof fibroblastsonstiffmaterialsisreduced[38].Themobilityofthe chainsthatconstitutethebrushescouldalsolead tosuperficial mechanicalinstabilityandthustoloweradhesion[41].
Despitetheadhesionofcells,thebiomaterialcanalsodisturb cellactivity.Aperturbationintheexocytosisresponseofcellsis revelatoryofthisperturbationinwaysthatcan,forinstance,be measuredbyhistologicalstudiesorcarbon-fibremicroelectrode amperometry(CFMA),asobservedbyReedetal.[10].Inthisstudy, theintroductionof PNIPAM(throughplasmadepositionorspin coating)onsurfaceshaveshowntoslowdownthecellexchange betweenvesiclesandextracellularspace.Additionally,spin-coated PNIPAMwereshowntohyper-activatetheexocytosisactivityof cells,whereasplasmadepositedPNIPAMonlyaffectedkinetics.If thishyper-activationbythesurfacecanpresentharmfuleffects, somestudiesseekforacceleratingtissueregenerationby modify-ingthesurface,suchasobservedbytheintroductionoffreeamino grouponPLLAsurfaces[42].Thesurfacearchitectureisalsoakey point.3Darchitecturesarecommonlymodulated inthefield of tissueengineering,astheyreproduceamorerealisticnatural bio-logicalenvironment.Anicereviewofthistopicwaspublishedby AbbottandKaplanin2015[43].
Inconclusion,thesurfacehydrophilicity(presenceoffunctional groups),thesurfacestructureandstiffnessarecharacteristicsthat must be considered for efficient bioadhesion. However, in the
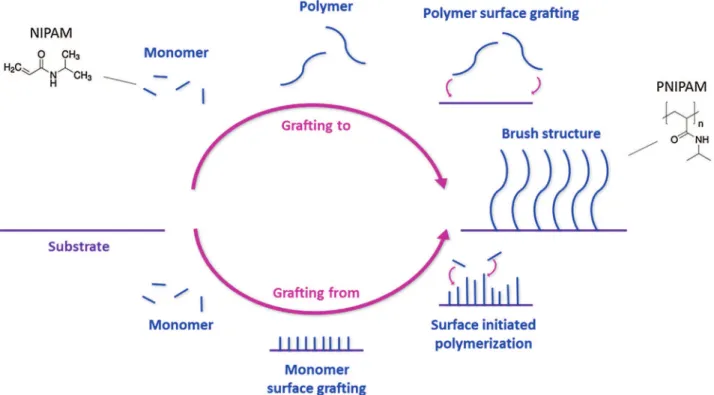
Fig.3. Twodifferentstrategiesofpolymergrafting(exampleofPNIPAM):thesurfaceinitiatedpolymerizationofamonomer(“graftingfrom”)orthegraftingofthepolymer (“graftingto”)onasurfacetoobtainabrushstructure.
presentreview,weonlyconsidersurfacemodificationthatdoes notinvolveanybulkmodification,inparticularintermof bioma-terialstiffness,andtheeffectofbrushstructuresonmechanical surfacepropertieswillnotbediscussed.Below,wefocusonakey step,i.e.thechoiceofsurfacemodificationmethodusedtoobtain brushstructuresonmaterialsurfacesthatenablegoodcontrolof thesurfacestateandproperties.
3. Surfacemodification 3.1. Brushstructure
Asdiscussedabove,thecontrolofbiomaterialsurfacesisa chal-lengeforscientistsinvolvedinthedevelopmentofmedicaldevices. Brushstructuresinparticularareinterestingintermsofprotein penetrationandcalibrationstudies[44].Thankstorecent devel-opmentsinchemistry,varioustechniquesareavailabletograftor coatbiomaterialswithpolymers.Forbiologicalapplications, cova-lentgraftingseemstobethebestchoicecomparedtophysically graftedsystems,duetorisksofdesorptionwiththislattermethod. Polymerbrushes,whichconsistonathinfilmofself-assembled polymers,areofinterest[10,45].Wettability,butalsothevarietyof possibleend-groupfunctions,thesubstrateandthechemistryare themainpointsthatmakethesesystemsattractive.The modular-ityofthepolymerbrushsynthesisisillustratedbythebroadrange ofsystemsthathavebeendevelopedinrecentyears:uniformed, patterned,orgradient(intermsofdensityorchemical composi-tion)brushlayershavebeenpreparedwithoneorseveralpolymers
[12,45].
Twodifferentapproachescanbeconsidered,namely“grafting to”(i.e.tothesurface)and“graftingfrom”,asshowninFig.3[45,46]. The “grafting to” method consists in coupling an end-functionalizedpolymerandareactivesurface.Thisapproachyields well-definedgraftedpolymer.However,thedepositedlayershave lowdensitiesandtheirthicknessislimited(100nm[47])duetothe sterichindranceduringgraftinganddiffusionprocesses[48–51].
Fig.4. AdvancedATRPmechanismscatalyzedbyCu(I)/Cu(II)complexes.Reprinted fromRef.[54],Copyright(2014),withpermissionofElsevier.
The“graftingfrom”methodconsistsinpolymerizationdirectly fromthesurface. Severaltechniquesare basedonthis process. Surface-initiatedpolymerization(SIP)basedonradicalchemistryis commonlyused[47,51,52].“Graftingfrom”techniquesyieldhigher graftedlayerdensitiesandovercomethicknesslimitations.These advantagesmadethisstrategythemostwidelyadopted.However, thecharacterizationofgraftedchainsismoredifficultand,apart frommodelsurfaces,stillremainsagreatchallenge.
Varioustechniquesarecommonlyusedforgraftingpolymers to surfaces, both for “grafting to” or “grafting from” strategies
[47,53,54].Amongthesetechniques,themostused,i.e.ATRPand RAFT,willbedevelopedinthenextsections.
3.2. Graftingtechniques
3.2.1. Atomtransferradicalpolymerization(ATRP)
Asisgenerallythecase incontrolledradicalpolymerization, ATRPchemistryreliesontheequilibriumbetweenactiveand dor-mantchains[46,47,55,56].ThemechanismisillustratedinFig.4. SurfaceinitiatedATRP(SI-ATRP)isa“graftingfrom”techniquethat
consistsinimmobilizingahalogenatedinitiatoronthesurface, fol-lowedbyATRP.Themainadvantages oftheATRParetheclose controloffilmthicknessandchainlengthwithlowpolydispersity
[47].Itisalsopossibletocontrolthicknessandgraftdensity sep-arately,bymodulatingthereactiontimeandstoichiometricratio, respectively[57,58].Inaddition,ATRPisknowntobeversatileand easytoperform(mildconditions)[51,58,59].Whilehigh temper-aturesmakethereactiontimeshorter,somestudiesalsoshowa slightreductionofthepolydispersity[60].Nevertheless,theneed ofmetalcatalystsisalimitationforbiomedicalapplications. Basi-cally,themetalisoxidized/reduced,andthusgeneratesorabsorbs aradical,leadingtotheactivation/deactivationofpolymerchains, respectively asshown inFig. 4.Atthe presenttime, due toits highcatalyticactivitycopperisusedmost[61,62].Iron catalyzed-ATRPcanbeperformedusinglowamountsofcatalyst,reducing thetoxicologicalrisksasiron isconsideredlesstoxicand more environmentallyfriendlythancopper[63–65].Ironisalsothemost abundantmetalonearthmakingitrelativelycheap;these charac-teristicshaveinitiatedalotofresearchandinterestinFecatalyzed organicchemistry,includingATRP,inlinewiththeperspectivesof “greenchemistry”[66,67].Thechoiceoftheligandisacomplex question;itdependsonthenatureofthepolymerandthecatalyst used[68]:forexample,pentamethyldiethylenetriamine(PMDETA) canbeusedincombinationwithCu[69],andtris(3,6-dioxaheptyl) amine(TDA)withFe[70].Furthermore,theactivatorsregenerated byelectrontransfer(ARGET)ATRP,developedbyMatyjaszewski etal.diminishtheamountofcatalystneeded(<50ppm)[71,72]. ThisadvancedATRP,derivedfromtheAGET-ATRP(foractivators generatedbyelectrontransferATRP)involvingareducingagentto initiatethereaction,consistsinusinganexcessofreducingagent (e.g.environmentallyfriendlyascorbicacid[73]).Thisinitiatordoes notonlygeneratebutalsomaintainsasufficientamountofCu(I) (inthecaseofcoppercatalyzedATRP)withouttheuseofaradical organiccompoundwhichcouldleadtosidereactions,cross-linking ortheformationofnewchains[55].Anotheradvantageof ARGET-ATRPisthattheoxidizedcatalyst,e.g.Cu(II)orFe(III),canbeused directlywithouttheneedforearly-stagereductionandcareful han-dling.Finally,thistechniqueincreasestheairtolerance andcan avoidtheneedofacontrolledatmosphere[74,75].Ascorbicacid (alsocalledvitaminC)ispreferabletoclassicSnreducingagents duetoitsnon-toxicitytowardshumanbeingsand the environ-ment.However,ascorbicacidpresentsthedisadvantageofbeing astrongreducingagent,soitsuseinwatercanleadtoanextensive conversionofCu(II)toCu(I)andcandiminishthecontrolofATRP. Onesolutionwouldbetousealessefficientsolvent,suchasanisole, todecreasethereducingactivityoftheascorbicacid[73,75].
