Publisher’s version / Version de l'éditeur:
Vous avez des questions? Nous pouvons vous aider. Pour communiquer directement avec un auteur, consultez la
première page de la revue dans laquelle son article a été publié afin de trouver ses coordonnées. Si vous n’arrivez pas à les repérer, communiquez avec nous à PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca.
Questions? Contact the NRC Publications Archive team at
PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca. If you wish to email the authors directly, please see the first page of the publication for their contact information.
https://publications-cnrc.canada.ca/fra/droits
L’accès à ce site Web et l’utilisation de son contenu sont assujettis aux conditions présentées dans le site LISEZ CES CONDITIONS ATTENTIVEMENT AVANT D’UTILISER CE SITE WEB.
Internal Report (National Research Council of Canada. Division of Building
Research), 1968-03-01
READ THESE TERMS AND CONDITIONS CAREFULLY BEFORE USING THIS WEBSITE.
https://nrc-publications.canada.ca/eng/copyright
NRC Publications Archive Record / Notice des Archives des publications du CNRC :
https://nrc-publications.canada.ca/eng/view/object/?id=5921117d-f194-48b8-b00c-88250d5efb93 https://publications-cnrc.canada.ca/fra/voir/objet/?id=5921117d-f194-48b8-b00c-88250d5efb93
NRC Publications Archive
Archives des publications du CNRC
For the publisher’s version, please access the DOI link below./ Pour consulter la version de l’éditeur, utilisez le lien DOI ci-dessous.
https://doi.org/10.4224/20377505
Access and use of this website and the material on it are subject to the Terms and Conditions set forth at
The atmospheric corrosion behaviour of three structural steels in eight
Canadian atmospheres: summary of ten years' results
NATIONAL RESEARCH COUNCIL O F CANADA DIVISION O F BUILDING RESEAR CH
THE ATMOSPHERIC CORROSION BEHAVIOWK O F T H R E E STRUCTURAL S T E E L S IN EIGHT CANADIAN ATMOSPHERES-
-
SUMMARY O F T E N YEARS' RESULTS
D.
B.
Clay and J. M. Tudhope, T h e S t e e l Company of Canada, L i m i t e d ,Hamilton, O n t a r i o
and
ANALYZED
v E. V. Gibbons,
Division of Building R e s e a r c h ,
National R e s e a r c h Council of Canada, Ottawa, Canada
A joint r e p o r t of t h e S t e e l Company of Canada, L i m i t e d and t h e Division of Building R e s e a r c h , National R e s e a r c h
Council of C a n a d a I n t e r n a l R e p o r t No. 3 5 7 of the Division of Building R e s e a r c h OTTAWA M a r c h 1968
P R E F A C E
T o fulfil t h e need f o r m o r e published i n f o r m a t i o n on the c o r r o s i o n b e h a v i o u r of d i f f e r e n t a r c h i t e c t u r e m e t a l s a t v a r i o u s l o c a t i o n s a c r o s s Canada, a n outdoor m e t a l s e x p o s u r e p r o g r a m w a s u n d e r t a k e n by t h e f o r m e r A s s o c i a t e C o m m i t t e e on C o r r o s i o n R e s e a r c h and P r e v e n t i o n . T h i s i n f o r m a t i o n i s f r e q u e n t l y r e q u e s t e d by t h o s e r e s p o n s i b l e f o r t h e d e s i g n and m a i n t e n a n c e of buildings. It w a s a c o -ope r a t i v e u n d e r t a k i n g involving C a n a d i a n m e t a l s u p p l i e r s , u s e r s and r e s e a r c h o r g a n i z a t i o n s . The f i r s t m e t a l s w e r e s e l e c t e d in 1953 f o r e x p o s u r e a t eight outdoor s i t e s a c r o s s Canada.
The i n i t i a l g r o u p of m e t a l s e x p o s e d included t h r e e t y p e s of s t e e l , t h r e e s t a i n l e s s s t e e l s , two m a g n e s i u m a l l o y s , a r o l l e d zinc, t h r e e
a l u m i n u m a l l o y s and r i v e t t e d galvanic c o u p l e s of a l u m i n u m 35 a l l o y coupled t o zinc, c o p p e r and m i l d s t e e l . In s u b s e q u e n t y e a r s s p e c i m e n s of l e a d a l l o y s , m o n e l , c o p p e r , m u n t z m e t a l and a n u m b e r of d i f f e r e n t m e t a l and o r g a n i c c o a t i n g s applied t o s t e e l s u b s t r a t e s w e r e s e t out a t t h e d i f f e r e n t s i t e s . T h i s r e p o r t d e s c r i b e s t h e 1 0 - y e a r p e r f o r m a n c e of t h e s t e e l s of t h r e e c o m p o s i t i o n s . R e p o r t s c o v e r i n g t h o s e m e t a l s t h a t h a v e c o m p l e t e d t h e i r 10 y e a r s of e x p o s u r e have been i s s u e d . T h e s p e c i m e n s of s t e e l w e r e f u r n i s h e d by t h e S t e e l Company of C a n a d a L i m i t e d , Hamilton, who w e r e a l s o r e s p o n s i b l e f o r a s s e s s i n g t h e i r p e r f o r m a n c e and i n compiling t h i s r e p o r t . T h i s p a r t of t h e s t u d y w a s u n d e r t h e d i r e c t i o n of M r .
D. B .
C l a y , with t h e a s s i s t a n c e of M r .J . M. Tudhope, The a r r a n g e m e n t s f o r e x p o s u r e and e x a m i n a t i o n of s p e c i m e n s , and s o m e o v e r - a l l c o - o r d i n a t i o n of t h e p r o g r a m w e r e c a r r i e d out u n d e r t h e d i r e c t i o n of M r . E. V. Gibbons of t h e Division of Building R e s e a r c h .
T h e D i v i s i o n i s i n d e b t e d , not only t o t h o s e c o m p a n i e s who c o - o p e r a t e d i n making t h i s study p o s s i b l e , but a l s o t o t h o s e who have c o n t r i b u t e d i n t h e p r o v i s i o n and s e r v i c i n g of c e r t a i n of t h e e x p o s u r e s i t e s . Ottawa M a r c h
1968
N.
