O
pen
A
rchive
T
OULOUSE
A
rchive
O
uverte (
OATAO
)
OATAO is an open access repository that collects the work of Toulouse researchers and
makes it freely available over the web where possible.
This is an author-deposited version published in :
http://oatao.univ-toulouse.fr/
Eprints ID : 19233
To link to this article : DOI: 10.1016/j.apsusc.2017.05.253
URL :
http://dx.doi.org/10.1016/j.apsusc.2017.05.253
To cite this version : Michau, Alexandre and Maury, Francis and Schuster,
Frederic and Boichot, Raphael and Pons, Michel Evidence for a Cr metastable
phase as a tracer in DLI-MOCVD chromium hard coatings usable in high
temperature environment. (2017) Applied Surface Science, vol. 422. pp.
198-206. ISSN 0169-4332
Any correspondence concerning this service should be sent to the repository
administrator:
staff-oatao@listes-diff.inp-toulouse.fr
Evidence
for
a
Cr
metastable
phase
as
a
tracer
in
DLI-MOCVD
chromium
hard
coatings
usable
in
high
temperature
environment
Alexandre
Michau
a,
Francis
Maury
a,∗,
Frederic
Schuster
b,
Raphael
Boichot
c,
Michel
Pons
caCIRIMAT,CNRS/INPT/UPS,4alléeE.Monso,31030Toulousecedex4,France bCEASaclay,DFP/DPg,91191GifSurYvette,France
cUniversityGrenobleAlpes,SIMAP,CNRS,38000Grenoble,France
Keywords: Chromiumcoatings MOCVD Crmetastablephase Structuraltransformation Tracer Hardcoatings
a
b
s
t
r
a
c
t
Crdepositsarewidelyusedasprotectivecoatingsbutmultifunctionalperformancesarerequiredinharsh environmentsmotivatingresearchonnewprocesses.MOCVDofCrmetalcoatingswascarriedoutby directliquidinjection(DLI)ofauniquesolutioncontainingbis(ethylbenzene)chromiumasmetalsource andthiophenolasinhibitorofcarbideformation.Alowamount(<6%)ofthemetastabled-Crphasewas foundembeddedinthestablea-Crphase.Theformationofthismetastablephaseoriginatesfromboth thelowdepositiontemperature(<723K)andtheuseofthiophenol.ItwasnotreportedunderotherCVD conditions.Densecoatingsweredepositedbyimplementingamultilayergrowthmode.Suchcoatings exhibitahighnanohardnessofabout17GPa.Thed-Crmetastablephaseundergoesanirreversible struc-turaltransformationtobcc-Crabove723K.Themechanicalpropertiesofcoatingsarenotaffectedbythe structuraltransformationbecauseofthesimilarityoftheircrystallographicstructures(bothcubic),their densityveryclose(avolumecontractionofonly0.4%duringthetransformation)anditslowcontent.This metastablephaseisasignatureoftheDLI-MOCVDprocessanditcanbeusedasatracerforCrcoatings operatinginhightemperatureenvironmentwithoutlossofthebasicproperties.
1. Introduction
Inordertogrowprotectivemetallicchromiumcoatings,wet routesgenerallyusealotoftoxicchemicalsincludingchromicacid solutionsbasedonhexavalentchromium[1–3]whichisnow for-biddenbyREACHregulation.Severalalternativesareinvestigated usingtrivalentchromium[4,5]buttheyshouldalsoresultina reg-ulationban.
Attheopposite,itispossibletodepositchromiumcoatingsby PVDprocesses ascathodicmagnetronsputtering [6,7],cathodic arcevaporation[8],hollowcathodedischarge[9]andhighpower impulsemagnetronsputtering(HIPIMS)[10]withoutusing danger-ouschemicals.Themaindrawbacksofthesephysicalmethodsare theircomplexityforthedepositiononlargepieces,therelatively lowgrowthratesand finallythefactthattheyareline-of-sight deposition techniques, which excludes conformal and uniform coatingsonthreedimensionalobjects.
∗ Correspondingauthorat:CIRIMAT,ENSIACET,4,alléeEmileMonso,BP44362, 31030,Toulousecedex4,France.
E-mailaddress:francis.maury@ensiacet.fr(F.Maury).
CVDprocessesarewellsuitedforconformalgrowthoncomplex shapes.Theirdrawbackcomesfromthehighworking tempera-turesusingtraditionalhalidesprecursors.Forinstance,Crmetal coatingsaredepositedbypackcementationabove1300K[11–13]. However,byusingmetalorganiccompounds(MOCVD),the depo-sitiontemperatureissignificantlydecreased,forinstancebelow 773KforCr-basedcoatingsdependingontheprecursor[14,15], andevenat523Kusingreactivetetra-alkylchromiumcomplexes [16].Asaresult,thinfilmdepositiononawidevarietyofsubstrate materialsispossiblewithoutstructuralanddimensionalchanges. InspiteofaC-richvaporphaseoriginatingfromtheorganicligands oftheprecursor,metallicCrthinfilmscanbedepositedbyMOCVD usinginhibitorsofcarbonincorporationandcarbideformationas chlorinated[14,17–20]andsulfur-containingderivatives[21].
Anevenmoresurprisingchallengewastofindthatwhenthe emergingtechnologyofdirectliquidinjection(DLI)isimplemented for supplying a cold-wall reactor operating under atmospheric pressure with high vapor flow rates, the carbon incorporation inhibitorswerestilleffectivewhilelargeamountsofhydrocarbon solvent were injected with theprecursor [22–24]. Indeed DLI-MOCVDisanalternativeprocesswheretheprecursorisdilutedor dissolvedinaliquidorganicsolvent.Thissolutionistheninjected
asapulsedsprayinaflashvaporizationchamberand the reac-tivegasphaseistransportedbyacarriergastotheCVDreactor. Thistechniquegenerateshighlystableandcontrolledvaporflow ratesoftheprecursorleadingtorelativelyhighgrowthrateatlow temperature(<773K).Anappropriatechoiceofprecursorprovides highyields[15].Thus,DLI-MOCVDprocessforCrdepositionhas beensuccessfulusingC6Cl6[22–24]andC6H5SH[24]asinhibitors
andbis(arene)chromiumasmetalsource.
Themicrostructuralcharacteristicsandthereforetheproperties ofchromiumcoatingsdependonboththeprocessesusedandthe depositionconditions.Therearetwodifferentcrystallinephases ofchromiuminstandardconditions.Thefirstonecorrespondsto thestablebcccubicstructurewithIm-3m(229)spacegroupand 2.88Åcellparameter(PDF00-006-0694),notedbcc-Cr(ora-Cr). ThesecondoneisaprimitivecubicmetastablephasewithPm-3n (223)spacegroupand4.59Åcellparameter(PDF00-019-0323) [25,26],notedd-Cr.
Excepthigh-temperatureCVDprocessesthatproduce thermo-chemical diffusion coatings,mostof theabove cited processes, includingDLI-MOCVD,lead totheformationofa-Cr thin films, althoughthegrowthconditionsarefarfromthermodynamic equi-librium. Theprocesses leadingtothegrowthof themetastable phase are rare. Evaporation and condensation of chromium in argonatlowpressures[25–27]ledtotheformationofcrystalline metastabled-Cr.Thesameauthorsreportedthestructural trans-formationofthemetastablephasetothestableoneabove723K.