Initiatorsforcontinuousactivatorregeneration(ICAR)ATRPuse theadditionofafreeradicalinitiatorsuchasazobisisobutyronitrile (AIBN)to(re)generatetheactivemetal[63,71].
Insupplementalactivatorandreducingagent(SARA)ATRPthe reducing agent is M(0), e.g. Cu(0) for a copper catalyzed ATRP
[67,76].Usingironpowder,thepolymerizationcanbecatalyzedby Fe(0)[60],withorwithouttheuseofFe(III)saltsbutinthislatter casealesscontrolledpolymerizationovertimecanoccur[63].
Many ATRP elaborations are involved for surface modifica-tion, nonetheless we would like to mention biomedical uses of this chemical route: biofouling surfaces or membranes [77], double responsive cellulose membranes [78,79], cell attach-ment/detachment(throughPNIPAMgrafting)[69,80].Toperform a“graftingfrom”,theideaistochemicallygrafttheinitiatoronthe surfacesothegrowthwillbedirectlyinitiatedonthematerial.This stepisfacilitatedbythefactthatATRPinitiatorsareacylbromides. Aself-assembledmonolayer(SAM)isgenerallyalsograftedbefore theinitiator. ATRPcanbeachievedincombination withplasma treatmentstohelpinitiatorimmobilization[81].Afewexamples ofSI-ATRParegiveninTable1.
ATRPcommonlyexhibitsapseudo-first-orderkinetics,atleast below high rates of conversion. The direct characterization of grafted polymer through the “grafting from” method is diffi-cult.Generally, a sacrificial initiator is used,assumingthat the polymer growth is similar on the surface and in the medium
[51,52,58,59,82–85].Anothermethodistousereversibleor break-ablesurfacebondsinordertodetachandstudythegraftedpolymer
[86].Amoredetaileddiscussiononbrushcharacterizationisgiven inSection3.3.
3.2.2. Reversibleaddition-fragmentationchaintransfer(RAFT) radicalpolymerization
SimilarlytoATRP,RAFTpolymerizationisaliving polymeriza-tionbasedontheequilibriumbetweenactive(i.e.bearingradicals) and dormant chains, and also shows pseudo-first-order kinet-ics.Initiationisperformedintraditionalways,e.g.usingthermal initiatorssuch as azobisisobutyronitrile(AIBN)or4,4′ -azobis(4-cyanovalericacid),whichhastheadvantageofbeingcarboxylic acidend-functionalized.Achaintransferagent(CTA)(alsocalled RAFTagent)ensuresthisequilibriumduringthepropagationsteps, asshowninFig.5[87,88].Thereductionofactivechain concen-trationresultsinanarrowdistributionofthechainlength,witha polydispersityindex(PDI),forPNIPAM,abletoreachvaluesaround 1.20,but PDIbelow1.10canbeobtainedinoptimalconditions
[89–91].Additionally,RAFTpolymerizationcanbeachievedwith abroadrangeoftemperatures,fromroomtemperatureto140◦C
[92].Ahighertemperatureallowsashorterreactiontime;lower polydispersitiescansometimesbeexpected.
OneofthemainadvantagesofRAFTpolymerizationcompared toATRPisthatitisametal-freechemicalroute.Incontrast,itcan requirethesynthesisoftheRAFTagent.InmodernRAFT polymer-ization,thisagentclassicallycontainsathiocarbonylthiomoiety, asforthecommonlyusedtrithio-carbonateanddithio-carboxylate type.AstheRAFTpolymerizationprocessreliesonthekineticsof additionand fragmentationofthis agent,thechoiceof its sub-stitute,classicallycalledZandR,iscrucial.Zisdedicatedtothe activationof thedouble bondby stabilizingthe adductradical, Risaleaving group.A completediscussion aboutthechoiceof theRAFTagentisavailableintheliterature[92].Thesurface initi-atedRAFT(SI-RAFT)polymerizationofPNIPAMcanallowtoobtain polydispersitiesbelow1.3[93].Thankstothesulfurylgroupsof theCTA,elementalanalysiscan,insomecases,beusedto deter-minethequantityofgraftedRAFTagent.Thetheoreticalmolecular massofthepolymer,Mnth,canbeestimatedthroughthefollowing equation:
Mnth= [Mono]0 [CTA]0
×MMono×con
v
.+MCTAwhereMMonocorrespondstothemolecularweightofthemonomer, [Mono]0toitsinitialconcentration,conv.istheconversionrateof themonomer,MCTAisthemolecularweightoftheCTAand[CTA]0 itsinitialconcentration[91,93].
InordertoperformSI-RAFTpolymerization,theinitiator[90]
ortheCTA[93]havetobepreviouslygraftedtothesurface.Inthe firstcasethehomolyticcleavageoftheinitiatorwillleadtogrowth eitheronthesurfaceorinthemedium,bothwiththefreeCTA.In thecaseofCTAgraftedsurfaces,theinitiatorandanotheramount oftheRAFTagentareintroducedtopermitthepolymerizationof freechainsandtheircharacterization.FewexamplesofSI-RAFT polymerizationaregivenTable1.
3.3. Characterizationofbrushes
Characterizationofthe“graftedfrom”polymerbrushisa chal-lengingtask.Inspecificcases,thegraftedchainscanberemoved fromtheirsubstrate[86].Inothercases,freechainsaregenerally
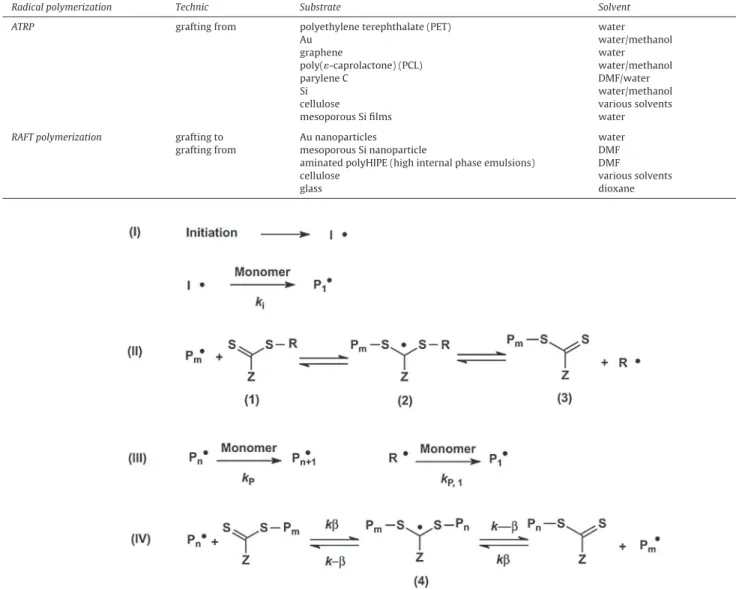
Table1
ExamplesofsurfacefunctionalizationwithPNIPAMbrushesusingATRPorRAFTpolymerizationprocess.
Radicalpolymerization Technic Substrate Solvent Refs.
ATRP graftingfrom polyethyleneterephthalate(PET) water [94]
Au water/methanol [95]
graphene water [96]
poly(«-caprolactone)(PCL) water/methanol [69]
paryleneC DMF/water [80]
Si water/methanol [97]
cellulose varioussolvents [98,99]
mesoporousSifilms water [100]
RAFTpolymerization graftingto Aunanoparticles water [89]
graftingfrom mesoporousSinanoparticle DMF [101]
aminatedpolyHIPE(highinternalphaseemulsions) DMF [93]
cellulose varioussolvents [98,102]
glass dioxane [90]
Fig.5. MechanismofaRAFTpolymerization.TheCTAplaystheroleofactivator/deactivator.ReproducedfromRef.[86],Copyright(2002),withpermissionofJohnWileyand Sons.
produced(seeSections3.2.1and3.2.2).Iffreepolymerisgenerated, themolecularweightcanbeeasilydeterminedbysize-exclusion chromatography (SEC)or viscosimetry.From opticalwaveguide lightmodespectroscopy(OWLS)themassofadepositedpolymer canalsobeobtained[23].
Moreover,themorphology,graft densityand thicknesshave tobedetermineddirectlyonthesurface.AFMisapowerfultool tostudymorphology[103,104](Fig.6).Forexample,inthecase ofPNIPAM,achangeinconformationaccompanyingachangein temperaturecanbeobservedbyAFM[103].