B . Hutcheon A s s i s t a n t D i r e c t o rTHE ATMOSPHERIC CORROSION BEHAVIOUR O F T H R E E STRUCTURAL S T E E L S IN EIGHT CANADIAN ATMOSPHERES-
-
SUMMARY O F T E N YEARS1 RESULTS
D. B. Clay a n d J. M. Tudhope, The S t e e l Company of Canada, L i m i t e d ,
H a m i l t o n , O n t a r i o
and
E. V. Gibbons,
Division of Building R e s e a r c h ,
N a t i o n a l R e s e a r c h Council of Canada, Ottawa, C a n a d a
Outdoor e x p o s u r e of v a r i o u s a r c h i t e c t u r a l m e t a l s a t C a n a d i a n s i t e s w a s u n d e r t a k e n by t h e A s s o c i a t e Comlzlittee on C o r - r o s i o n R e s e a r c h a n d P r e v e n t i o n of t h e N a t i o n a l R e s e a r c h C o u n c i l of Canada. T h e p u r p o s e w a s t o p r o v i d e a b e t t e r u n d e r s t a n d i n g of t h e b e h a v i o u r of m e t a l s and m e t a l c o a t i n g s e x p o s e d f o r long d u r a t i o n s in v a r i o u s a r e a s of Canada. T h e e x p o s u r e s i t e s u s e d w e r e s e t up a n d
o p e r a t e d by t h e Division of Building R e s e a r c h of t h e Council. T h e f i r s t g r o u p of m e t a l s e x p o s e d , in a d d i t i o n t o t h e t h r e e s t r u c t u r a l s t e e l s b e i n g r e p o r t e d on a t t h i s t i m e , included s p e c i n l e n s oi a l u i n i n u m , and
r i v e t t e d g a l v a n i c c o u p l e s of 3 s a l u m i n u m a l l o y t o c o p p e r , zinc a n d s t e e l , t h r c e s t a i n l e s s s tee1 a l l o y s , two magnesiurm al.1 oys, r o l i e d zinc, two l e a d a l l o y s , m o n e l c o p p e r and m u n t z m e t a l . T h e s t e e l s p e c i n l e n s w e r e i u r n i s h e d by t h e S t e e l C o m p a n y of C a n a d a L i m i t e d anci w e r e s e t out f o r e x p o s u r e in 1953. T h i s r e p o r t d e s c r i b e s t h e 1 0 - y e a r p c r f o r - m a n c e of t h e s e m a t e r i a l s and g i v e s a b r i e f d e s c r i p t i o n of the t e s t i n g p r o c e d u r e a n d t e s t i n g s i t e s . A m o r e d e t a i l e d d e s c r i p t i o n of t e s t i n g p r o c e d u r e a n d c o r r o s i o n s i t e s w a s p u b l i s h e d e a r l i e r (1, 2 , 3, 4 ) . TESTING P R O C E D U R E FOR S T E E L M A T E R I A L S T h e s t e e l m a t e r i a l s , f r o m n o r m a l p r o d u c ion, had b e e n h o t r o l l e d and pickled. The pickling o p e r a t i o n w a s p e r l o r n l e c i on a con- t i n u o u s s u l p h u r i c a c i d p i c k l i n g line. Then 4-in. by
6-32,
s a m p l e s w e r e s h e a r e d off, identified by d r i l l i n g 1 / 8 in. h o l e s t h r o u g h t h e s a m p l e s , a n dweighed on an a n a l y t i c a l b a l a n c e p r i o r to s h i p m e n t to t h e i r a p p r o - p r i a t e e x p o s u r e site.
E a c h m a t e r i a l was exposed in t r i p l i c a t e f o r p e r i o d s of one, two, f i v e and ten y e a r s a t each c o r r o s i o n s i t e on s p e c i a l l y con-
s t r u c t e d f r a m e s which held the s a m p l e s in p l a c e with p o r c e l a i n i n s u l a t o r s a t 30" to the h o r i z o n t a l and facing due south.
After completing the p r e s c r i b e d e x p o s u r e p e r i o d the s a m p l e s w e r e photographed, d e s c a l e d and reweighed. S e s c a l i n g was a c c o m p l i s h e d in a molten c a u s t i c bath containing 1 t o 2 p e r c e n t s o d i u m hydride. One t e n - y e a r s a m p l e of e a c h s t e e l composition was withheld f o r m i c r o s c o p i c examination.
TEST SITES
Seven t e s t s i t e s of the National R e s e a r c h Council and
one s i t e of Consolidated Mining and Smelting Company of Canada, Limited, n e a r T r a i l w e r e u s e d f o r t h e s e e x p o s u r e s . A b r i e f d e s c r i p t i o n of the location and c l a s s i f i c a t i o n of e a c h s i t e follows.
S i t e No. 1, Ottawa
-
T h i s s i t e i s l o c a t e d on the e a s t e r l y c i t y l i m i t s and c a n be c l a s s i f i e d a s s e m i - r u r a l .S i t e No. 2, Saskatoon
-
T h i s i s a r u r a l s i t e located on the c a m p u s of t h e Univ.ersity of Saskatchewan on t h e o u t s k i r t s of Saskatoon.S i t e No. 3, M o n t r e a l
-
T h i s is a n i n d u s t r i a l s i t e located on t h e roof of a CNR S t o r e s building in the P o i n t St. C h a r l e s d i s t r i c t .S i t e No. 4, Halifax
-
This i s a m a r i n e - i n d u s t r i a l s i t e l o c a t e d on the roof of a F e d e r a l Public building in t h e downtown a r e a , two c i t y b l o c k s f r o m the h a r b o u r .S i t e No. 5, York Redoubt
-
This i s a r u r a l - m a r i n e site, located a p p r o x - i m a t e l y 7 m i l e s f r o m Halifax. I t i s a t an elevation of about 100 f t and 300 f t f r o m t h e ocean.S i t e No.
6,
N o r m a n Wells-
This i s a f a r n o r t h e r n s i t e located in t h e M a c k e n z i e R i v e r Valley a p p r o x i m a t e l y 9 0 m i l e s south of t h e A r c t i c C i r c l e .S i t e No. 7, E s q u i m a l t , B, C.