TheaimofthispaperistostudyCrcoatingsdepositedat temper-aturesbelow723Kinahot-wallDLI-MOCVDreactor.Weshowthey havetheparticularityofcontainingsmallamountsofmetastable d-Crphaseembeddedina-Crpolycrystallinematrix.Furthermore, theseCrcoatingsaresupersaturatedwithcarbonwhichgivesthem unusualmetallurgicalproperties.Theresultinghighnanohardness makesthemgoodcandidatesashardprotectivecoatings.Thiswork alsodemonstratesthatforapplicationsinharshenvironments,if theoperatingtemperatureexceeds 723K, themetastable phase irreversiblytransformsintothestablebccphase,actingasatracer withoutlossofthebasicpropertiesofthecoating.
2. Experimental 2.1. Depositionprocess
DepositionswerecarriedoutbyDLI-MOCVDin ahorizontal, hot-wall,pyrextubularreactor(300mmlongand24mminternal diameter)withanisothermalzonearound150mm.Si(100)and stainlesssteel(304L)plateswereusedassubstratesandplacedona planarhorizontalsubstrate-holderintheisothermalzone.Thetotal pressurewaskeptconstantat6.7kPaandthegrowthtemperature wasfixedateither673(400◦C)or723K(450◦C).
Commercial BEBC, bis(ethylbenzene)chromium (from Strem Chemicals,CAS12212-68-9,infactamixtureof[(C2H5)xC6H6-x]2Cr
where x=0–4)wasusedaschromiumprecursor. Itis aviscous liquidthatwasmixedwithanhydroustoluene(99.8%from Sigma-Aldrich,CAS108-88-3)at3×10−1molL−1(4gofBEBCin50mL oftoluene).Asulfur-containinginhibitorofcarbonincorporation, thiophenol,C6H5SH(fromSigma-Aldrich,purity>99%),was
intro-ducedintothissolutionwithamolerationthiol/nBEBC=2%togrow
preferentiallyCrmetalcoatingsinsteadofchromiumcarbide coat-ings.
The liquid solution containing both the precursor and the inhibitorwasinjectedinaflashvaporizationchamber(473K)at 0.9mLmin−1 byappropriately tuninginjection parameters
(fre-quencyandopeningtime).Nitrogenwasusedascarriergaswith a500sccmflowrateandwasheatedat453Kbeforeenteringthe flashvaporizationchambertopreventcondensation.
Table1
GrowthconditionsofCrmetalcoatingsbyDLI-MOCVDinahorizontalhot-wall reactorusinganinjectionofasinglesolutionofBEBCandthiophenolintoluene,as chromiumsourceandinhibitorofcarbideformation,respectively.
Coatingtype Monolayer Multilayers
(9layers)
Depositiontemperature(K) 673;723 673;723
Totalpressure(kPa) 6.7 6.7
CarriergasN2(sccm) 500 500
Solvent Toluene Toluene
BEBCconcentration(mol/L) 3.5×10−1 3.5×10−1
Thiophenolconcentration(mol/L) 7.0×10−3;3.5×10−2 7.0×10−3
Injectionfeedrate(mL/min) 0.9 0.4
Injectionfrequency(Hz) 10 3
Openingtimeofinjector(ms) 0.5 0.5
Growthduration(min) 60 140
Inafirstmode,coatingsweredepositedasmonolithiclayers, namelymonolayers,bykeepingconstantalltheconditionsduring thedepositionrun.Howeverthefirstanalysesrevealeda colum-narmorphologywithahighporositybetweenthecolumnswhich is not suitable for protective coatings. In order toincrease the compactness,inaseconddepositionmode,thecoatingswere struc-turedinmultilayersbystoppingtheprecursorinjectionfor5min every15minwithoutsignificanttemperaturechangeorventing. Thetemporaryshutdownofthegrowthcreatedcleaninterfaces. Atthebeginningofeachinjectionperiodanewnucleationstep andcrystalgrowthoccurredandwasstoppedbeforethe forma-tionofthecolumns,i.e.after15min,whichlimitsthethicknessof individuallayersandhindersthedevelopmentofcolumns.Thus, densemultilayercoatingswith9layerswereproduced. Character-izationswillbeshownforbothtypesofcoatings.Table1detailsthe experimentaldepositionconditions.
2.2. Coatingcharacterization
The morphology of the Cr coatings were characterized by scanning electron microscopy (SEM; Leo-435VP) and their microstructurewasstudiedbyElectronBackScatteredDiffraction (NordlysNanoEBSDDetectorwiththeAZtecHKLsoftwareinstalled ona SEM-FEGJEOLJSM-7100TTLS LV)and TEM(JEOLJEM2100 equippedwitha200kVFEG).Theircrystallinestructureand ther-malstabilitywereanalyzedbyXRDandin-situXRDvstemperature under Ar atmosphere, respectively (Bruker D8-2 diffractometer equippedwithagraphitemonochromator;Bragg-Brentano con-figuration;CuKaradiation).
Thefilmcompositionwasanalyzedbyelectronprobe micro-analysis(EPMA;CamecaSXFive,15kVand20nA)andthechemical environmentofeachelementofthecoatingwasinvestigatedusing anXPSspectrometer(ThermoScientificK-Alpha),equippedwith a monochromaticAlX-raysourceanda low energyAr+ gun(1
keV) forsurfacecleaning anddepthprofileanalysis.Rutherford BackscatteringSpectrometry(RBS;1.5MeVH+beam;detectionat
160◦)wasusedtoestimatethedensityofcoatings.
3. Resultsanddiscussion
3.1. Depositionofthebcc-Crstablephase
ManyCVDreactorconfigurationsand sizes,aswellas depo-sitionconditionshavebeenpreviouslytestedtodepositmetallic Cr [14,17–24]. These studies on MOCVD and DLI-MOCVD pro-cesses are summarized in Table 2. In the case of DLI-MOCVD process,thepresenceofalargeamountofhydrocarbonsolvent vaporisworthnoting.Experimentalsetsinvolving(i)additionof areactivegas(H2)totheinertcarriergas(N2),(ii)hot-walland
Table2
ExperimentalconditionsusedforCrdepositionbyMOCVDandDLI-MOCVD.Bis(benzene)chromium(BBC)andbis(ethylbenzene)chromium(BEBC)wereusedasprecursors andeitherC6Cl6orC6H5SHwereusedasinhibitorsofcarbideformation.
Process Reactora Carboninhibitor BBC BEBC
Atmb P(kPa) T(K) Ref. Atmb P(kPa) T(K) Ref.