Surfaceplasmonresonance(SPR)givesthewetthicknessofthe layer,i.e.itsthicknessinaliquidenvironment,takingintoaccount swellingphenomena.Thedrythickness,i.e.intheabsenceofwater, ofthegraftedlayercanbemeasuredbyellipsometry[103].AFM canalsobeusedtodeterminethedrythickness,butsystematic errorswerereportedduetoAFMtipsbeingattractedbythePNIPAM layer[105].PNIPAMgraftingdensitycanbededucedfromthedry thicknessvalue,throughthefollowingequation:
s= hrNA Mn
withsthegraftdensity,hthedrythickness,NAAvogadro’s num-berandMnthemolecularweight.ThedensityofdryPNIPAM,r,is sometimesarbitrarilytakenequalto0.95g/cm3bycertainauthors
[106],buttheactualdensitycanbemeasuredbyU-tubeoscillation
[107]orbyX-rayreflectometry[108].Fouriertransforminfrared (FTIR)spectroscopycanalsobeused,insomecases,todetermine thegraftdensity,asreportedbyMizutanietal.[109].
Chemical analysis can beperformed through classic surface analysis:X-rayphotoelectronspectrometry(XPS),attenuatedtotal reflectionFTIRspectroscopy(ATR-FTIR),Ramanspectroscopy.For nanolayerstudies,XPSispreferabletoATR-FTIRspectroscopydue toitslowerpenetrationdepth(afewnmforXPSagainstupto1mm forATR-FTIRspectroscopy)[28].Secondaryionmassspectroscopy (SIMS)techniquescanbeevenmoresurfacelocalized.Inthis tech-nique,thesurfaceisetchedbyanionbeamandsputteredmaterial iscollectedbyadetector.Elementsandchemicalstructurecanbe determined.
Quartzmicrobalance(QCM)isaninterestingtoolforthe eval-uationoftheamountofadepositedlayer.Thedeterminationof thequantityofproteinsadsorbedonabiomaterialsurfacecanbe obtainedbymeasuring,insitu,thefrequencyshiftofthequartz
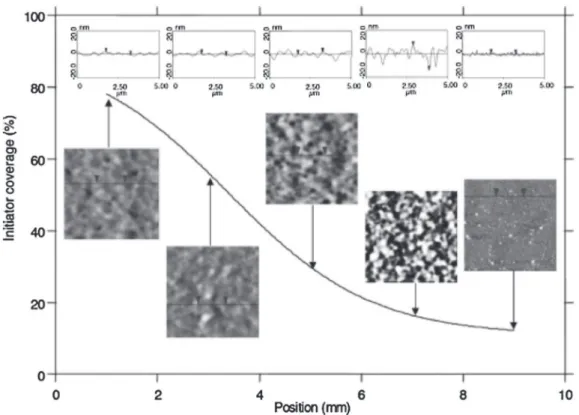
Fig.6. RelationshipofPNIPAMfilmmorphologytolocalgraftingdensityastrackedthroughtheinitiatordensity.Thegraftingdensityincreases,fromadiscontinuous mushroomstructure(left,lowgraftingdensity),toaheterogeneouspatchystructure(middle,intermediategraftingdensity).Athighgraftingdensity(right),asmoother, presumablymoreextended,structureisobtained.ReprintedfromRef.[102].Copyright(2006),AmericanChemicalSociety.
[24].Quartzmicrobalancewithdissipationmonitoring(QCM-D)is usefultoobtaintheswellingbehaviorofpolymerbrushes[97].
Finally,carbonfibremicroelectrodeamperometry(CMFA)can giveinformationaboutbiochemicalexocytosisofcells(kineticsand amountofrelease)andthusevaluatehowthesubstrateimpactsthe excretionactivityofcells[10].
Mostofthesetechniquesarenotapplicableto“brushgraftedon polymer”systemsduetotherelativelyhighroughnessofthistype ofsurface,whichexplainsthelackofreliablebrushcharacterization techniquesintheliterature.
4. Smartbio-surfaces:exampleof
poly(N-isopropylacrylamide)thermosensitivesurfaces
Poly(N-isopropylacrylamide)isathermo-responsivepolymer. Its structure is shown in Fig. 3. Indeed, PNIPAM changes its water affinityaccording tothetemperatureof thesurrounding medium,turningfrom hydrophilictohydrophobic.Thischange occurs at around32◦C, whose temperature is calledthe lower criticalsolutiontemperature(LCST),andleadstoachangeinits conformation.Abovethistemperature,PNIPAMcollapsesin solu-tion.Thiscoiltoglobuletransitionisendothermicandisrelated towater/polymerand polymerinterand intra-molecule hydro-genbonding.In otherwords,below theLCSTPNIPAMisbound towaterthroughamide/water(C O···H O)hydrogenbonding.As thetemperatureincreasesaboveLCST,thepolymerbecomes dehy-dratedandamide/amine(C O···H N)hydrogenbondingappears
[110,111].InthecaseofsurfacesgraftedwithPNIPAM,itmeans thata“brush”systemcanbeturnedintoa“mushroom” conforma-tionabovetheLCST.TheLCSTofPNIPAMwasreportedtodependon thechainlengthandthegraftingdensitywhileremainingbetween roomandphysiologicaltemperaturemakingthispolymer interest-ingforvariousbiomedicalapplications[105].Additionally,theuse ofPNIPAMinacopolymersystem[112],aswellasthepresenceof
saltscanstronglyinfluencethisLCST[113,114].Cl−andCH 3COO− haveaparticularlystronginfluenceaspredictedbythe Holfmeis-terseries.Moreover,ionconcentrationsaregenerallylow(below 0.15M)inbothculturemediaandbodyfluids,itsinfluencethus hastoberelativized.Itisnoteworthytomentionthatproteins,if concentrated,canalsoaffecttheLCSTofPNIPAMfromadecrease of2.6◦Ctoanincreaseof1.5◦C,dependingontheproteininvolved
[111].
Surfaceinteractionscanbemodulatedbythetemperatureof thePNIPAM.Thisvariationoftheinteractionsisclearlyobservedby AFMmeasurements[105,115].Bovineserumalbumin(BSA)bound toAFMtipswasusedtostudythevariation ofprotein/PNIPAM surface interactions. It appeared that interactions are tempera-turedependent,withaproteinadsorptionphenomenonoccurring aboveLCST [116].Thisphenomenon wasalsoobservedusing a QCM.ThemechanismisrelatedtoPNIPAMhydrationbutisnot wellunderstoodatthepresenttime. Variousstudieswere per-formedtoevaluatetheabilityofPNIPAMtotriggercellattachment. InadditiontotheirslightcontrolofLCST,itappearsthatthegrafting densityandthechainlengthalsoplayanimportantroleon bioad-hesion.Thus,thematerialseemstoberesistanttotheadsorption ofeitherproteinsorcellswhenchaindensityandchainlengthare bothhigh[117].Thisisexplainedbythedifficultyforproteinsto enterthePNIPAMlayerduetosterichindrancewhenchaingrafting isdense.Halperinetal.proposedatheoreticalapproachof mech-anismsforharvestingcellsculturedonthermoresponsivePNIPAM polymerbrushes[118].
First,two interaction modeshave tobeexaminedwhen we considerparticles(e.g.proteins)andabrushstructure.The com-pressivemode,wherethebrushesarecompressedbyaparticle, occurswhenitssizeisgreaterthanthespaceavailablebetween chains,takingintoaccounttheabilityofchainstorearrange them-selvesaroundtheparticle.Thisistypicallythecaseforcells.The other mode, theinsertive mode, occurs for small particles, e.g.
Fig.7. Thethreedifferentmodesofproteinadsorptionthroughbrushes:(A)primary adsorption,(B)ternaryadsorptionand(C)secondaryadsorption.AdaptedfromRef.
[117],Copyright(2012),withpermissionofElsevier.
extracellularproteinssuchasfibronectin.Here,weseethe impor-tanceofthegraftingdensity:highdensitylimitsproteininsertion. Then,thedepthoftheinsertedproteincanalsovary,formingthree modes:primary,secondaryandternaryadsorption(Fig.7).High affinitybetweenECM proteinsandsubstrate promotesprimary adsorption,whereasahighgraftingdensitytendstosuppress pri-maryandmaybeternaryadsorption[117,118].Primaryandternary adsorptionfavorbioadhesion,whereasatheoreticalmodelpredicts that secondary adsorption preferably occurs for large cylindri-calproteins[44,119].Nonetheless,ifternaryadsorptionmediated bioadhesionwasshowntobepossible,itseffectonprotein denatu-rationisstilltobedemonstrated[118].Finally,athinlayer(i.e.low molecularweight)facilitates primaryandternaryadsorbed pro-teins/cellsinteractions,leadingtoanincreaseofcelladhesion.In addition,proteinadsorptionisalsoconcentrationdependentand, athighconcentrations,theadsorptionratebelowandaboveLCST canbecomeclose[120].Then,thegraftdensitywasshownto influ-encethebrush structure. Ahighgraft density cancause phase separation[121],andmoregenerallythedensitywillmodulatethe proteinadsorptionratewithinthebrushes,asforBSA[44].Malham etal.showedthatchainrearrangementsovertimecouldslightly increaseinter-chainadhesionandrelated thisto−NHand C O hydrogenbonding[121].It isobviousthatinter-chainattractive interactionswouldplayaroleonproteininclusion.