-
T h i s i s a r u r a l - m a r i n e s i t e l o c a t e d a t t h e s o u t h e a s t e x t r e m i t y of Vancouver Island, 15 m i l e s f r o m t h e c i t y of V i c t o r i a . I t is a t a n e l e v a t i o n of 50 f t a n d a p p r o x i m a t e l y 1500 f t f r o m t h e ocean. S i t e No. 8, T r a i l , B. C.-
T h i s i s a s e m i - r u r a l s i t e l o c a t e d a t B i r c h b a n k i n t h e C o l u m b i a R i v e r Valley6
m i l e s n o r t h of t h e c i t y of T r a i l , I n a d d i t l o n t o m e t e o r o l o g i c a l d a t a a v a i l a b l e f r o m D e p a r t m e n t of T r a n s p o r t w e a t h e r s t a t i o n s n e a r t h e s i t e s , t h e s u l p h u r d i o x i d e (SO ) c o n t e n t of t h e a t m o s p h e r e w a s d e t e r m i n e d , u s i n g t h e l e a d 2 p e r o x i d e m e t h o d . T h i s m e t h o d d o e s n o t m e a s u r e t h e v o i u m e t r l c c o n - c e n t r a t i o n of SO2 i n t h e a t m o s p h e r e d i r e c t l y , b u t p r e s e n t s an i n t e g r a t e d m e a s u r e of t h e SO ' ' a c t i v i t y " d u r i n g a p e r i o d o l e x p o s u r e . I t was found 2 t h a t t h i s i n s t r u m e n t w a s well s u i t e d to m e a s u r e t h e r e l a t l v e l e v e l s of SO2 a t t h e e x p o s u r e s i t e s when m e t a l s a r e u n d e r t e s t . T h e m e a s u r e - m e n t s w e r e m a d e on a m o n t h l y b a s i s a t e a c h s i t e . T h e r e s u l t s h a v e b e e n c o n d e n s e d a n d t h e a v e r a g c l e v e l s d u r i n g t h e t e n - y e a r e x p o s u r e of t h e s t e e l s p e c i m e n s a r e given in T a b l e I. F i g u r e 1 i s a g r a p h i c a l r e - p r e s e n t a t i o n of t h e a v e r a g e c o n c e n t r a t i o n of SO a t e a c h t e s t s i t e . 2 The A. S. T . M . c o r r o s i v e n e s s i n d e x (5,6)
w a s d e r i v e d b y ex;)tislng : r ~ i ? r l s t e e l s a m p l e s a t s i t e s a c r o s s C a n a d a a n d U. S , A., n l e a s u r j n g t h e m a g n i t u d e of c o r r o s i o n p r o d u c t s lorme,cl a f t e r a s i \ - e n t i m e on e a c h s a m p l e a n d e x p r e s s i n g t h e a m o u n t of c o r r o s i o n ~ v i t h r e l a t i o n t o s i m i l a r s a m p l e s e x p o s e d a t S t a t e College, Pa., v:hich w e r e taken a s b e i n g unity. T h e A.S. T.M. i n d i z c ~ s f o r the s i t e s u s e d in t h i s p r o g r a m m e a r e a s follows: H a l i f a x ( m a r i n e - i n d u s t r i a l ) H a l i f a x ( m a r i n e - r u r a l ) M o n t r e a l T r a i l OttawaS a s k a t o o n
-
0.6
N o r m a n Wells
-
0. 03As a n e x a m p l e , the i n d i c e s show t h a t Halifax ( m a r in e - i n d u s t r i a l ) i s
127 t i m e s m o r e c o r r o s i v e than N o r m a n Wells a f t e r one y e a r of e x p o s u r e .
MATERIALS T E S T E D
S t e e l s of t h r e e c o m p o s i t i o n s w e r e s e l e c t e d and g r o u p e d a s low carbon, low c a r b o n c o p p e r - b e a r i n g , a n d low a l l o y c o p p e r - n i c k e l - b e a r i n g . T h e s e s t e e l s w e r e d e s i g n a t e d a s A3, Aq, a n d A5, r e s p e c t i v e l y .
T a b l e I 1 g i v e s t h e c h e m i c a l c o m p o s i t i o n of t h e s e t h r e e s t e e l s .
RESULTS
T h e r e s u l t s of the c o m p l e t e t e n - y e a r t e s t p e r i o d f o r t n e t h r e e s t e e l m a t e r i a l s h a v e been c o m p i l e d in t h e Appendix. The weight l o s s , c o r r o s i o n r a t e , and t h i c k n e s s l o s s v a l u e s a r e inciuded.
B a r g r a p h s showing the c o r r o s i o n r a t e s a f t e r e a c h
c o r r o s i o n p e r i o d f o r the s t e e l m a t e r i a l a t t h e e i g h t c o r r o s i o n s i t e s a r e shown in F i g u r e 2. C o r r o s i o n r a t e s h a v e d e c r e a s e d s i g n i f i c a n t l y on a l l the s t e e l s a t all c o r r o s i o n s i t e s e x c e p t N o r m a l Wells, w h e r e t h e a m o u n t of c o r r o s i o n even a f t e r ten y e a r s i s v e r y s m a l l .
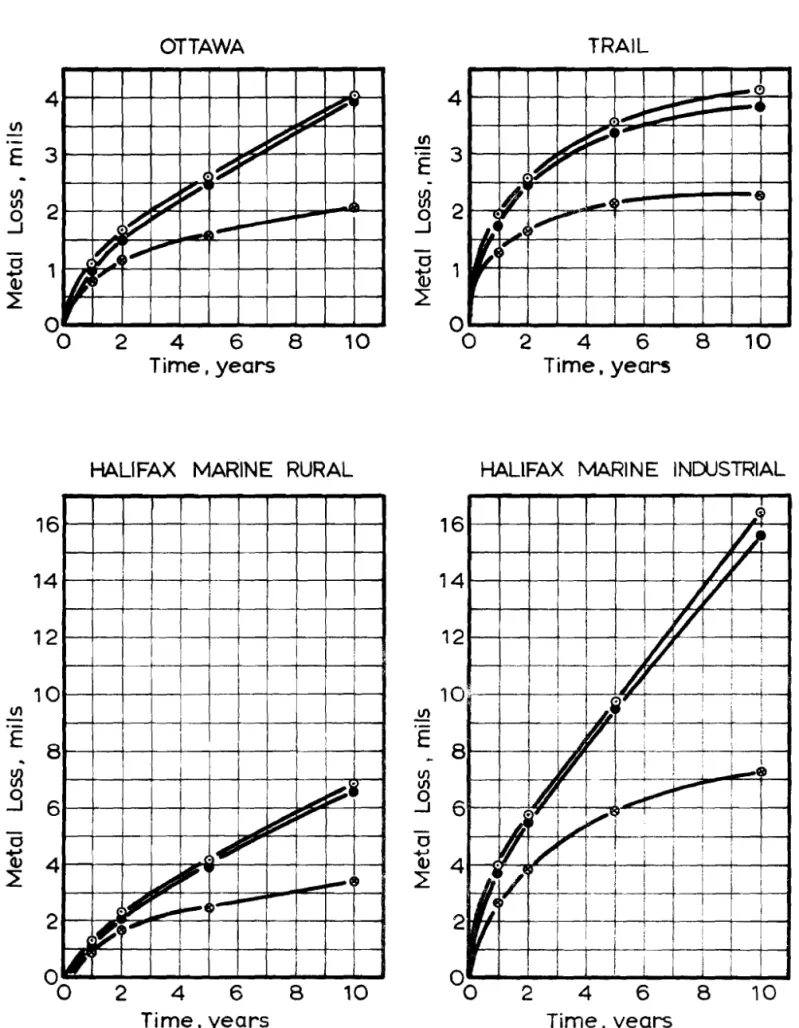
C o r r o s i o n v s , t i m e g r a p h s f o r t h e t h r e e s t e e l s at e a c h t e s t s i t e a r e shown m F i g u r e
3.