MOCVDc hw C 2Cl6;C6Cl6 – – – – nr nr 573–748 [17] MOCVDc hw (C 6H5CH2)2S;(C6H5)2S – – – – vacuum 1.3×10−3 573–723 [21] MOCVD cw C6Cl6 H2 0.8 673 [14] – – – – MOCVD hw C6Cl6 H2 0.4–2.0 573–673 [14,18,19] H2 0.8 623 [18]
DLI-MOCVD cw C6Cl6 Tol;Cyclo 6.7;101 753–793 [22,23] – – – –
DLI-MOCVD hw C6Cl6 Tol 6.7 748 [24] – – – –
DLI-MOCVD hw C6H5SH Tol 6.7 748 [24] – – – –
DLI-MOCVD hw C6H5SH Tol 6.7 723 Thiswork Tol 6.7 673;723 Thiswork
acwmeanscold-wallverticalreactorandhwmeanshot-wallhorizontaltubularreactor.
bAtmmeansatmosphere;TolandCyclomeantolueneandcyclohexaneusedassolventstoinjecttheprecursorsolution.
c Theprecursorused,namelyBarkhoswasamixtureofbis(arene)chromiumslightlydifferentfromtheBEBC(nrmeansnotreported). orbis(ethylbenzene)chromium(BEBC)asbis(arene)chromium(0)
precursor,(iv)differentcarbideinhibitors,(v)totalpressurerange fromatmospherictolowpressure(0.4kPa),and(vi)growth tem-peraturerangefrom573to793Kwerestudied.Themainreason toexploretheeffectsofalltheseconditionscomesfromthekey roleplayedbysurfacechemistryforthecompetitivedepositionof metallicCrandcarbides.Obviouslythissurfacechemistrycanbe influencedbythegasphasechemistrywhichcanbedependenton experimentalconditionsandreactorconfigurations.
So far, MOCVD and DLI-MOCVD processes only led to the growthofsingle-phasepolycrystallinebcc-Crcoatings.Thisisthe stablebody-centered cubicstructure of themetal(spacegroup Im-3m).This common phase was obtainedby adding a chlori-nated[14,17,22,24]orasulfur-containing[21,24]inhibitortothe bis(arene)chromiumprecursor. The present work complements thesepreviousstudiesandisdistinguishedonseveralpoints:(i) theDLI-MOCVDprocesswasused,(ii)withBEBCandC6H5SHin
auniquesolutioncontainingboththeprecursorandtheinhibitor, (iii)H2wasnotaddedinthereactivegasphase,(iv)alargerCVD
reactorwasused,and(v)operatingtemperatureswerelowerthan 723K,asspecifiedinTable2.
Amonolayercoatingdepositedaccordingtomode1at723Kby DLI-MOCVDonsiliconsubstrateexhibitstypicallyacolumnarand porouschromiumstructure.Itisgenerallycomposedofthestable bcccrystallinephase.Noevidenceofcarbideformationwasfound byXRD.TheSEMmicrographofFig.1ashowsa5.7mmthickcoating
consistingofdisjointcolumnsperpendiculartothesurfaceofabout 1mmwidegrownfromadenser1mmthicksub-layerwherethe columnsarenotyetformed.ThecorrespondingXRDpatterninthe 2urange35–80◦exhibitsthetwomainpeaksofthestablebcc-Crat
44.4◦(110)and64.6◦(200).TheKa2contributioncanalsobeseen
asashoulderofthe(200)Crpeak,aswellastheKbcontributionfor themostintense(110)Crpeak(Fig.1b).Thisstablebcc-Crphase exhibitsapreferentialorientationofthegrowthalongthe[110] directionwithatexturecoefficientof1.8whichisconsistentwith acolumnarmorphology.
Asexpected,thecoatingsdepositedaccordingtothemode2 have a multilayer structure with a greater compactness and a smoothersurfacemorphologycomparedwithmonolayercoatings. Fig.2showsacross-sectionviewofacleavedsampleonSisubstrate grownat673K.Itconsistsof9layerswithclearinterfacesandthe totalthicknessis3.4mm.Theaveragegrainsizeislowerandless dispersedthanformode1;typicallyoftheorderofmagnitudeof thethicknessoftheindividuallayers(≤380nm).
Theinterfacesareclearbetweeneachlayerbecausethestopping oftheinjectionlastedlongenough(5min)tomakethetransient effects negligible and to suppress any nutrient species for the growthofthecoating.Indeed,theevacuationofthegaseous reac-tantsisrapidunderatotalpressureof6.7kPaandmaintainingthe carriergasflowrate(500sccm)duringthestoppingoftheprecursor injectionensuresanefficientpurgeofthereactor.
Fig.1.(a)SEMmicrographofacross-sectionand(b)correspondingXRDpatternofamonolayerCrcoatingdepositedonSisubstratebyDLI-MOCVDaccordingtomode1 (723K;6.7kPa;inhibitorC6H5SH).Thecoatingissingle-phasedandexhibitsatexturedbcc-Crstructure.
Fig.2. SEMmicrographofthecross-sectionofatwo-phasemultilayercoating grownat673Kand6.7kPaonSisubstrate(accordingtomode2)showingthe9 layerscomposingitanditshighcompactness.
Aboutthetransienteffectsinducedbythesemultilayer deposi-tionconditions,transientchangescanbeexpectedfortemperature and pressure.However thetotal pressureis independently and automaticallyregulatedatafixedvalueanditdoesnotaffectthe microstructureofcoatingswhenitisvariedinanarrowrange.Asa result,thetransientchangesofpressureinducedbythisstepwise deliveryofprecursorareinsignificant.Regardingthesubstrate tem-perature,thetemperatureprofilecalculatedinthis3-dimensionnal reactorshowedacoldfingerattheentranceofthereactoroverafew centimetersoriginatingfromtheinjectionofcoldervaporphase (473K).Thestoppingoftheinjectionoftheprecursorsolutioninthe toluenedecreasesthetotalflowrateofthegasphasebyabout30% (about750sccmcomparedto500sccmwithandwithout precur-sorinjectionrespectively)andconsequentlysubstantiallyreduces thelengthofthistemperaturegradientzoneattheentrance.This transienteffectonthetemperatureisnotharmfulsinceitexpends theisothermalzoneofthereactor.
Bycombining theatomic densitymeasuredbyRBSwiththe thicknessofthecoating(2mm)measuredbySEManditsatomic compositionmeasuredbyEPMA,adensityof7.7±0.6g/cm3 was found.Usuallythedensityofcoatingsislowerthanthatofbulk materials. Despite a significant experimental error,the density measuredisofthesameorderofmagnitudeasthatofbulk bcc-Cr(7.19g/cm3).ThisconfirmsthehighdensityofDLI-MOCVDCr
multilayercoatingsgrownaccordingtomode2.
ChemicalanalysesofpreviousMOCVDandDLI-MOCVDcoatings typicallyrevealedalowcarboncontent(from3to8at.%)[14,22,24] andaslightcontaminationbyoxygenbecauseofaresidualoxygen inthereactororimpuritiesintheprecursorandinhibitor. Depend-ingonthegrowthconditions,coatingsalsocontainasmallamount ofchlorine(<1at%)orsulfur(<3at%)originatingfromtheinhibitors ofcarbideformation.