VariouscellsadheretoheatedPNIPAMsurfacesandarereleased during cooling [122]. Typically, cell detachment is achieved at T=20◦C [118].Thispropertyallowscellstobeseededand gen-tlydetachedthem,withoutaneedoftrypsin:itisusedinthefield ofcellsheetengineering[123].
Monoormultilayercellsheetsarethusproducedandusedfor tissueregeneration[2].Thesebiologicallayersdemonstrategood integrationintissues.Invivostudieswereperformedtotreat vari-ousdiseases:cartilagedegeneration[124],damagedcornealtissues
[125]orcardiactissues[126].Interestingly,itappearedthatafter celllift-off,alayerofECMremainsattachedtothesurface.Research hastriedtodeterminethecompositionofthis remnantprotein layer[127]anditwasshownthatmostofthefibronectinleaves thesurfacewiththecells.Nonetheless,thisremnantlayerpromotes newcellgrowth,showingitsviability.
Alotofsystemshavealready beendevelopedusingPNIPAM brushsurfaces.PNIPAMbrushesweresuccessfullygraftedthrough ATRP[69,128]and RAFTpolymerization [90,93].Byintroducing reactivegroupsthroughplasmatreatments,PNIPAMcanbegrafted via“graftingto”amidebinding[8]orsurfaceinitiatedATRP[81]. ATRPproducedSi-PNIPAMbrushhybridswhichwereshowntobe efficientinthethermo-triggeredadhesion/de-adhesionof fibro-blastcells[128].InthiscasethethickerthePNIPAMlayeris,the moreprofitablethesurfaceisforcellproliferationafter2days.In allcases,noadhesionisobserved fortemperatures belowLCST independently of the thickness (3nm, 11nm or 31nm).It also appearedthattheantifoulingpropertiesofthepoly(ethylene gly-col) monomethacrylate (PEGMA) in combination with PNIPAM increasesthecellreleaseabilitiesofPNIPAM.Forbovine endothe-lialcells,athicknessofPNIPAMbrushontissueculturepolystyrene
around15nmshowedoptimaladhesion/de-adhesionproperties
[129].Theyalsoreportednoadhesionabove30nm,whereas Mitzu-tanietal.observedthatendothelialcelladhesiononpolystyrene ATRPgraftedPNIPAMsurfacesissuppressedforthicknessgreater than60nm[109].Moreover,thebestadhesionwasobtainedfor thinnerPNIPAMlayers(1.8nm).Takahashietal.developed surface-initiated RAFT polymerization brushes on glass coverslips and studiedboth graftdensity andmolecular weightof PNIPAMon reversiblebioadhesion[90].Thestudyshowedthattheamountof cellsalsoincreasedonloweringthegraftdensity.Inaddition, bet-terbioadhesionisobservedforshorterbrushesbutde-adhesion needsathickenoughlayer.Theexplanationisrelatedtothe neces-sitytopushcellsfromthesurface,asthePNIPAMbrushesbecome extendedonreducingthetemperature.Thiscanbethegeneral con-clusion,ifpossible,ofthicknessconsiderations:abalancebetween theabilityforcellstoattach(thinbrushlayer)anddetach(thick brushlayer)astobefound.Consequently,athickPNIPAMlayer canbeusefultoproduceproteinresistantsurfaces.
Zhao et al. studied the anti-fouling properties of PNIPAM graftedpolyurethanesurfacesagainstfibrinogenandhumanserum albumin (HAS) proteins at 37◦C [131]. It appeared that the thermosensitivity of the hydrophilicity was not significant on lowPNIPAMthickness,andthattheproteinadsorptionstrongly decreased asthis thicknessincreased.Thiseffectcanbedue to higherhydrophilicityofthickerlayers.Asaconsequence,cellsdo notadheretothickbrushesandthusanti-adhesionsurfacescanbe producedbytheuseofPNIPAM.Yuetal.showedthickness depen-dent thermo-sensitivity of PNIPAMgrafted Si (surface initiated ATRP)surfacesandmanagedtoproduceHSArepellent,evenwith thinPNIPAMlayers(<15nm)[132].Thevariationofcontactangle andHSAadsorptionbetween27and37◦Cisnotsonotableatlow PNIPAMthickness.However,greatertemperaturesensitivitywas observedathighergraftthicknessbothoncontactangleandHSA proteinadsorption.Moreinterestingly,at37◦CHSAadsorptionis notlinearlydependentwithPNIPAMthicknessand,asthe thick-nessincreases,adecreaseofsensitivityfollows.Thisobservation wasattributedtopossibleadsorptionontheSi-initiatorsurfaceat lowgraftthickness.Ascontactangleshowedhydrophobicsurfaces (higherthantraditionalanti-foulingpolymer),theauthorsdeduced thattheanti-foulingpropertiesoflowPNIPAMthicknesswerenot duetothehydrophilicityofthePNIPAMsurface,buttothe inter-actionsbetweenPNIPAMandthesubstrate.Indeed,shortPNIPAM brushendchainscanalsointeractwiththesubstrateandreducethe freedomofconformationchanges,reducingthetemperatureeffect
[132].Thisstudyalsoshowedtheimportanceoftheproteinsizeon adsorption.Indeed,thesizeoftheproteinmoleculeisofimportance asthepenetrationwillbedependentonsterichindrance phenom-ena.Inaddition,itcanbenotedthatthethreeproteinsstudied,HSA, fibrinogenproteinandlysozymealsohavedifferentcharge char-acteristics.Thesmallestprotein,thelysozymes,adsorbedwhether ornotthePNIPAMwasincollapsedorextendedregime.An expla-nationcanbetheabilityofthissmallproteintopassthroughthe PNIPAMbrushesandthentointeractwiththesubstrate(primary adsorption).Astheproteinsizeincreased,theproteinswereno longerabletoefficientlygothroughPNIPAMchainsbelowtheLCST (extendedregime),butareable,abovetheLCST,tointeractwiththe outermostregionofPNIPAMwhenhydrophobicandmaybewith thesubstrate(collapsedregime).
Comparingthesetwolastresults,itappearsthatinthecaseof polyurethanesubstratethehydrophilicitytendstoincreasewith thethickness,leadingtoa decreaseofproteinadsorption[131], whereastheSisubstrategraftledtoanincreaseofprotein adsorp-tion, as the hydrophobicity increased [132]. Thus, we see the importanceofthesubstrate,andtheresultingsurfaceproperties willdependontheabilityofitssubstratetoallowprimarybinding andonthehydrophilicity/hydrophobicitybalanceoftheresulting
surface.Infact,adsorptionofHSAwasofthesameorderof magni-tudewhateverthesubstrate,forthickerPNIPAMlayers,thelatter havingalsothesamehydrophilicity.
Nanostructuredorpatternedsurfaceswerealsoinvestigated. Silicon nanowires were thus used as a substrate for SI-ATRP
[133].Theadditionof PNIPAMstronglyreducedplatelet activa-tionandadhesion,bothaboveandbelowtheLCST.Asexpected, thenanostructurationofPNIPAMsurfaces(i.e.theincreaseof sur-facearea)involvesanexacerbationofthehydrophilic/hydrophobic surfacestate.Infact,Chenetal.highlightedthatthese nanostruc-tures,whichpresentahighaspectratio,tendtotrapwater.This entrappedwaterledtoareductionoftheplateletprotein/surface interactions,whateverthecoilorglobulestateofPNIPAMbrushes. Theseresultsopennewfieldsofapplicationasplateletactivation andadhesioncanleadtobloodcoagulationandthrombosis.More recently,siliconnanopillarswereshowntobeabletoreversibly attach/detach to/from breast cancer cells, through specific and selectiveinteractions[134].ComparedtoflatSi-PNIPAMsurfaces, theintroduction ofnanopillar architecturewidenedthe overall potentialcontactsurfacebutlimitedtheavailablespacefor inter-actions between cells and surfaceswhen adhered. As a result, the3Darchitectureofthesesurfacesenhancedcellcapture, but diminishedthetendencyofcellstospread,makingreleaseeasier. NanopatternedPNIPAMsurfaceswerealsousedtotrap,killand deliverbacteria[135].Inthiswork,biocidesweregraftedbetween patternedSI-ATRPPNIPAMbrushes.Additionally,nanopatterning isapotentialsolutiontoovercomethicknesslimitations:eventhick brushesallowscellstoattach,sothenecessitytohavethickenough brushesinordertodetachcellscanbemoreeasilyfulfilled[136]. OwingtothefactthatthickPNIPAMbrushesdonotsupportcell attachmentbut becomebioadhesive when nanopatterned, con-trolledspatializationofcellcultureispossible[136].