S a n l p l e s e x p o s e d a t t h e Halifax : m a r m e - i n d u s t r i a l s i t e s u f f e r e d t h e g r e a t e s t a m o u n t of c o r r o s i o n and t h o s e a t N o r m a n W e l l s the l e a s t . T h e c o r r o s i o n r a t e c u r v e s of t h e i o x ~ c a r b o n and low c a r b o n c o p p e r - b e a r i n g m a t e r i a l s show only a s l i g h t d i f f e r e n c e in c o r r o s i o n r a t e s a t e a c h of the eight t e s t s i t e s .F i g u r e s 4,
5,
and6
show t h e c o r r o s i o n r a t e c u r v e s of e a c h type of s t e e l a t t h e e i g h t c o r r o s i o n s i t e s . At t h e enci of the t e n - y e a r period, t h e s l o p e of t h e c u r v e s f o r t h e t h r e e m a r i n e s i t e s is s t e e p e r than t h a t f o r the o t h e r s i t e s . T h e c u r v e s f o r t h e low c a r b o n and the c o p p e r - b e a r i n g s t e e l s a t t h e Ottawa s i t e do n o t follow t h e s a m e p a t t e r n a s t h o s e f o r the o t h e r n o n - m a r i n e s i t e s . T h i s s i t e m a y have u n d e r g o n e a change in c o r r o s i v e n e s s d u r i n g the t e s t p e r i o d .T a b l e I11 g i v e s t h e a v e r a g e c o r r o s i o n r a t e in m i l s p e r y e a r b e t w e e n t h e fifth and t e n t h y e a r f o r t h e t h r e e s t e e l m a t e r i a l s . T h e a v e r a g e c o r r o s i o n r a t e f o r the c o p p e r - n i c k e l s t e e l a t S a s k a t o o n i s o m i t t e d f r o m T a b l e I11 a s a n e g a t i v e r e s u l t w a s i n d i c a t e d ; h o w e v e r , f r o m t h e v a l u e s f o r t h e o t h e r two m a t e r i a l s t h e c o r r o s i o n r a t e e x p e c t e d would b e l e s s t h a n 0.01
8
r n i l s p e r y e a r . Weight l o s s r e s u l t s m a y n o t i n d i c a t e t h e e x t e n t of l o c a l i z e d pitting; f o r t h i s r e a s o n v i s u a l examinatiol-1 of d e s c a l e d s a m p l e s w a s u n d e r t a k e n . E x a m i n a t i o n showed t h a t c o r r o s i o n h a d o c c u r r e d u n i f o r m l y o v e r t h e e n t i r e s u r f a c e with t h e u n d e r s u r f a c e a t t a c k e d m o r e s e v e r e l y t h a n t h e u p w a r d s u r f a c e ( 7 ) . No e x c e s s i v e pitting w a s n o t e d a t a n y of t h e e d g e s . R e s u l t s frorrl v i s u a l e x a m i n a t i o n c o n f i r m t h e w e i g h t l o s s r e s u l t s a s to e x t e n t of c o r r o s i o n . F i g u r e 7 shosvs t h e d e s c a l e d u p - w a r d s u r f a c e of t h e c o p p e r - n i c k e l - b e a r i n g s t e e l f r o m t h e v a r i o u s c o r-
r o s i o n s i t e s . T h e c r o s s - s e c t i o n s of t h e c o r r o d e d s t e e l s a m p l e s t a k e n f r o m t h e N o r m a n W e l l s , Ottawa a n d H a l i f a x m a r i n e - i n d u s t r i a l s i t e s a f t e r t e n y e a r s of e x p o s u r e a r e shown in F i g u r e s 8 , 9, a n d 10. T h e s e t h r e e s i t e s r e p r e s e n t t h e r a n g e of c o r r o s i o n e n c o u n t e r e d in t h i s p r o - g r a m m e . V e r y l i t t l e c o r r o s i o n h a s t a k e n p l a c e on a n y 01 t h e s a m p l e s f r o m N o r m a n W e l l s ( F i g u r e 8), a l t h o u g h i t i s e v i d e n t t h a t t h e lolver e x p o s e d s u r - f a c e h a s b e e n a t t a c k e d t n o r e s e v e r e l y t h a n t h e u p p e r e x p o s e d s u r i a c e . Tli.:, a m o u n t of c o r r o s i o n on t h e Ottawa s a m p l e s ( F i g u r e 9 ) i s c o n s i d e r-
a i ~ l y trlorc t h a n 'chat on the s a m p l e s f r o m N o r m a n W e l l s . T h e low c a r b o n and low c a r b o n c o p p e r - b e a r i n g m a t e r i a l s h a v e c o r r o d e d m o r e t h a n t h e c o p p e r - n i c k e l - b e a r i n g m a t e r i a l . T h e m e t a l s u r f a c e s h a v e b e c o m e p i t t e d , c a u s i n g s o m e l o c a l d e c r e a s e in c r o s s - s e c t i o n .F i g u r e 10 s h o w s t h e sn3cjunt of c o r r o s i o n a t the H a i i i a x m a r i n e - i n d u s t r i a l s i t e a f t e r t e n y e a r s of e x p o s u r e . T h e c r o s s - s e c t i o n s of the low c a r b o n and low c a r b o n c o p p e r - b e a r i n g s t e e l s f r o r n t h i s s i t e i l l u s t r a t e t h e c o n s i d e r a b l e r e d u c t i o n in t h i c k n e s s o f t h e s e s a n l p l e s c o m p a r e d with t h o s e e x p o s e d a t t h e o t h e r two site:; ( F i g u r e s 8 a n d 9 ) . Comparing the c o p p e r - n i c k e l - b e a r i n g s t e e l e x p o s e d a t H a l i f a x m a r i n e - indl.istria1 s i t e with t h e o t h e r two s i t e s ( F i g u r e s 6 anrl 9 ) it i s o b v i o u s t h a t c o n s i d r : r a b l e pitting h a s o c c u r r e d b u t t h e t h i c k n e s s of tn:s r i ~ a t e r i a l h a s n o t d e c r e a s e d n e a r l y a s m u c h a s t h e t h i c k n e s s of tht: iow c a r n o n o r lolv c a r b o n c o p p e r - b e a r i n g m a t e r i a l s .