In this work, the two types of coating deposited according to modes 1 (monolayer) and 2 (multilayer) using thiophenol as inhibitor of carbon incorporation have similarcompositions whichareingoodagreementwithpreviousstudies[14,22,24].For instance,EPMAanalysisofmultilayercoatingsdepositedat673and 723Kunderlowpressure(6.7kPa)andusingamoleratio thiophe-nol/BEBCequalsto2%intheinjectedsolutionhavethecomposition Cr0.92C0.04O0.03S0.01.
Fig.3.XRDpatternsofatwo-phasecoatingcomprisingthemetastabled-Crphase andthestablebcc-Crphase(topandblackpattern;multilayercoating)andofastable bcc-Crmonolayercoating(bottomandgreypattern)depositedbyDLI-MOCVDon Sisubstrate,respectivelyat673Kand723K(otherconditions:6.7kPa;C6H5SH).
3.2. Evidenceofthemetastablephaseı-Cr
ForallpreviousMOCVDfilmsdepositedat623and673Kwith C6Cl6asinhibitor,thestablebcc-Crphasewastheonlyone
identi-fiedbyXRD[14,18,19].ThesameresultwasfoundbyDLI-MOCVD usingC6Cl6inthetemperaturerange748–793K[22–24].The
coat-ingsgrownat748Kusingthiophenolasinhibitoralsoexhibita single-phase bcc structure [24]. In the present work, thin film growthbyDLI-MOCVDusingthiophenolat673and723Kallowed toobservetheformationoftwocrystallinephases:thestable bcc-Croneandthemetastabled-Crphasewhichhasacubicstructure (space group Pm-3n) witha larger latticeparameter of 4.59Å. TheXRDpatternofapartiallymetastablecoatinggrownat673K accordingtomode2(multilayers)isshowninFig.3andcompared withapurestablebcc-Crmonolayercoatinggrownat723K(mode 1).
For multilayer coatingsdeposited at 673K, both stable and metastablephasesareidentified,whateverthesubstrateused(Si orstainlesssteel).Thestablebcc-Crphaseshowsapolycrystalline structureconsistingofwellcrystallizedgrainswhoseaveragesize islargeenough(>300nm)nottoinduceameasurablewideningof theXRDpeaks.Onthecontrary,theXRDpeaksofthemetastable d-CrphasehavelargeFWHMwhichreflectsthenanometricaverage sizeofthecrystallites,estimatedat17and28nmusingtheScherrer equationfromthepeaks(211)and(200),respectively.
FromtherelativeintensityofexperimentalXRDpeaks(Ihkl)of
Fig.3comparedtothoseofPDFfilesofrandomlyorientedcoatings (I0
hkl),atexturecoefficient(Thkl)forbothphaseswascalculated
accordingtoThkl=(Ihkl/I0hkl)/[n·S(Ihkl/I0hkl)],wherenisthenumber
ofdiffractionpeaksconsideredinagivenangularrange. Contrar-ilytocoatingsgrowninthemonolayermode,multilayercoatings grownat673Kdonotexhibitpreferentialorientationforthe dom-inantbcc-Crphase;T110=0.9comparedto1.8forthemonolayer
coating.Thisisdirectlytheresultofthemultilayerstructurewhich hindersthecolumnargrowth,decreasestheaveragegrainsizeand subsequentlyleadstoahigherdensityofcoatings.Forthis two-phase coating, the metastable phase d-Cr exhibits a noticeable preferentialorientationinthe[211]directionwithanestimated
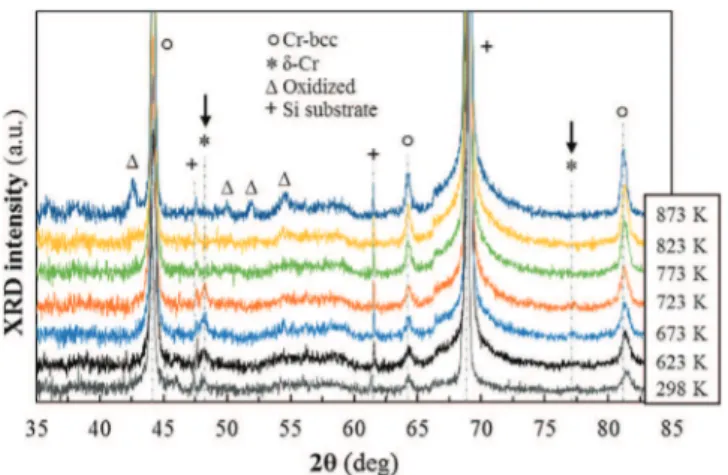
Fig.4. InsituXRDofapartiallymetastablechromiummonolayercoatingat dif-ferenttemperaturesunderAratmosphere(depositionat673Kand6.7kPaonSi substrate).Arrowsshowdisappearanceofthemetastabled-Crphase.Thesample washeatedwithatemperaturerampof1Kmin−1anditwasmaintained35minat
eachtemperaturetorecordthepattern.
texturecoefficientof1.8despitethelowintensityofthediffraction peaks.
TheXRDpeaksofthemetastablephasearealwayslessintense thanthoseofthestablephase,indicatingsmallamounts.Fromthe ratiooftherelativeXRDintensityofthemostintensepeaksofd-Cr (211)andbcc-Cr(110),andsoneglectingthelowtextureofthe metastablephase, acoarseproportionofd-Crwasestimatedat about6%.Thismethodsuffersfromtwoapproximations;(i)the lowtextureofd-Croverestimatestheintensitythatshouldbe con-sideredford-Cr(211)and(ii)thecrude intensityofbcc-Cr(110) isalsooverestimatedbecauseofoverlapwiththed-Cr(210)peak. Interestingly,bothapproximationscompensatewhatmakessense tothemethod.
InsituXRDanalysesallowedustheverificationofthethermal stability,inargonatmosphere,ofthemetastablephaseembedded inthestablebccmatrix.ThearrowsonXRDpatternsofFig.4show thecompletetransformationofthemetastabled-Crphaseintothe stablebcc-Cronebeyond723K,ingoodagreementwithliterature data[25–27].Tracesofchromiumoxideappearabove823K,mainly duetotheresidualoxygenpartial pressurein theXRDanalysis chamber.Onreturningtoroomtemperature,theXRDpatternis thesameasat873Kconfirmingthatthed-Cr→a−Cr transforma-tionisirreversible.FromthisseriesofXRDpatternsrecordedinsitu onadiffractometerdifferentfromthatofFig.3,themean crystal-litesizeestimatedwiththed-Cr(211)peakis21,32and30nmat thetemperatures623,673and723Krespectively.Thesevaluesare consistentwiththatmeasuredatroomtemperaturewithdataof Fig.3.Nosignificantvariationoftheaveragesizeofd-Cr crystal-liteswasfoundforthisheattreatment.Above723K,themetastable phasedisappears.
Noevidenceforthemetastabled-CrphasewasfoundbyTEM analysis,probablybecauseoftheverylowamountofthisphase comparedtothestableone.Anotherhypothesis isthatitcould undergoaphasetransformationundertheTEM200kVelectronic beam. Electron-beam induced phase transformation is a well-known phenomenon as reported in several papers relative to metallicalloys[28],minerals[29]oroxides[30].