Inthefieldofbodyimplantsandsurgicalbiomaterials,Chenet al.grafted(“graftingto”)PNIPAM-COOHontochitosanthroughan amidebondresultinginacomb-likepolymerstructure(branched PNIPAMonachitosanbackbone)whichformsagel[137].The sur-facefunctionalizationwasfollowedbyastudyofchondrocytesand meniscuscellsbioadhesion.Thethermosensitivebehaviorof PNI-PAM(brushtomushroomthermotriggeredconformationchange) wasshowntoprovokeaphasetransition,liquidtosolid-like hydro-gel.Thegelificationwouldoccurinsidethebodyafterinjection. Fibronectinadsorptionwasobservedbyfluorescenceusing rho-daminelabelledfibronectin.Polypropylene-g-chitosan-g-PNIPAM wasperformedthrougha“graftingto”processwithaviewto eas-ilystrippingoffoftheskinwounddressing[138].Non-toxicityand temperature-responsivenessbehaviorwerefulfilled.
Whilebrushes dopresent someinterestingproperties,other non-brushsystemshavebeenusedtodevelopthesamekindof functionalities. Ignacio et al. made a wound dressing using UV grafted PNIPAMpolyurethane membranes [139]. New subcuta-neousconnectivetissuegrewbutnotoxicitywasobserved.The detachmentonmiceskinwoundswastriggeredbythereduction of temperaturebelow theLCST. We canalsomentionthe easy removalofretinalimplantsachievedwithPNIPAMsurfaces[140]. Inthisstudy,bioadhesion,measuredbyapull-offtest,appearedone minuteafterpassingthroughtheLCST.Thecorrelationbetweenthe cellculturebehaviorandthermo-sensitivetissueadhesionclearly indicates thatbioadhesionontissuesisrelated totheability of PNIPAMtoadsorbproteinsandthuscatchcells.
5. Conclusion
Bioadhesionisacharacteristicofinteractionsbetween materi-alsandcells.Thisphenomenonisnowbetterunderstoodandgives risetointerestingfieldsinbiomedicalsciencesuchascellsheet
engineering.Brushstructureshavebeenshowntobeefficientfor celladhesion,andtooffertheadvantageofawell-controlled prepa-rationprocess.The“graftingfrom”approachenablesdensebrush layerstobemadewithoutsterichindrancelimitationsandthus leadstohomogeneouslayers,especiallyforroughsurfaces.Living polymerizationprovidesawaytocontrolthegrowthofthese lay-ers.Varioustechniquesexist,suchasFe-catalyzedARGET-ATRPor RAFTpolymerizations.It isknownthatthegraftingmethodcan leadtodifferentproperties(e.g.cross-linking),andimpactthe sur-faceinteractionwithcells.PNIPAM,asathermo-sensitivepolymer, is widelystudied and is a promising polymerin the cellsheet generationarea,butitsapplicationscanbewider,including,for example,implants.Thus,severalparameterssuchasgraftdensity, layerthicknessandgraftingmethodhavetobestudied, character-izedandcomparedintermsofcytotoxicityandbioadhesion.Asyet, nosolutionshavebeenfoundtothoroughlycharacterizeandstudy brushesdirectlyonpolymersubstrates,this challengewillhave tobeovercomeinthefuture.Thislimitationputsabrakeonthe controlofthesurfacestate,whichisakeypointforthe prospec-tiveworkinbioadhesionandcanallowtheinvestigationofnew insightsinthebioadhesionfield,boththeoreticallyandinterms ofapplications.Insomecases,(i.e.biomedicalimplants) antifoul-ingsurfacesaresoughtinordertolimitthebiologicalcolonization oranyimmunologicalresponse.Onthecontrary,tissue engineer-ingneedsgoodintegration,andthusbioadhesion,ofcellswithin biomaterials.Strongeffortsareneededtofurtherinvestigatethe effectsofthephysio-chemicalparametersofsurfaces: hydrophilic-ity,roughness,mechanicalproperties,patterns.Thedevelopment ofinnovativebiomaterialswillbedependantoftheseadvances. Acknowledgement
Authors would like to thank the French National Research Agency for its financial support (ANR-14-CE17-0002-01, FP-BioPrevproject).
References
[1]B.Kasemo,Biologicalsurfacescience,Surf.Sci.500(2002)656–677,http:// dx.doi.org/10.1016/S0039-6028(01)01809-X.
[2]J.Yang,M.Yamato,C.Kohno,A.Nishimoto,H.Sekine,F.Fukai,etal.,Cell sheetengineering:recreatingtissueswithoutbiodegradablescaffolds, Biomaterials26(2005)6415–6422,http://dx.doi.org/10.1016/j.biomaterials. 2005.04.061.
[3]M.A.Cooperstein,H.E.Canavan,Assessmentofcytotoxicityof(N-isopropyl acrylamide)andpoly(N-isopropylacrylamide)-coatedsurfaces,
Biointerphases8(2013)19,http://dx.doi.org/10.1186/1559-4106-8-19. [4]A.Tourrette,N.DeGeyter,D.Jocic,R.Morent,M.M.C.G.Warmoeskerken,C.
Leys,Incorporationofpoly(N-isopropylacrylamide)/chitosanmicrogelonto plasmafunctionalizedcottonfibresurface,ColloidsSurf.APhysicochem. Eng.Aspects352(2009)126–135,http://dx.doi.org/10.1016/j.colsurfa.2009. 10.014.
[5]H.Lim,Y.Lee,S.Han,Y.Kim,J.Song,J.Kim,Wettabilityof
poly(styrene-co-acrylate)ionomersimprovedbyoxygen-plasmasourceion implantation,J.Polym.Sci.PartBPolym.Phys.41(2003)1791–1797,http:// dx.doi.org/10.1002/polb.10536.
[6]L.Bacakova,E.Filova,M.Parizek,T.Ruml,V.Svorcik,Modulationofcell adhesion,proliferationanddifferentiationonmaterialsdesignedforbody implants,Biotechnol.Adv.29(2011)739–767,http://dx.doi.org/10.1016/j. biotechadv.2011.06.004.
[7]A.M.G.Borges,L.O.Benetoli,M.a.Licínio,V.C.Zoldan,M.C.Santos-Silva,J. Assreuy,etal.,Polymerfilmswithsurfacesunmodifiedandmodifiedby non-thermalplasmaasnewsubstratesforcelladhesion,Mater.Sci.Eng.C 33(2013)1315–1324,http://dx.doi.org/10.1016/j.msec.2012.12.031. [8]A.Sdrobis¸,G.E.Ioanid,T.Stevanovic,C.Vasile,Modificationof
cellulose/chitinmixfiberswithN-isopropylacrylamideand
poly(N-isopropylacrylamide)undercoldplasmaconditions,Polym.Int.61 (2012)1767–1777,http://dx.doi.org/10.1002/pi.4268.
[9]Y.M.Lee,J.K.Shim,PreparationofpH/temperatureresponsivepolymer membranebyplasmapolymerizationanditsriboflavinpermeation, Polymer(Guildf)38(1997)1227–1232, http://dx.doi.org/10.1016/S0032-3861(96)00548-4.
[10]J.A.Reed,S.A.Love,A.E.Lucero,C.L.Haynes,H.E.Canavan,Effectofpolymer depositionmethodonthermoresponsivepolymerfilmsandresulting
cellularbehavior,Langmuir28(2012)2281–2287,http://dx.doi.org/10. 1021/la102606k.
[11]L.Moroni,M.KleinGunnewiek,E.M.Benetti,Polymerbrushcoatings regulatingcellbehavior:passiveinterfacesturnintoactive,ActaBiomater. 10(2014)2367–2378,http://dx.doi.org/10.1016/j.actbio.2014.02.048. [12]A.Olivier,F.Meyer,J.-M.Raquez,P.Damman,P.Dubois,Surface-initiated
controlledpolymerizationasaconvenientmethodfordesigningfunctional polymerbrushes:fromself-assembledmonolayerstopatternedsurfaces, Prog.Polym.Sci.37(2012)157–181,http://dx.doi.org/10.1016/j. progpolymsci.2011.06.002.
[13]G.Bao,S.Suresh,Cellandmolecularmechanicsofbiologicalmaterials,Nat. Mater.2(2003)715–725,http://dx.doi.org/10.1038/nmat1001.
[14]M.Gao,M.Sotomayor,E.Villa,E.H.Lee,K.Schulten,Molecularmechanisms ofcellularmechanics,Phys.Chem.Chem.Phys.8(2006)3692–3706,http:// dx.doi.org/10.1039/b606019f.
[15]W.Xu,H.Baribault,E.D.Adamson,Vinculinknockoutresultsinheartand braindefectsduringembryonicdevelopment,Development125(1998) 327–337http://www.ncbi.nlm.nih.gov/pubmed/9486805.
[16]K.Anselme,L.Ploux,A.Ponche,Cell/materialinterfaces:influenceofsurface chemistryandsurfacetopographyoncelladhesion,J.Adhes.Sci.Technol.24 (2010)831–852,http://dx.doi.org/10.1163/016942409X12598231568186. [17]D.G.Castner,B.D.Ratner,Biomedicalsurfacescience:foundationsto
frontiers,Surf.Sci.500(2002)28–60, http://dx.doi.org/10.1016/S0039-6028(01)01587-4.