DISCUSSION
The c o r r o s i o n r a t e c u r v c s f o r t h e low c a r b o n a n d the low c a r b o n c o p p e r - b e a r i n g m a t e r i a l s show only a s l i g h t d i f f e r e n c e in r a t e s a t e a c h of t h e e i g h t t e s t s i t e s . T h e c h e m i c a l c o m p o s i t i o n of the two m a t e r i a l s i s v e r y s i m i l a r e x c e p t t h a t t h e low c a r b o n m a t e r i a l c o n - t a i n s 0.090 p e r c e n t c o p p e r a n d t h e low c a r b o n c o p p e r - b e a r i n g m a t e r i a l c o n t a i n s 0.205 p e r c e n t c o p p e r . D.M. Buck found a t t h e t u r n of the c e n t u r y t h a t s m a l l a d d i t i o n s of c o p p e r g r e a t l y influt,nit-cl the c o r r o s i o n r e s i s t a n c e of s t e e l . T h e m o r e r e c e n t w o r k s of Hudson a n d S t a n n e r s
( 8 )
a n d L a r a b e e and C o b u r n( 9 )
s u b s t a n t i a t e t h e s e e a r l i e r f i n d i n g s . L a r r a b e e and C o b u r n found t h a t t h e g r e a t e s t c h a n g e s in c o r r o s i o n with s m a l l c h a n g e s i n c o p p e r c o n t e n t a r e c a u s e d by a n i n c r e a s e i n c o p p e r f r o m 0 . 0 1 t o 0.04 p e r cent. T h i s c h a n g e w a s found t o d e c r e a s e t h e t h i c k n e s s l o s s by c o r r o s i o n t o a p p r o x i m a t e l y 2 0 m i l s o v e r a 1 5 . 5 - y e a r p e r i o d a t K e a r n y , N. J. I n c r e a s i n g t h e c o p p e r c o n t e n t f r o m 0.10 t o 0 . 2 4 p e r c e n t only d e c r e a s e d t h e c o r r o s i o n l o s s by a b o u t 1 . 9 m i l s o v e r t h e s a m e p e r i o d . Evidently, t h e r e s i d u a l c o p p e r p r e s e n t in t h e low c a r b o n s t e e l w a s s u f f i c i e n t to c a u s e i t to b e h a v e in a s i m i l a r m a n n e r to a c o p p e r - b e a r i n g s t e e l . A low c a r b o n s t e e l without r e s i d u a l c o p p e r would be e x p e c t e d t o h a v e a h i g h e r c o r r o s i o n r a t e t h a n t h e m a t e r i a l t e s t e d . C o r r o s i o n r a t e v s . t i m e c u r v e s show t h a t t h e m e t a l l o s s d e c r e a s e d rapicily o v e r thc f i r s t two y e a r s , a n d then a p p r o a c h e d a m o r e c o n s t a n t r a t e w i t h i l l c r e a s i n g t i m e . T h e s l o p e of t h e c o r r o s i o n t i i n e c u r v e b c t w c e n t h e f i f t h and t e n t h y e a r i n d i c a t e s t h e e f f e c t i v e n e s s of t h e c o r r o s i o n l a y e r in p r o t e c t i n g thc st.eel f r o m f u r t h e r c o r r o s i o n (7, 10). T h e c o p p e r - n i c k e l - b e a r i n g s t e e l i o r r i ? e d a p r o t e c t i v e f i l m which k e p t t h e c o r r o s i o n r a t e a f t e r five y e a r s t o q u i t e low v a l u e s , e s p e c i a l l y in r u r a l a n d i n d u s t r i a l a t m o s p h e r e s . A s m a y b e e x p e c t e d , the t h r e e m a r i n e l o c a t i o n s h a v e t h e h i g h e s t c o r r o s i o n r a t e s b e t w e e n t h e f i f t h and tenth y e a r ; i n d i - c a t i n g t h a t c o r r o s i o n p r o d u c t s f o r m e d in c h l o r i d e a t r l l o s p h e r e s o f f e r l e s s p r o t e c t i o n t h a n t h o s e f o r m e d in o t h e r e n v i r o n m e n t s . T h e h i g h e s t c o r r o s i o n r a t e f o r t h e coppe:--nicrkel-bearing s t e e l b e t w e e n t h e fifth a n d tenth y e a r w a s only(1.28