Fig. 5 illustrates a TEManalysis of a two-phase monolayer coatingcharacterizedbyacolumnarmorphology.APrecisionIon PolishingSystem(PIPS)wasusedtopreparethesample,resulting inathinsliceofthecoatingapproximatelyparalleltothesurface ofthefilm.ThereforeFig.5ashowsacross-sectionofonecolumn (andapartofanotherone)perpendiculartoitslongitudinalaxis. TheinsertinFig.5bpresentsthecorrespondingelectron
diffrac-tionpattern attributedtothestablebccphase with[011]zone axis.Anexperimentallatticeparameterof2.92Åwasdeducedfrom electrondiffractionpatternsofseveralcrystallitesingood agree-ment, within2%,withthe literaturevalueof bcc-Cr(2.88Å).A high-resolutionpictureinthesameareashowninFig.5crevealsa veryhomogeneouspacking.ItsFouriertransformintheinsert(d) isinagreementwiththeexperimentalpatternin(b).Moreover, columnsobserved by TEMappear polycrystallinein agreement withthefactthatthetextureisnotveryimportantevenifitexists formonolayercoatings(Fig.1b).Thecross-sectionconfirmsthese columnshaveanaveragediameterofabout1mm,asfoundinSEM observations(Fig.1a).
A two-phase multilayer coating comprising the metastable componentwasanalyzedbySEM,coupledtoanEBSDdetector. Itisinfactacross-sectionofthismultilayercoatingpreparedwith across-sectionpolisher(JEOLIB-19510CP)whichwasstudied.An EBSDbandcontrastexampleispresentedonFig.6a.Itissimilarto classicalsecondaryelectronsSEMimages,asshowninFig.2,except thatthecontrastmakesiteasiertoseethegrains.EBSDphase map-pingis showninFig.6b.Thestablebcc-Crandmetastabled-Cr phasesareidentifiedwithtwodistinctcolors,respectivelyblueand red.
EBSDstatisticalanalysiswasperformedonapproximately1000 grains.IdentificationofbothmetastableandstableCrphaseswas possible.Themetastabled-Crphaseproportionisaround3%.This comparesquitewellwiththe6%determinedbyXRDassumingthat bothphaseswerenottextured.Themetastablephaseisessentially foundatinterfacesofthemultilayerandatgrainboundariesofthe dominantbccphasewherethereisthehighestdensityofstructural defectsandinlowEBSDsignalareas.Itwascomplicatedto dis-cernthetwophasesbecausetheyhavethesamecompositionand bothexhibitcubicstructures(bccforthestablephaseandprimitive cubicforthemetastableone).Inthistwo-phasemultilayercoating, theaveragegrainsizeofbcc-Crisapproximately300nmwhileitis onlyafewnanometersford-Cr.Inapreviouswork,Itwasreported thattheparticlesofthismetastabled-Crphasedidnotexhibita definitecrystalshapeandtheiraveragesizewaslowerthan20nm [26].
NospecificgrainorientationwasobservedbyEBSDanalysisfor thestablebccphase,asitcanbeseenontheinversepolefigurein Fig.6c.Thisconfirmsthatnopreferentialorientationofthegrowth occursusingthismultilayerCVDmode.Thisresultisinverygood agreementwithXRDanalysiswhichshowedtheabsenceoftexture forthestablephasewithatexturecoefficientof0.9forthe[110] direction(Fig.3).
XPSanalyseswereperformedtostudytheenvironmentofatoms andanalyzethenatureofimpuritiesinthesecoatings.Asurvey scanafterthesurfacecleaningbyAr+sputtering,reportedas
sup-plementaryinformation,revealsthepresenceofonlyCr,O,Cand S(Fig.S1).Itisnotpossibletodistinguisha-Crfromd-Cr.Fig.7 presentsXPSregionspectraofCr2p,O1s,C1sandS2plevelsfrom atwo-phasemultilayerCrcoatingdepositedonaSisubstrate.Three differentsputteringtimesaredisplayedforeachelectroniclevel: 0s(as-deposited),120sand2012s.As-depositedsampleexhibits asurfacecontaminationcontainingchromiumoxides(Cr2p3/2at
576.6eVandO1sat530.8eV),adventitiouscarbon(C1sat284.8 and288.3eV)andsulfateanions(S2p3/2at168.7eV).
Forthelongestcleaningtimebysputtering,thiscontaminated surfaceissufficientlycleanedanddataaremorerepresentativeof thecoatingcomposition.ThemaincontributionofCr2p3/2isshifted
to574.0eVwhichischaracteristicofmetalCr,evenifanoverlap existswithasmallcontributionduetoCr Cbonds(Fig.7a).Traces ofoxygenleadtoaresponseofCr Obondsasashoulderat576.6eV forCr2p3/2andacontributiontoO1sat530.8eV(Fig.7b).This
oxy-genwasattributedtoaslightoxidationofthecoatingsinagreement withEPMAdata(3at%).Fig.7cshowscarbonincorporatedintwo
Fig.5.(a)TEMobservationofatwo-phasemonolayerCrcoating;(b)correspondingselectedareaelectrondiffractionpattern([011]zoneaxis);(c)Highresolutionmicrograph; (d)correspondingFouriertransformofthehigh-resolutionview.
Fig.6. EBSDanalysisofacross-sectionofatwo-phasemultilayercoatinggrownat673Kand6.7kPaonSisubstrate:(a)bandcontrast;(b)phasemapping;(c)inversepole figure(zaxis).
forms:asfreecarbon(C1sat284.6eV,dominantcontribution)and ascarbide(C1sat282.8eV).Bycombiningtherelativeintensityof freeCandcarbidecomponentswiththetotalCcontentdetermined byEPMA(4at%),theamountofCinthecarbideformisestimated atabout0.8at%.ThisissignificantlyhigherthanCsolubilityinCr whichwasreportedtobe<0.1at%atabout750K[31].
Aftersurfacecleaning,theS2plevel,althoughnotveryintense, givesevidenceforauniqueenvironmentintheformofS Crbonds withS2p3/2shiftedat161.6eV,withashoulderat162.8eVforS
2p1/2(Fig.7d).ThisischaracteristicofmetalsulfideorofS
solubi-lizedinthemetalstructure.Inthissample,totalScontentanalyzed byEPMAwasonly0.5at%whichishigherthanthesolubilityof sulfurinchromiumat673K[32].Consequentlyasulfide
contam-inationlikelyexistsbuttheamountistoolowtobeanalyzedby XRDorTEM.
3.3. Originofthemetastablephaseı-Cr
Themetastablephased-Crwasclearlyidentifyinsmallamount byXRDandEBSDinDLI-MOCVDCrmetalcoatingsdepositedboth inmode1(monolayer)andmode2(multilayer).Itisbetter visi-bleinmultilayercoatingsthaninmonolayerbecausethecolumnar growthand textured structure in thelattermake it difficult to observe,sincetherelativeintensityofd-CrXRDpeaksiscrushed bythatofthepreferentialorientationofa-Cr.Severalfactorsseem toinfluencetheformationofthismetastablephase.