[18]J.H.Lee,J.W.Lee,G.Khang,H.B.Lee,Interactionofcellsonchargeable functionalgroupgradientsurfaces,Biomaterials18(1997)351–358,http:// dx.doi.org/10.1016/S0142-9612(96)00128-7.
[19]A.Tamura,M.Oishi,Y.Nagasaki,EfficientsiRNAdeliverybasedon PEGylatedandpartiallyquaternizedpolyaminenanogels:enhancedgene silencingactivitybythecooperativeeffectoftertiaryandquaternaryamino groupsinthecore,J.Control.Release.146(2010)378–387,http://dx.doi. org/10.1016/j.jconrel.2010.05.031.
[20]A.Tamura,M.Nishi,J.Kobayashi,K.Nagase,H.Yajima,M.Yamato,etal., Simultaneousenhancementofcellproliferationandthermallyinduced harvestefficiencybasedontemperature-responsivecationic
copolymer-graftedmicrocarriers,Biomacromolecules13(2012)1765–1773,
http://dx.doi.org/10.1021/bm300256e.
[21]D.Fischer,Y.Li,B.Ahlemeyer,J.Krieglstein,T.Kissel,Invitrocytotoxicity testingofpolycations:influenceofpolymerstructureoncellviabilityand hemolysis,Biomaterials24(2003)1121–1131,http://dx.doi.org/10.1016/ S0142-9612(02)00445-3.
[22]H.Lodish,A.Berk,C.A.Kaiser,M.Krieger,A.Bretscher,H.Ploegh,etal., L’intégrationcellulairedansdestissus,in:DeBoeck(Ed.),Biol.Moléculaire LaCell,4eed.,2014,pp.925–975.
[23]J.Pei,H.Hall,N.D.Spencer,Theroleofplasmaproteinsincelladhesionto PEGsurface-density-gradient-modifiedtitaniumoxide,Biomaterials32 (2011)8968–8978,http://dx.doi.org/10.1016/j.biomaterials.2011.08.034. [24]J.Andersson,K.N.Ekdahl,J.D.Lambris,B.Nilsson,BindingofC3fragments
ontopofadsorbedplasmaproteinsduringcomplementactivationona modelbiomaterialsurface,Biomaterials26(2005)1477–1485,http://dx.doi. org/10.1016/j.biomaterials.2004.05.011.
[25]K.N.Ekdahl,J.D.Lambris,H.Elwing,D.Ricklin,P.H.Nilsson,Y.Teramura, etal.,Innateimmunityactivationonbiomaterialsurfaces:amechanistic modelandcopingstrategies,Adv.DrugDeliv.Rev.63(2011)1042–1050,
http://dx.doi.org/10.1016/j.addr.2011.06.012.
[26]E.A.Vogler,Structureandreactivityofwateratbiomaterialsurfaces,Adv. ColloidInterfaceSci.74(1998)69–117, http://dx.doi.org/10.1016/S0001-8686(97)00040-7.
[27]A.J.Pertsin,M.Grunze,Computersimulationofwaternearthesurfaceof oligo(ethyleneglycol)-terminatedalkanethiolself-assembledmonolayers, Langmuir16(2000)8829–8841,http://dx.doi.org/10.1021/la000340y. [28]Z.Ma,Z.Mao,C.Gao,Surfacemodificationandpropertyanalysisof
biomedicalpolymersusedfortissueengineering,ColloidsSurf.B Biointerfaces60(2007)137–157,http://dx.doi.org/10.1016/j.colsurfb.2007. 06.019.
[29]T.Suzuki,Y.Mizushima,Characteristicsofsilica-chitosancomplex membraneandtheirrelationshipstothecharacteristicsofgrowthand adhesivenessofL-929cellsculturedonthebiomembrane,J.Ferment. Bioeng.84(1997)128–132, http://dx.doi.org/10.1016/S0922-338X(97)82541-X.
[30]C.D.Tidwell,D.G.Castner,S.L.Golledge,B.D.Ratner,K.Meyer,B.Hagenhoff, etal.,Statictime-of-flightsecondaryionmassspectrometryandx-ray photoelectronspectroscopycharacterizationofadsorbedalbuminand fibronectinfilms,Surf.InterfaceAnal.31(2001)724–733,http://dx.doi.org/ 10.1002/sia.1101.
[31]B.G.Keselowsky,D.M.Collard,A.J.García,Surfacechemistrymodulates fibronectinconformationanddirectsintegrinbindingandspecificityto controlcelladhesion,J.Biomed.Mater.Res.A66(2003)247–259,http://dx. doi.org/10.1002/jbm.a.10537.
[32]M.Cockerill,M.K.Rigozzi,E.M.Terentjev,MechanosensitivityoftheIInd kind:tGFbmechanismofcellsensingthesubstratestiffness,PLoSOne (2015)1–11,http://dx.doi.org/10.1371/journal.pone.0139959. [33]D.E.Discher,P.Janmey,Y.Wang,Tissuecellsfeelandrespondtothe
stiffnessoftheirsubstrate,Science310(2005)1139–1143,http://dx.doi.org/ 10.1126/science.1116995.
[34]Q.S.Li,G.Y.H.Lee,C.N.Ong,C.T.Lim,AFMindentationstudyofbreastcancer cells,Biochem.Biophys.Res.Commun.374(2008)609–613,http://dx.doi. org/10.1016/j.bbrc.2008.07.078.
[35]M.S.Yousafzai,F.Ndoye,G.Coceano,J.Niemela,S.Bonin,G.Scoles,etal., Substrate-dependentcellelasticitymeasuredbyopticaltweezers indentation,Opt.LasersEng.76(2016)27–33,http://dx.doi.org/10.1016/j. optlaseng.2015.02.008.
[36]M.G.Haugh,C.M.Murphy,R.C.McKiernan,C.Altenbuchner,F.J.O’Brien, Crosslinkingandmechanicalpropertiessignificantlyinfluencecell attachment,proliferation,andmigrationwithincollagenglycosaminoglycan scaffolds,TissueEng.PartA.17(2011)1201–1208,http://dx.doi.org/10. 1089/ten.TEA.2010.0590.
[37]C.M.Lo,H.B.Wang,M.Dembo,Y.L.Wang,Cellmovementisguidedbythe rigidityofthesubstrate,Biophys.J.79(2000)144–152,http://dx.doi.org/10. 1016/S0006-3495(00)76279-5.
[38]S.Kidoaki,T.Matsuda,Microelasticgradientgelatinousgelstoinduce cellularmechanotaxis,J.Biotechnol.133(2008)225–230,http://dx.doi.org/ 10.1016/j.jbiotec.2007.08.015.
[39]H.-W.Liu,C.-P.Lin,Y.-J.Liou,K.-W.Hsu,J.-Y.Yang,C.-H.Lin,NBT-IIcell locomotionismodulatedbyrestrictingthesizeoffocalcontactsandis improvedthroughEGFandROCKsignaling,Int.J.Biochem.CellBiol.51 (2014)131–141,http://dx.doi.org/10.1016/j.biocel.2014.04.009.
[40]F.G.M.Russel,J.M.Bindels,C.H.Van,Celllocomotionandfocaladhesionsare regulatedbysubstrateflexibility,Proc.Natl.Acad.Sci.U.S.A.94(1997) 13661–13665,http://dx.doi.org/10.1073/pnas.95.20.13661.
[41]L.Bacakova,E.Filova,D.Kubies,L.Machova,V.Proks,V.Malinova,etal., Adhesionandgrowthofvascularsmoothmusclecellsincultureson bioactiveRGDpeptide-carryingpolylactides,J.Mater.Sci.Mater.Med.18 (2007)1317–1323,http://dx.doi.org/10.1007/s10856-006-0074-1. [42]Y.Zhu,C.Gao,X.Liu,T.He,J.Shen,Immobilizationofbiomacromolecules
ontoaminolyzedpoly(L-lacticacid)towardaccelerationofendothelium regeneration,TissueEng.10(2004)53–61,http://dx.doi.org/10.1089/ 107632704322791691.
[43]R.D.Abbott,D.L.Kaplan,Strategiesforimprovingthephysiological relevanceofhumanengineeredtissues,TrendsBiotechnol.33(2015) 401–407,http://dx.doi.org/10.1016/j.tibtech.2015.04.003.
[44]C.Xue,B.-C.Choi,S.Choi,P.V.Braun,D.E.Leckband,Proteinadsorption modesdeterminereversiblecellattachmentonpoly(N-isopropyl
acrylamide)brushes,Adv.Funct.Mater.22(2012)2394–2401,http://dx.doi. org/10.1002/adfm.201103056.
[45]M.A.C.Stuart,W.T.S.Huck,J.Genzer,M.Müller,C.Ober,M.Stamm,etal., Emergingapplicationsofstimuli-responsivepolymermaterials,Nat.Mater. 9(2010)101–113,http://dx.doi.org/10.1038/nmat2614.