rrij1.s a y e a r , c o m p a r e d with 2.72 m i l s a y e a r f o r t h e sarlle m a t e r i a l . a n d l o c a t i o n a f t e r t h e f i r s t y c a r of e x p o s u r e . T h i s w a s a l-l~arirl;: s i t e ; t h e r u r a l and i n d u s t r i a l s i t e s had c o n s i d e r a b l y l o w e r v a l u e s .T h e c o r r o s i o n r a t e s on t h e low c a r b o n a n d low c a r b o n c o p p e r - b e a r i n g m a t e r i a l h a v e d e c l i n e d c o n s i d e r a b l y when c o m p a r e d t o the r a t e s d u r i n g i n i t i a l y e a r s ; h o w e v e r , t h e r a t e s f o r t h e s e s t e e l s s t i l l r e m a i n c o n s i d e r a b l y h i g h e r t h a n t h o s e f o r t h e c o p p e r - n i c k e l - b e a r i n g s t e e l . T h e s u p e r i o r c o r r o s i o n r e s i s t a n c e of t h e c o p p e r - n i c k e l - b e a r i n g s t e e l is c l e a r l y e v i d e n t when F i g u r e s 7 and
9
a r e c o m p a r e d . CONCLUSIONS 1. A f t e r t h e i n i t i a l y e a r s of e x p o s u r e , c o r r o s i o n r a t e s d e c r e a s e d s i g n i f i c a n t l y f o r a l l s t c e l m a t e r i a l s a t e v e r y c o r r o s i o n s i t e e x c e p t N o r m a n Wells, w h e r e t h e c o r r o s i o n r a t e s w e r e low e v e n f r o m t h e s t a r t of the t e s t s . 2. T h e r e s i d u a l c o p p e r c o n t e n t of t h e low c a r b o n s t e e l m a t e r i a l w a s s u f f i c i e n t t o d e c r e a s e i t s c o r r o s i o n r a t e . 3 . T h e c o p p e r - n i c k e l - b e a r i n g s t e e l f o r m e d a n a d h e r e n t , p r o t e c t i v e o x i d e f i l m which m a i n t a i n e d t h e c o r r o s i o n r a t e s a t low v a l u e s a f t e r f i v e y e a r s 1 e x p o s u r e . T h i s w a s p a r t i c u l a r l y n o t i c e a b l e a t t h e r u r a l a n d i n d u s t r i a l s i t e s , 4. The t h r e e m a r i n e l o c a t i o n s h a v e t h e h i g h e s t c o r r o s i o n r a t e s b e t w e e n t h e fifth and t e n t h y e a r . 5. The c o r r o s i o n p r o d u c t f o r m e d u n i f o r n l l y o v e r the s u r f a c e s of a l l s a m p l e s , but thc l o w e r e x p o s e d s u r f a c e s w e r e a t t a c k c d m o r e s e v e r e l y t h a n t h e u p p e r e x p o s e d s u r f a c e s . R E F E R E N C E S 1. Gibbons,E.
V. A s s o c i a t c ConlmitCee on C o r r o s i o n R e s e a r c h a n d P r e v e n t i o n S u b - C o m r n ~ t t e e C , "The C o r r o s i o n Behaviour of M a j o r A I - c h i t e c t u r a l a n d S t r u c ~ u r a l M e t a l s in C a n a d i a n A t n i o s p h e r e s : Surnrnar y of Two- y e a r R e s u l t s , National R e s e a r c h Councll,Ottawa, F e b r u a r y 19 59.
2. Gibbons, E. V. C o r r o s i o n 17, " A t r n o s p h e r i c C o r r o s i o n T e s t i n g 01 M e t a l s in Canada!' (19 6 1 j, ! ! 8t - 32Ot.
3. F o r a n ,
M.
R., Gibbons, E. V., a n d Wellington, J. R. " T h e M e a s u r e m e n t of A t m o s p h e r i c S u l p h u r Dioxide a n d C h l o r i d e s ; ' C h e m i s t r y in Canada, 10, (19 58), p. 3 3 -41. 4. Gibbons,E.
V. "Outdoor E x p o s u r e S i t e s of t h e N a t i o n a l R e s e a r c h Council1' C h e m i s t r y i n Canada, 12, (1960), p.44-48. 5. L a r r a b e e , C. P. a n d E l l i s , 0 . B. I 1 P r o g r e s s r e p o r t of s u b g r o u p of S u b c o m m i t t e e VII, on c o r r o s i v e n e s s of variou:; a t m o s p h e r i c t e s t s i t e s a s m e a s u r e d by s p e c i m e n s of s t e e l and z i n c , A, S. T. M e P r o c e e d i n g s 53, (1953), p. 194-197. 6. L a r r a b e e , C. P. and E l l i s , 0, B. " R e p o r t of s u b g r o u p of S u b c o m m i t t e e VII, on c o r r o s i v e n e s s of v a r i o u s a t m o s p h e r i c t e s t s i t e s a s m e a s u r e d by s p e c i m e n s of s t e e l a n d zinc. A . S . T.M. P r o c e e d i n g s 59, (1959), p. 183-201.7. Copson, H. R. I 1 T h e o r y of M e c h a n i s m of R u s t i n g of Low Alloy S t e e l s in ~ t m o s ~ h e r e : ' A. S.
T.
M., P r o c e e d i n g s , 45, (1945), p554-580.8. Hudson,
J.
C. a n d S t a n n e r s , J. F. J o u r n a l I r o n a n d Steel, 180, (1955), p. 271-284.9. L a r r a b e e , C.
P.