Fig.7. XPSanalysesofatwo-phasemultilayerCrcoatingcomprisingthemetastabled-Crandthestablea-Crphasesdepositedat673K.Spectralregionsof(a)Cr2p,(b)O 1s,(c)C1sand(d)S2parereportedfordifferentAr+sputteringtimes(1keV):0s(as-deposited;blackspectrum,bottom),120s(blue,middle)and2120s(red,top).Anoffset
alongtheYaxishasbeenappliedforclarityandadeconvolutionofthemostrepresentativespectrumofthecoatingisplotted.(Forinterpretationofthereferencestocolour inthisfigurelegend,thereaderisreferredtothewebversionofthisarticle.)
It wasnever reported in Crcoatingsgrown by MOCVD and DLI-MOCVDusing chlorinatedinhibitors[14,17–19,22–24] even fordepositiontemperatureaslow as573K.Whenusing sulfur-containinginhibitorsitwasnotmentionedbyMOCVD[21]norby DLI-MOCVD[24](Table2).Ithasbeenobservedinthisworkby DLI-MOCVDusingC6H5SHasinhibitorofcarbonincorporationonlyfor
depositiontemperature≤723K.Thisisingoodagreementwiththe factthatbeyondthiscriticaltemperatureastructuralphase trans-formationintobcc-Croccurs,asreportedby[25–27]andconfirmed byinsituXRDanalysisinthisstudy(Fig.4).
Whenthedepositioniscarriedoutat723K,theobservation ofthisphasedependsonthepositionofthesamplesina quasi-isothermalreactor,whichmeansitdependsonthelocaldeposition conditions,i.e. mainlyonthe substratetemperature.Numerical modeling(notreportedhere)showedforourDLI-MOCVD reac-torthatwhenthetemperatureofthefurnaceissetto723K,the substratetemperaturewasapproximately50Klower,i.e.around 673K.Thistemperaturedecreasecomesfromofthepresenceofa coldfingerattheentranceofthereactoroverseveralcentimeters intheaxialdirection[33].Thisisduetothelowertemperatureof thereactivegasphase(approximately473K)comingfromtheflash vaporizationchamberwhichentersthereactor.Consequently,the formationofthemetastablephaseisthenpossiblebecausethe sub-stratetemperatureislowerthanthecriticalvaluebeyondwhich thestructuraltransformationoccurs.Forthesamefurnace
tem-perature(723K),butusinganothergeometryandsizeoftheCVD reactordifferenttransportconditionshavetobeusedleadingto differenttemperaturefields.Consequently,thetemperature dif-ferencebetweenthesubstrateand thefurnaceset-pointwillbe differentthaninthisworkandthemetastablephasemaynotbe observedifthereactorismoreisothermal.Thisisconsistentwitha sharpstructuralphasetransformationatthiscriticaltemperature of723K.So,forthedepositionofmetallicCrcoatingscontaining d-Crasaminorphase,DLI-MOCVDisasuitableprocess.The sub-stratetemperaturemustbelowerthan723KandC6H5SHisthe
bestinhibitor.
Structuralphasetransformationinmetalsinducedby impuri-tiesisacommonphenomenon[34].Moreover,magneticordering of chromium is affectedby impurities (doping) which induces changesinphasetransition[35].Thiophenollikelypromotesthe formationofthemetastablephase.Sulfurimpuritiesare incorpo-ratedinthecoating(<1at.%)andtheycouldinducethestructural phasetransformationwithinCrcoatings.Thiophenolactionwas foundefficientstartingfromamoleratiothiophenol/BEBCof2% (thetestedvalueswereintherange1–10%).Foramoleratioof only1%,noinhibitoryactionwasobservedsincetheformationof chromiumcarbidesoccurredinsteadofmetallicchromium.
TherelativelylargeFWHMofXRDpeaksofd-Crphasetogether withtheEBSDanalysisgivesevidencethatthismetastablephase ispresentintheformofnanocrystallites.Thenanometricsizeof
d-Crparticlesmaybeanothercauseofitsstability.Indeedsucha nanometriceffectisknownforinstancetostabilizethemetastable anataseform of titania relative torutile although this last one isthermodynamicallythemoststablephase.Thisisbecausethe surfaceenergyofanataseissignificantlylowerleadingtostable anatasenanoparticlesbelowacriticalsizeofabout15nm[36].Our observationofd-Cronlyintheformofnanoparticlesisconsistent with[26]whereauthorsalsoreportedd-Crasnanoparticles,which couldbeexplainedbyalowersurfaceenergythanthatofbcc-Cr.
Asmentionedabovethed-Crphaseismoreeasilyseenin multi-layercoatingsthaninthemonolayercoatings.Furthermore,EBSD analyseshaveshownthatitislocatedbothattheinterfacesand grainboundaries(Fig.6).Fromfundamentalsof nucleationand crystalgrowthitcanbeassumedthatthemetastablephase depo-sitioniscontrolledbyaheterogeneousnucleationstepratherthan asteadystategrowthregimebecausethesurfaceseemstoplay animportantroleanditscrystallitesizestayslimitedatthe nano-metricscale.Thishypothesissuggeststhatitsproportionwould increasewiththenumberofgrainboundariesandinterfacesas obtainedinthemultilayercoatings.Sobyincreasingthe nanos-tructurationwhichmeansbyincreasingthenumberofindividual layersinthecoating,thenumberofbothgrainboundaries(dueto loweraveragecrystallitesize)andinterfaceswillincrease,which couldfavorthegrowthofd-Cr.Thisideahasnotyetbeenverified. FromtheXRDpatternofFig.3,theproximityofthemostintense peaksofeachphase suggeststhatanepitaxialrelationshipmay existwhichwouldfavorthenucleationofd-Cr(211)planeon a-Cr(110)andwouldexplainthepreferentialorientationofd-Crin the[211]direction.
3.4. Unusualproperties
These DLI-MOCVD Cr coatings are promising candidates as protectivemetallurgicalcoatings.Theyaredepositedatlow tem-perature(T<723K)witharelativelyhighgrowthrate(∼5mm/h). The multilayer coatings exhibit a density as high as that of bulkmetallicCr(7.19g/cm3).Forapplicationsinharsh
environ-ments,iftheoperatingtemperatureincidentallyexceeds723K,the metastablephasewillbeirreversiblytransformedintothestable bccphase(Fig.4).Accordingtotheircrystallographicstructures,the metastabled-Crphaseisslightlylessdensethana-Cr(7.16g/cm3).
Thismeansthatatthetimeofthephasetransformationtheoverall volumecontractionwillbeonly0.4%.Furthermoretheproportion ofd-Crdoesnotexceed6%inmultilayercoatingsasdeducedfrom XRDandEBSDanalyses.Thisindicatesthatitcanactasatracer likely withoutloss of thebasicproperties of thecoatings.This isparticularlyusefulinnon-destructivetestingofmanufactured structuralcomponentsprotectedwithsuchacoating.