[46]J.Ran,L.Wu,Z.Zhang,T.Xu,Atomtransferradicalpolymerization(ATRP):A versatileandforcefultoolforfunctionalmembranes,Prog.Polym.Sci.39 (2014)124–144,http://dx.doi.org/10.1016/j.progpolymsci.2013.09.001. [47]S.Edmondson,V.L.Osborne,W.T.S.Huck,Polymerbrushesvia
surface-initiatedpolymerizations,Chem.Soc.Rev.33(2004)14–22,http:// dx.doi.org/10.1039/b210143m.
[48]J.Draper,I.Luzinov,S.Minko,I.Tokarev,M.Stamm,Mixedpolymerbrushes bysequentialpolymeraddition:anchoringlayereffect,Langmuir20(2004) 4064–4075,http://dx.doi.org/10.1021/la0361316.
[49]B.Zdyrko,I.Luzinov,Polymerbrushesbythegraftingtomethod,Macromol. RapidCommun.32(2011)859–869,http://dx.doi.org/10.1002/marc. 201100162.
[50]K.S.Iyer,B.Zdyrko,H.Malz,J.Pionteck,I.Luzinov,Polystyrenelayersgrafted tomacromolecularanchoringlayer,Macromolecules36(2003)6519–6526,
http://dx.doi.org/10.1021/ma034460z.
[51]M.Husseman,E.E.Malmström,M.McNamara,M.Mate,D.Mecerreyes,D.G. Benoit,etal.,Controlledsynthesisofpolymerbrushesbylivingfreeradical polymerizationtechniques,Macromolecules32(1999)1424–1431,http:// dx.doi.org/10.1021/ma981290v.
[52]N.Ayres,C.D.Cyrus,W.J.Brittain,Stimuli-responsivesurfacesusing polyampholytepolymerbrushespreparedviaatomtransferradical polymerization,Langmuir23(2007)3744–3749,http://dx.doi.org/10.1021/ la062417+.
[53]U.Mansfeld,C.Pietsch,R.Hoogenboom,C.R.Becer,U.S.Schubert,Clickable initiators,monomersandpolymersincontrolledradicalpolymerizations–a prospectivecombinationinpolymerscience,Polym.Chem.1(2010)1560,
http://dx.doi.org/10.1039/c0py00168f.
[54]P.Liu,Modificationofpolymericmaterialsviasurface-Initiated Controlled/Livingradicalpolymerization,EPolymers7(2007)1–3,http:// dx.doi.org/10.1515/epoly.2007.7.1.725.
[55]P.Król,P.Chmielarz,RecentadvancesinATRPmethodsinrelationtothe synthesisofcopolymercoatingmaterials,Prog.Org.Coat.77(2014) 913–948,http://dx.doi.org/10.1016/j.porgcoat.2014.01.027.
[56]C.-Y.Hsiao,H.-A.Han,G.-H.Lee,C.-H.Peng,AGETandSARAATRPofstyrene andmethylmethacrylatemediatedbypyridyl-iminebasedcopper complexes,Eur.Polym.J.51(2014)12–20,http://dx.doi.org/10.1016/j. eurpolymj.2013.11.013.
[57]N.Singh,X.Cui,T.Boland,S.M.Husson,Theroleofindependentlyvariable graftingdensityandlayerthicknessofpolymernanolayersonpeptide adsorptionandcelladhesion,Biomaterials28(2007)763–771,http://dx.doi. org/10.1016/j.biomaterials.2006.09.036.
[58]W.Feng,J.Brash,S.Zhu,Non-biofoulingmaterialspreparedbyatom transferradicalpolymerizationgraftingof2-methacryloloxyethyl
phosphorylcholine:separateeffectsofgraftdensityandchainlengthon proteinrepulsion,Biomaterials27(2006)847–855,http://dx.doi.org/10. 1016/j.biomaterials.2005.07.006.
[59]J.Pyun,T.Kowalewski,K.Matyjaszewski,Synthesisofpolymerbrushes usingatomtransferradicalpolymerization,Macromol.RapidCommun.24 (2003)1043–1059,http://dx.doi.org/10.1002/marc.200300078.
[60]J.Qin,Z.Cheng,L.Zhang,Z.Zhang,J.Zhu,X.Zhu,Ahighlyefficient iron-mediatedAGETATRPofmethylmethacrylateusingFe(0)powderas thereducingagent,Macromol.Chem.Phys.212(2011)999–1006,http://dx. doi.org/10.1002/macp.201000737.
[61]R.Gong,S.Maclaughlin,S.Zhu,Surfacemodificationofactivemetals throughatomtransferradicalpolymerizationgraftingofacrylics,Appl.Surf. Sci.254(2008)6802–6809,http://dx.doi.org/10.1016/j.apsusc.2008.04.101. [62]C.Hou,R.Qu,C.Sun,C.Ji,C.Wang,L.Ying,etal.,Novelionicliquidsas
reactionmediumforATRPofacrylonitrileintheabsenceofanyligand, Polymer(Guildf)49(2008)3424–3427,http://dx.doi.org/10.1016/j.polymer. 2008.06.013.
[63]Y.Wang,Y.Zhang,B.Parker,K.Matyjaszewski,ATRPofMMAwithppm levelsofironcatalyst,Macromolecules44(2011)4022–4025,http://dx.doi. org/10.1021/ma200771r.
[64]J.Cao,L.Zhang,X.Jiang,C.Tian,X.Zhao,Q.Ke,etal.,Facileiron-mediated dispersant-freesuspensionpolymerizationofmethylmethacrylatevia reverseATRPinwater,Macromol.RapidCommun.34(2013)1747–1754,
http://dx.doi.org/10.1002/marc.201300513.
[65]L.Zhang,Z.Cheng,Z.Zhang,D.Xu,X.Zhu,Fe(III)-catalyzedAGETATRPof styreneusingtriphenylphosphineasligand,Polym.Bull.64(2009) 233–244,http://dx.doi.org/10.1007/s00289-009-0139-7.
[66]C.Bolm,J.Legros,J.LePaih,L.Zani,Iron-catalyzedreactionsinorganic synthesis,Chem.Rev.104(2004)6217–6254,http://dx.doi.org/10.1021/ cr040664h.
[67]R.Poli,L.E.N.Allan,M.P.Shaver,Iron-mediatedreversibledeactivation controlledradicalpolymerization,Prog.Polym.Sci.39(2014)1827–1845,
http://dx.doi.org/10.1016/j.progpolymsci.2014.06.003.
[68]S.-I.Nakanishi,M.Kawamura,H.Kai,R.-H.Jin,Y.Sunada,H.Nagashima, Well-definedironcomplexesasefficientcatalystsforgreenatom-transfer radicalpolymerizationofstyrene,methylmethacrylate,andbutylacrylate withlowcatalystloadingsandcatalystrecycling,Chemistry20(2014) 5802–5814,http://dx.doi.org/10.1002/chem.201304593.
[69]M.KleinGunnewiek,A.DiLuca,X.Sui,C.A.vanBlitterswijk,L.Moroni,G.J. Vancso,Controlledsurfaceinitiatedpolymerizationof
N-isopropylacrylamidefrompolycaprolactonesubstratesforregulatingcell attachmentanddetachment,Isr.J.Chem.52(2012)339–346,http://dx.doi. org/10.1002/ijch.201100118.
[70]J.Liu,W.He,L.Zhang,Z.Zhang,J.Zhu,L.Yuan,etal.,Bifunctional nanoparticleswithfluorescenceandmagnetismviasurface-initiatedAGET ATRPmediatedbyanironcatalyst,Langmuir27(2011)12684–12692,
http://dx.doi.org/10.1021/la202749v.
[71]K.Matyjaszewski,W.Jakubowski,K.Min,W.Tang,J.Huang,W.A. Braunecker,etal.,Diminishingcatalystconcentrationinatomtransfer radicalpolymerizationwithreducingagents,Proc.Natl.Acad.Sci.U.S.A. 103(2006)15309–15314,http://dx.doi.org/10.1073/pnas.0602675103. [72]W.Jakubowski,K.Matyjaszewski,Activatorgeneratedbyelectrontransfer
foratomtransferradicalpolymerization,Macromolecules38(2005) 4139–4146,http://dx.doi.org/10.1021/ma047389l.
[73]K.Min,H.Gao,K.Matyjaszewski,Useofascorbicacidasreducingagentfor synthesisofwell-definedpolymersbyARGETATRP,Macromolecules40 (2007)1789–1791,http://dx.doi.org/10.1021/ma0702041.
[74]B.V.Bhut,K.a.Conrad,S.M.Husson,Preparationofhigh-performance membraneadsorbersbysurface-initiatedAGETATRPinthepresenceof dissolvedoxygenandlowcatalystconcentration,J.Memb.Sci.390–391 (2012)43–47,http://dx.doi.org/10.1016/j.memsci.2011.10.057.
[75]P.Shivapooja,L.K.Ista,H.E.Canavan,G.P.Lopez,ARGET–ATRPsynthesisand characterizationofPNIPAAmbrushesforquantitativecelldetachment studies,Biointerphases7(2012)1–9, http://dx.doi.org/10.1007/s13758-012-0032-z.