a n d Coburn, S. K. F i r s t I n t e r n a t i o n a l C o n g r e s s on M e t a l l i c C o r r o s i o n , London, B u t t e r wor th' s S c i . Publ. ( 1 9 62), p. 276-285.10. Copson, H. R. t'Long-Ti:ne A t m o s p h e r i c C o r r o s i o n of Low-Alloy S t e e l s ,
"
A . S . T . M . P r o c e e d i n g s , 60, (1960), p.650-665.T A B L E I
AVERAGE AMOUNT O F SULPHUR DIOXIDE I N T H E ATMOSPHERE
AT EACH T E S T S I T E FROM 1 9 5 4 - 19 64 S i t e No. L o c a t i o n MgSO / d m 2 / d a y 3 Halifax M o n t r e a l T r a i l Ottawa Y o r k Redoubt S a s k a t o o n E s quimalt N o r m a n W e l l s
T A B L E I 1 C H E M I C A L C O M P O S I T I O N O F T H E S T E E L S T E S T E D L o w c a r b o n ( A 3 0 . 0 9 0 0 . 2 9 0 . 0 0 2 0 . 0 6 4 0. 0 1 3
-
0. 035 L o w c a r b o n c o p p e r - b e a r i n g ( A 4 ) 0 . 2 0 5 0 . 3 2 0 . 0 0 2 0. 060 0 . 0 1 4-
0 . 0 4 8 L o w a l l o y c o p p e r - n i c k e l - b e a r i n g ( A 5 ) 0 . 7 3 8 0 . 7 8 0 . 0 2 1 0 . 1 2 4 . 0 . 0 9 5 0 . 6 8 0 0 , 0 3 2 T A B L E 111 A V E R A G E M I L S P E R YEAR P E N E T R A T I O h T B E T W E E N F I F T H A N D T E N T H Y E A R S a s k a - M o n - H a l i - H a l i - N o r m a n E s q u i - M . a t e r i a l O t t a w a t o o n t r e a l fax** f a x * W e l l s m a l t T r a i l - L o w c a r b o n ( A 3 ) 0 . 2 9 4 0 . 0 1 8 0. 1 3 0 1. 3 3 6 0 . 4 5 8 0. 042 0 . 2 0 0 0. 1 2 0 L o w c a r b o n c o p p e r - b e a r i n g ( A 4 ) 0 . 2 8 2 0. 0 1 8 0. 142 1. 1 6 2 0 . 4 7 0 0. 032 0 . 1 9 2 0 . 0 8 6 L o w a l l o y c o p p e r - n i c k e l - b e a r ing ( A 5 ) 0 . 1 0 6-
0 . 0 4 6 0 . 2 8 0 0. 1 8 6 0 . 0 2 2 0. 1 2 4 0 . 0 2 0 L*. -E m a r i n e - i n d u s t r i a l**
m a r i n e - r u r a lF I G U R E
1
A V E R A G E A M O U N T O F S U L P H U R D I O X I D E I N T H E A T M O S P H E R E
A T E A C H T E S T S I T E F R O M 1 9 5 4 - 1 9 6 4
ATMOSPHERIC CORROSION RATES FOR THREE TYPES OF STEEL
A 3
Low Carbon
Steel
I
Year Exposure
A 4
Copper Bearing
Steel
U
2 Years Exposure
A 5
Copper
-
Nickel Bearing
Steel
B€l
5
yea^Exposure
10
Years Exposure
OTTAWA
SASKATOON
MONTREAL
HALIFAX
MAR1 N
E
l
NDUSTRlAL
ATMOSPHERIC CORROSION RATES FOR THREE TYPES OF STEEL
HALIFAX MAR1 NE RURAL
ESOU
IMALT
i
1
i
--+.--..--I---+
I'
4.-.-
Ij
! II
I &---4.-
1
ii
I II
I
I 1 I 1NORMAN WELLS
TRAIL
FIG
2
(Cont'd)
I "._ ___._"I___
-
-
-- -- -- -.--CORROSION
VS TIME CURVES
FOR
THREE TYPES OF STEEL
o
Low Carbon Steel
Copper Bearing Steel
@
Copper -Nickel Bearing Steel
NORMAN
WELLS
SASKATOON
0
2
4
6
8
10
0
2
4
6
8
1 0
Time. years
Time ,years
0
2
4
6
8
10
Time, years
MONTREAL
Time ,years
CORROSION VS
TIME
CURVES
FOR THREE TYPES OF S T E E L
OTTAWA
TRAIL
"0
2
4
6
8
10
Time, years
"0
2-
4
6 8
10
Time,
years
HALIFAX MARINE RURAL
HALLFAX
MAR1
N
E
l NDUSTRIAL
-0
2
4
6
8
10
Time,
years
-0
2
4
6
8
1 0
Time,
years
CORROSION VS
TIME CURVES FOR
LOW
CARBON
STEEL AT VARIOUS CANADIAN CORROSION SITES
0
1
2
3
4
5
6
7
8
9
1 0
Time,
years
CORROSION VS TIME CURVES FOR COPPER
BEARING S T E E L AT VARIOUS
CANADIAN
CORROSION SITES
0
0
'1
2
3
4
5
6
Time,
years
CORROSION
VS
TIME
CURVES FOR COPPER-
NICKEL BEARING STEEL AT
VARIOUS CANADIAN
CORROSION
SITES
-
. O
1
2
3
4
5
6
7
8
9
10
Time, years
Ottawa
M o n t r e a l (31 Halifax (Marine Industrial)
($1
Mag.
IX
Halifax (Marine R u r a l ) ( 5 ) Norman W e l l s
6)
Trail (8)
Mag.
lX
Steel material exposed ten years
at Norman W e l l s Mag. 18%
Steel material exposed ten
years at Ottawa M a g .
10X
Steel material exposed ten y e a r s
at
Kalidax(marine indue tr ial)
Mag.
POXAPPENDIX
$:[ y' ]i KC)
1.
QI! 5.'kL\il!.---.
---
---
---
Lot NO.
Plot31
Sa:.,~gl.oQ:c.:lzj.nal.
;?:l.na?.
\it 3~1~.i;fi]--Coi*xo;:.on
Bzte
Doe:I.~:n;~tion 17vn1bo:c
V-i:.W?;
j ~ o s a ? - o m ( r , i i l n1
(TI!,J:J/<C~.:Q.,
-."a. _.-.U-.l....l.----"~-."..--IIYI.1.-..CII-Y.....-.----....-.--
-I*.---
--.--
-
---.
A 4
1
237.4.
%30,9
5,
5
021602
2
2j703
230.8
6.5
L o
O?
1..023
23'1.5
291.1
6.4-"-
:L-
OX
3. ,, O i.a-
-
Ave,
1.02 ai.ve..
I-,
02A
5
1. 3 . 8 7 . ~la;!,.j;
5-1
0 , O ~ J 0 ~ 0 02
3.87.6
182.55.1
0,80 0.803
189.7
1.84-0'4 0 9
--
0.77
-
C o77
CI.hve.
0.79
Avc
.
0070
o.,
GT
0892 0,25Ava.
0'04LO.!.
. :I..
5
9
7.7
I.
.
.
'
2
'1.12,12
6,3
3..oa
.,.3..-...-Xvc.
1.14.
-.&.-,-".."--.---.-
---. -.
...--
.--
.,d -0-----.--..-.-
...___.a--e-Mota:l Btunplo
dr:~-~:ind.
3'j.rml kitI,]."
CorrooJ-on
l h t eLot No,
Deai~fantion
Nusnbov !V-b.
lI Loou N i l u 1-00t
Mllo/Yurir1 A 3
1
251e6
240-011-6
1
a, 021.82
2250.9
233.1
l l n a
1005
2.1853
25605
24498
11.7
---
3. 0 134--
1.04
Avo.1,04
Ave
.