Indeed carbon-steels and alloys undergo specific heat treatments to optimize their mechanical properties (ductility, hardness...) and theiroperating temperaturemust not exceed the temperatureof the last treatmentundergone as hardening ortemperingannealing.Beyondthecriticaltemperatureasteel structurecannotcarrytheserviceloadforwhichitwasdesigned. Thephasetransformationofd-Croccursinthetemperaturerange oftheheattreatmentsofmanysteelsandmetallicalloys,asfor instanceZircaloy-4 usedin nuclearindustrywhich willloseits metallurgicalstateascladdingmaterialabove753K[37].IfaCr coatingisdepositedbyDLI-MOCVDonthisalloy,the disappear-anceofthemetastablephased-Crwillrevealincidentalconditions inservicegreaterthan723Kwhichwillalsoaffecttheproperties of thezirconiumalloy.Obviously, theexposuretime alsoplays animportantroleinthisd-Cr/a-Crtransformation,butakinetic studyofthistransformationwasnottheprimarypurposeofthis paperanditwillbeinvestigatedshortly.
Preliminary results of mechanical properties were obtained usingananoindenter(Nanoscratchtester,CSMinstrument)for monolayer and multilayercoatings deposited onsteel at 673K usingC6H5SHasinhibitor.ThedataarereportedinTableS1
(Sup-plementary material) and are compared to previous values for similarcoatings.The nanohardness ofa columnar 3.5mmthick monolayercoatingis9.7GPa.Thisisconsistentwiththe13.0GPa found for columnarmonolayer coatingsdeposited at thesame temperaturewithC6Cl6asinhibitor[18].Moreinterestingfor
tech-nologicalapplicationisthehighernanohardnessof16.9GPafound for a 5.5mm thickmultilayer coatingthat exhibitsa high den-sity(depositedaccordingmode2).WhenCrcoatingsgrownby MOCVDandDLI-MOCVDusingC6Cl6 asinhibitoraresufficiently
dense,theyalsoexhibitacomparablehighnanohardnessof19.0 [18]and17.0GPa[22],respectively.Whatevertheinhibitorused (C6Cl6 orC6H5SH)densecoatingshaveanelasticmodulusinthe
range270–310GPawhichisclosetothe285GPaofbulkCr. Further-moretheyallexhibitacompressiveresidualstresscloseto0.6GPa (determinedfromthecurvaturechangeofsamples).
Wewillnolongercommentonthesepreliminaryproperties,but therearetwostrengthsthatmustberetained:(i)DLI-MOCVDCr metalcoatingsaremuchharderthanthosedepositedbyother tech-niques,e.g.electrodeposition[1–3]orarcevaporation[8],and(ii) thereisnosignificantdifferenceforMOCVDandDLI-MOCVD coat-ingsdependingonthenatureoftheinhibitorused,whichmeans thatthepresenceofthemetastablephased-Crdoesnot signifi-cantlyaffectmechanicalproperties.Theveryhighhardnessofthe sameorderofmagnitudeasthatofchromiumcarbidesisprobably duetocarbonsupersaturationinCrasrevealedbyXPSanalyses (Fig.7).Thefactthatthemetastablephasewouldnotaffectthe mechanicalpropertiesofthetwo-phasecoatingswouldbedueto thecubicstructureofeachcomponent,theirverysimilardensity andthesmallamountofd-Cr.
4. Conclusions
Alow amount(<6%)ofthecubicmetastabled-Crphase was foundmixedwiththestablebcc-CrphaseinDLI-MOCVDcoatings. Theformationofthismetastablephaseresultsfromboththelow temperatureofdeposition(<723K)andtheuseofthiophenolas inhibitor ofcarbide formation.It wasnot reportedunderother CVDconditions.Densecoatingsweredepositedbyimplementing amultilayergrowthmodeinordertoavoidacolumnarandporous morphology.Suchcoatingsexhibitahighnanohardnessofabout 17GPa.
Accordingtotheliterature,thismetastablephaseisirreversibly transformedintothestablebcconefrom723Kininertatmosphere. Thisstructuraltransformationshouldnotaffectthepropertiesof thecoatingduetothesimilarityoftheircrystallographicstructures (bothcubic)andtheirveryclosedensity(avolumecontractionof only0.4%atthetimeofstructuraltransformation).Thepresence ofthismetastablephaseconstitutesasignatureoftheDLI-MOCVD process.Itcanbeusedasatracerforcoatingsoperatinginhigh temperatureenvironmentwithoutlossofthebasicpropertiesof thecoatings.Thisisparticularlyusefulinnon-destructivetesting ofmanufacturedstructuralcomponents.
Acknowledgements
The authors thank Dr. Arnaud Proietti and Marie-Christine LafontfortheirhelpinEBSDandTEManalysesrespectively,and R.Laloofornanohardnessmeasurement.RBSanalysesweremade byH.GuéganinARCANE-CENBG,Gradignan,France.Thisworkwas partiallysupportedbyCNRS,CEA,INPTandtheCentreofExcellence ofMultifunctionalArchitecturedMaterials(CEMAM).
AppendixA. Supplementarydata
Supplementarydataassociatedwiththisarticlecanbefound,in theonlineversion,atdoi:10.1016/j.apsusc.2017.05.253.
References
[1]S.Hoshino,H.A.Laitinen,G.B.Hoflund,Theelectrodepositionandproperties ofamorphouschromiumfilmspreparedfromchromicacidsolutions,J. Electrochem.Soc.133(1986)681–685.
[2]N.Imaz,M.Ostra,M.Vidal,J.A.Díez,M.Sarret,E.García-Lecina,Corrosion behaviourofchromiumcoatingsobtainedbydirectandreversepulseplating electrodepositioninNaClaqueoussolution,Corros.Sci.78(2014)251–259. [3]G.A.Lausmann,Electrolyticallydepositedhardchrome,Surf.Coat.Technol.
86–87(Part2)(1996)814–820.
[4]A.Liang,L.Ni,Q.Liu,J.Zhang,Structurecharacterizationandtribological propertiesofthickchromiumcoatingelectrodepositedfromaCr(III) electrolyte,Surf.Coat.Technol.218(2013)23–29.
[5]V.S.Protsenko,F.I.Danilov,V.O.Gordiienko,A.S.Baskevich,V.V.Artemchuk, ImprovinghardnessandtribologicalcharacteristicsofnanocrystallineCr-C filmsobtainedfromCr(III)platingbathusingpulsedelectrodeposition,Int.J. Refract.Met.HardMater.31(2012)281–283.
[6]A.Aubert,R.Gillet,A.Gaucher,J.P.Terrat,Hardchromecoatingsdepositedby physicalvapourdeposition,ThinSolidFilms108(1983)165–172.
[7]F.Cosset,G.Contoux,A.Celerier,J.Machet,Depositionofcorrosion-resistant chromiumandnitrogen-dopedchromiumcoatingsbycathodicmagnetron sputtering,Surf.Coat.Technol.79(1996)25–34.
[8]G.Cholvy,J.L.Derep,M.Gantois,Characterizationandwearresistanceof coatingsintheCr-C-Nternarysystemdepositedbyphysicalvapour deposition,ThinSolidFilms126(1985)51–60.