[76]Y.Zhang,Y.Wang,C.Peng,M.Zhong,W.Zhu,D.Konkolewicz,etal., Copper-MediatedCRPofmethylacrylateinthepresenceofmetalliccopper: effectofligandstructureonreactionkinetics,Macromolecules45(2012) 78–86,http://dx.doi.org/10.1021/ma201963c.
[77]C.H.Worthley,K.T.Constantopoulos,M.Ginic-Markovic,R.J.Pillar,J.G. Matisons,S.Clarke,Surfacemodificationofcommercialcelluloseacetate membranesusingsurface-initiatedpolymerizationof2-hydroxyethyl methacrylatetoimprovemembranesurfacebiofoulingresistance,J.Membr. Sci.385–386(2011)30–39,http://dx.doi.org/10.1016/j.memsci.2011.09.017. [78]X.Qiu,X.Ren,S.Hu,Fabricationofdual-responsivecellulose-based
membraneviasimplifiedsurface-initiatedATRP,Carbohydr.Polym.92 (2013)1887–1895,http://dx.doi.org/10.1016/j.carbpol.2012.11.080. [79]K.Pan,X.Zhang,R.Ren,B.Cao,Doublestimuli-responsivemembranes
graftedwithblockcopolymerbyATRPmethod,J.Memb.Sci.356(2010) 133–137,http://dx.doi.org/10.1016/j.memsci.2010.03.044.
[80]C.Zhang,P.T.Vernier,Y.H.Wu,W.Yang,Surfacechemicalimmobilizationof paryleneCwiththermosensitiveblockcopolymerbrushesbasedon N-isopropylacrylamideandN-tert-butylacrylamide:synthesis,
characterization,andcelladhesion/detachment,J.Biomed.Mater.Res.—Part BAppl.Biomater.100B(2012)217–229,http://dx.doi.org/10.1002/jbm.b. 31941.
[81]W.Zhen,C.Lu,Surfacemodificationofthermoplasticpoly(vinyl alcohol)/saponitenanocompositesviasurface-initiatedatomtransfer radicalpolymerizationenhancedbyairdielectricdischargesbarrierplasma treatment,Appl.Surf.Sci.258(2012)6969–6976,http://dx.doi.org/10.1016/ j.apsusc.2012.03.145.
[82]J.-K.Chen,C.-Y.Hsieh,C.-F.Huang,P.Li,Characterizationofpatterned poly(methylmethacrylate)brushesundervariousstructuresuponsolvent immersion,J.ColloidInterfaceSci.338(2009)428–434,http://dx.doi.org/10. 1016/j.jcis.2009.06.040.
[83]Y.Liu,V.Klep,B.Zdyrko,I.Luzinov,Synthesisofhigh-densitygrafted polymerlayerswiththicknessandgraftingdensitygradients,Langmuir21 (2005)11806–11813,http://dx.doi.org/10.1021/la051695q.
[84]S.J.Eichhorn,A.Dufresne,M.Aranguren,N.E.Marcovich,J.R.Capadona,S.J. Rowan,etal.,Review:currentinternationalresearchintocellulose nanofibresandnanocomposites,J.Mater.Sci.45(2009)1–33,http://dx.doi. org/10.1007/s10853-009-3874-0.
[85]J.Lindqvist,E.Malmström,Surfacemodificationofnaturalsubstratesby atomtransferradicalpolymerization,J.Appl.Polym.Sci.100(2006) 4155–4162,http://dx.doi.org/10.1002/app.23457.
[86]C.Kang,R.M.Crockett,N.D.Spencer,Molecular-weightdeterminationof polymerbrushesgeneratedbySI-ATRPonflatsurfaces,Macromolecules47 (2014)269–275,http://dx.doi.org/10.1021/ma401951w.
[87]M.Barsbay,O.Güven,Ashortreviewofradiation-inducedraft-mediated graftcopolymerization:apowerfulcombinationformodifyingthesurface propertiesofpolymersinacontrolledmanner,Radiat.Phys.Chem.78 (2009)1054–1059,http://dx.doi.org/10.1016/j.radphyschem.2009.06.022. [88]C.Barner-Kowollik,T.P.Davis,J.P.a.Heuts,M.H.Stenzel,P.Vana,M.
Whittaker,RAFTingdownunder:talesofmissingradicals,fancy architectures,andmysteriousholes,J.Polym.Sci.PartAPolym.Chem.41 (2003)365–375,http://dx.doi.org/10.1002/pola.10567.
[89]K.Kusolkamabot,P.Sae-ung,N.Niamnont,K.Wongravee,M.
Sukwattanasinitt,V.P.Hoven,Poly(N-isopropylacrylamide)-Stabilizedgold nanoparticlesincombinationwithtricationicbranched
phenylene-Ethynylenefluorophoreforproteinidentification,Langmuir29 (2013)12317–12327,http://dx.doi.org/10.1021/la402139g.
[90]H.Takahashi,M.Nakayama,M.Yamato,T.Okano,Controlledchainlength andgraftdensityofthermoresponsivepolymerbrushesforoptimizingcell sheetharvest,Biomacromolecules11(2010)1991–1999,http://dx.doi.org/ 10.1021/bm100342e.
[91]J.Bigot,D.Fournier,J.Lyskawa,T.Marmin,F.Cazaux,G.Cooke,etal., Synthesisofthermoresponsivephenyl-andnaphthyl-terminated poly(NIPAM)derivativesusingRAFTandtheircomplexationwith cyclobis(paraquat-p-phenylene)derivativesinwater,Polym.Chem.1 (2010)1024–1029,http://dx.doi.org/10.1039/c0py00085j.
[92]HandbookofRAFTPolymerization,in:C.Barner-Kowollik(Ed.),Wiley-VCH VerlagGmbH&Co.KGaA,Weinheim,Germany,2008,http://dx.doi.org/10. 1002/9783527622757.
[93]F.Audouin,A.Heise,Surface-initiatedRAFTpolymerizationofNIPAMfrom monolithicmacroporouspolyHIPE,Eur.Polym.J.49(2013)1073–1079,
http://dx.doi.org/10.1016/j.eurpolymj.2013.01.013. [94]H.Alem,A.-S.Duwez,P.Lussis,P.Lipnik,A.M.Jonas,S.
Demoustier-Champagne,Microstructureandthermo-responsivebehavior ofpoly(N-isopropylacrylamide)brushesgraftedinnanoporesof track-etchedmembranes,J.Membr.Sci.308(2008)75–86,http://dx.doi. org/10.1016/j.memsci.2007.09.036.
[95]I.Lokuge,X.Wang,P.W.Bohn,Temperature-Controlledflowswitchingin nanocapillaryarraymembranesmediatedbyPoly(N-isopropylacrylamide) polymerbrushesgraftedbyatomtransferradicalpolymerization†, Langmuir23(2007)305–311,http://dx.doi.org/10.1021/la060813m. [96]L.Ren,S.Huang,C.Zhang,R.Wang,W.W.Tjiu,T.Liu,Functionalizationof
grapheneandgraftingoftemperature-responsivesurfacesfromgraphene byATRPonwater,J.NanoparticleRes.14(2012),http://dx.doi.org/10.1007/ s11051-012-0940-3.
[97]P.Zhuang,A.Dirani,K.Glinel,A.M.Jonas,Temperaturedependenceofthe surfaceandvolumehydrophilicityofhydrophilicpolymerbrushes, Langmuir32(2016)3433–3444,http://dx.doi.org/10.1021/acs.langmuir. 6b00448.
[98]W.Wu,J.Li,W.Zhu,Y.Jing,H.Dai,Thermo-responsivecellulosepapervia ARGETATRP,FibersPolym.17(2016)495–501,http://dx.doi.org/10.1007/ s12221-016-5877-1.
[99]L.L.Yang,J.M.Zhang,J.S.He,J.Zhang,Z.H.Gan,Synthesisand characterizationoftemperature-sensitive
cellulose-graft-poly(N-isopropylacrylamide)copolymers,Chin.J.PolymSci. (EnglishEd.)33(2015)1640–1649, http://dx.doi.org/10.1007/s10118-015-1703-2.
[100] R.B.Vasani,S.J.P.McInnes,M.A.Cole,A.M.M.Jani,A.V.Ellis,N.H.Voelcker, Stimulus-responsivenessanddrugreleasefromporoussiliconfilms ATRP-graftedwithpoly(N-isopropylacrylamide),Langmuir27(2011) 7843–7853,http://dx.doi.org/10.1021/la200551g.
[101] P.-W.Chung,R.Kumar,M.Pruski,V.S.-Y.Lin,Temperatureresponsive solutionpartitionoforganic-Inorganichybrid
poly(N-isopropylacrylamide)-coatedmesoporoussilicananospheres,Adv. Funct.Mater.18(2008)1390–1398,http://dx.doi.org/10.1002/adfm. 200701116.
[102] A.Hufendiek,V.Trouillet,M.A.R.Meier,C.Barner-Kowollik,Temperature responsivecellulose-graft-copolymersviacellulosefunctionalizationinan