[9]S.Komiya,S.Ono,N.Umezu,Hardnessandgrainsizerelationsforthick chromiumfilmsdepositedbyhallowcathodedischarge,ThinSolidFilms45 (1977)473–479.
[10]H.Högberg,L.Tengdelius,M.Samuelsson,J.Jensen,L.Hultman,b-Taanda-Cr thinfilmsdepositedbyhighpowerimpulsemagnetronsputteringanddirect currentmagnetronsputteringinhydrogencontainingplasmas,Phys.B: Condens.Matter439(2014)3–8.
[11]R.G.I.Leferink,W.M.M.Huijbregts,Chromiumdiffusioncoatingsforthe protectionoflow-alloysteelinasulphidizingatmosphere,Corros.Sci.35 (1993)1235–1242.
[12]V.A.Ravi,Packcementationcoatings,in:S.D.Cramer,B.S.CovinoJr.(Eds.), ASMHandbookVolume13A:Corrosion:Fundamentals,Testing,and Protection,ASMInternational,2003,2017,pp.763–771.
[13]A.R.Castle,D.R.Gabe,Chromiumdiffusioncoatings,Int.Mater.Rev.44(1999) 37–58.
[14]F.Maury,L.Gueroudji,C.Vahlas,Selectionofmetalorganicprecursorsfor MOCVDofmetallurgicalcoatings:applicationtoCr-basedcoatings,Surf.Coat. Technol.86–87(Part1)(1996)316–324.
[15]A.Michau,F.Maury,F.Schuster,R.Boichot,M.Pons,E.Monsifrot,Chromium CarbideGrowthatLowTemperaturebyaHighlyEfficientDLI-MOCVDProcess inEffluentRecyclingMode,Surf.Coat.Technol.(2017),submittedfor publication.
[16]F.Maury,F.Ossola,Evaluationoftetra-alkylchromiumprecursorsfor organometallicchemicalvapordepositionI:Filmsgrownusing Cr[CH2C(CH3)3]4,ThinSolidFilms207(1992)82–89.
[17]V.B.Polikarpov,A.S.Luzin,V.A.Dodonov,E.K.Klement,Chromiumfilms obtainedbypyrolysisofchromiumbisarenecomplexesinthepresenceof chlorinatedhydrocarbons,IzvestiyaAkademiiNaukSSSR20(1984) 1839–1842.
[18]F.Maury,C.Vahlas,S.Abisset,L.Gueroudji,Lowtemperaturemetallorganic ChemicalVaporDepositionroutestochromiummetalthinfilmsusing bis(benzene)chromium,J.Electrochem.Soc.146(1999)3716–3723. [19]F.Maury,F.-D.Duminica,F.Senocq,NovelMOCVDprocessforthelow
temperaturedepositionofthechromiumnitridephases,in:M.D.Allendorf, M.L.Hitchman(Eds.),CVDXV:ProceedingsoftheFifteenthInternational SymposiumonChemicalVaporDeposition,TheElectrochemicalSociety, Pennington,NJ,2000,pp.260–267.
[20]C.Vahlas,F.Maury,L.Gueroudji,AthermodynamicapproachtotheCVDof chromiumandofchromiumcarbidesstartingfromCr(C6H6)2,Chem.Vap.
Depos.4(1998)69–76.
[21]A.S.Luzin,V.B.Polikarpov,V.A.Dodonov,E.K.Klement,Chromiumfilms obtainedbypyrolysisofbis(arene)chromiumcomplexesinpresenceof sulfur-containingadditives,Zh.Prikl.Khim.61(1988)1235–1239. [22]F.Maury,A.Douard,S.Delclos,D.Samelor,C.Tendero,Multilayerchromium
basedcoatingsgrownbyatmosphericpressuredirectliquidinjectionCVD, Surf.Coat.Technol.204(2009)983–987.
[23]A.Douard,C.Bernard,F.Maury,Thermodynamicsimulationofatmospheric DLI-CVDprocessesforthegrowthofchromium-basedhardcoatingsusing bis(benzene)chromiumasmolecularsource,Surf.Coat.Technol.203(2008) 516–520.
[24]G.Boisselier,F.Maury,F.Schuster,Growthofchromiumcarbideinahotwall DLICVDreactor,J.Nanosci.Nanotechnol.11(2011)8289–8293.
[25]K.Kimoto,I.Nishida,Anelectrondiffractionstudyonthecrystalstructureofa newmodificationofchromium,J.Phys.Soc.Jpn.22(1967)744–756. [26]I.Nishida,K.Kimoto,Crystalhabitandcrystalstructureoffinechromium
particles:anelectronmicroscopeandelectrondiffractionstudyoffine metallicparticlespreparedbyevaporationinargonatlowpressures(III),Thin SolidFilms23(1974)179–189.
[27]J.Forssell,B.Persson,Growthandstructureofthinchromiumfilms condensedonultra-highvacuumcleavedNaClandKClcrystals,J.Phys.Soc. Jpn.29(1970)1532–1545.
[28]J.Reyes-Gasga,G.R.Garcia,M.Jose-Yacaman,Electron-beam-induced structuretransformationofthequasicrystallinephasesoftheAl62Cu20Co15Si3
alloy,Radiat.Phys.Chem.45(1995)283–291.
[29]K.Yin,Y.Xia,Z.Liu,J.Yin,L.Sun,Electron-beaminducedphasetransformation inb-Ag2Sethinfilms,Phys.StatusSolidi(a)209(2012)135–138.
[30]U.Golla-Schindler,G.Benner,A.Orchowski,U.Kaiser,Insituobservationof electronbeam-inducedphasetransformationofCaCO3toCaOviaELNESat
lowelectronbeamenergies,Microsc.Microanal.20(2014)715–722. [31]W.D.Klopp,Recentdevelopmentsinchromiumandchromiumalloys,JOM21
(1969)23–32.
[32]J.Oudar,N.Barbouth,Solubilityofsulphuriniron-chromiumalloys,Scr. Metall.15(1981)41–43.
[33]A.Michau,F.Maury,F.Schuster,I.Nuta,R.Boichot,M.Pons,Chromium carbidegrowthbydirectliquidinjectionchemicalvapordepositioninlong andnarrowtubes,experiments,modelingandsimulation,submittedfor publication.
[34]R.G.Hennig,D.R.Trinkle,J.Bouchet,S.G.Srinivasan,R.C.Albers,J.W.Wilkins, Impuritiesblockthe[alpha]to[omega]martensitictransformationin titanium,Nat.Mater.4(2005)129–133.
[35]R.S.Fishman,S.H.Liu,Effectofimpuritiesonthemagneticorderingin chromium,Phys.Rev.B45(1992)12306–12318.
[36]D.A.H.Hanaor,C.C.Sorrell,Reviewoftheanatasetorutilephase transformation,J.Mater.Sci.46(2011)855–874.
[37]S.Fourgeaud,J.Desquines,M.Petit,C.Getrey,G.Sert,Mechanical characteristicsoffuelrodcladdingsintransportconditions,Packag.Transp. StorageSecur.Radioact.Mater.20(2009)69–76.