1
e 8 4 A5
1
19500
3.06.6 6 - 43."52
I.
-
3~ r,2
195-0
186.7
0 . 3 Z Y70
1 - 303
183.8 100.39 - 5
----
1049
-
I
- 49
Ave.
1-37'
dve ,?_,?i
2
..
4-7
2.402,,T3
--m..Ava.
2,,42
2 h 4 4 2 " 5-1 2 - 4 0 -. . c . -Avo. 2,.36I n
57 3..5'7
L
,,5
*I
--.-,..
Avo,3.,57
1.; 23l.23
1.619
---
Avo,1-
21
/?21!.'T20,G
3-
2 4 .
2'35.'7 20.0 j1 27231.1
20-1
3-16
-. " " . -aAvo.
3 * 2 2 220.919,5
.
>,06.
21-'10%19-8
7 * 1""2 1 5 e l
19.5
..
~ o ! x
--.-
Avo. 3,013 0,650 -
STO.,
6 3
Ave
,0.
GC.
0 , 3 5 0 , 5 7 Q .57
.---..
.jb.re '9o,,
7':
E e t c ~ l S a m j ~ l o
O~!.,@nnl
P<<n:xl ':it Caryoa:i.on 1:ri-t~Lot
130.
Dooigi~ation
E~unber
Vt
!PI; LorleW.il.a.loot;
l,'iila/y~ccr4
A
31
25d.:L-
..
-.
.a2
258,O
233-324.7
5.
Ct3 0029
3
249.6
225.1
24a5---..
5
*-!Xi
0.38
--
Avo.
5.07
JLVO.0.39
SITE
HO0
4.
I A (1hj:ine Indur.l;rial)-.--~-ccI..Imnnn-.--.--.---* ~~-LYLY~LY----LY---
I.!e
-i;aZ
SulitploOr:i.g?jinal
Final
\ I t C o r Y o ~ L a n X a t oLot
130.Deuignation
:Ttunbsr W-i; Yl i; L O W S 111P13 1 0 ~ 3 l i i l ~ f l o a rC--U...U-W.U.IL.---- ...----PIIS
ns
3. 1.6~- G 2.~14~4 ~ - 7 ~ 2 2:10 2 .,y i ~
2
1313~4
7
7 .
3.7 0 2.67 2 ,5.;
5
290,1
1'7
2 ., 4. I.?,'!
:!
e78
:!. $6 -.. % ,..-.-.a.--.
7..
rlva, 2,,72 : Y J ~ . ;2u'izMe
trr?. SL?.IIIQ'I.B Or1{;?.):i:2.1 Fl;:tid. 71 '6 CorromionR a t o
Lot.
No,
Dao:l.:;l~nt.ion llr\nnbcr Y.t, II?%JJOCU
MLLa l o o t~ i l n / ! ! c s r
--.~-,-cc.~~..Ic--.-.--------II---I--C----
--
2
A
5
1
169.6
I65.5
24.1
5
079
1,)
892
1.94.9
1.6'3.62 5 a 3
7
63'7
293
3 1 0 9 n 0165u2
24.G--"-
7 - 8 5
-
1-
93Ava,
3.0'(I...94
3
A3
1
25004
1.813n0 62°C9 %
Ft!)
3.-96
2 250.8190.3
G0,5
3 * 50l..?O
S
246.0
183,g
6201
9
075
h - - r1295
..--
Ave,
9.63
Ave.
1,94
A
4 3. 235.3 227,3 8.G
Xi35
1-55
224yo6
229,3
6.3 -" "-
3 G
.
3. n -20 23
2jB,.7
2Y0*3
6 ,4
1,j2 3.,.
32 * - e m ,....--
h ~ e t ,1,.52
AVO,Ie32
L
107*7
182.5
6.2 0.397
O e 9 72
m g 0 6
179-3
6 , ~ 0.99 0,;99
3
187n4 i . D l n l6.7
.,-.-..,-
O e g 9 0,99 ----aAvo. 0,30 Avo, (je,'3E
c . -. . ull- AVO. 2 , 2 3 i ~ v e ,
1-11
1
2jS.1
224.211,9
2.18 ?-<0 9
2235.9
222.213.'1
2.1.5
3. a'So
3
238.0
224.1
13.9
2,13 7.. 09 Y--
Avo. 2.27Ave.
3.,0(;1
394.7183.6
1.1.1
1
* 74- O,Z7 2 1886%177.3.
11.
L1-74
O.,t373
18609
7-76oo10.9
-...I1
u '77,...-.-...-
0,85Avov-
1,,73
I\.VO,0 ~ 6 7
Avo,
4,,11
Ave*
0,02A
5
3.188
., 2-.
-
-
.., 2195.8
174,*7
2 l n l
3
d,51
0,.53
3
136.6
174*8
21,s
3.,
I?. 2 0.. 3 4 -. . -a---
h ~ e .3*37
~lvc?, O n > ~ ! ---.--.I---.-~--.--.,-.CY~U-U--------.~--'-..---
SITENO
,. 5 I?OI@I:IIU vnFJ;.Ls ..
. - . U S , - W I . . L . . - - - - . - - - - m ~ . - r - - ~ = . * ~ = - . . - ^ l - - -Islet
sl
3wnple Orii:inalPPnnl
Vt
C o r r o c i o c lZaLeLot
:lo.D o l j i ~ n a t i o n
Nv.lni?or
W-1; lip& TIOOO1ki.I.a
L G O - ~ . l t j . l c / ~ ~ z x ~-.-
-ICU--LC-..-O-.;ll.,.---.-1
A3
1
2 5 1 e 3
251.n2 0.Z
0.. 02 0,0.?2
251.04
252050-0
0.
Oi, 0, (!03
251.5
251.5
0,O 0,OO0.00
---."
-*.-.. AiN!..
-
,',\7~-.,
A 41
23'7*3
237.3
0.0 0.00 o,C?2
236,.7
231.8
0.0
O,OOi\.no
3 235.6235.5
0,1
---
0,0;2 0. a2 --.-L Ave,-
~ L V e.
-
f"5
3.194
1l.95
+9
O,, 2 0.03 0 , C S2
5
195.5
0
..0
0 .GO
0. GO3
107
.,
7
167.7
0. i ) O.GO 0.00 .A,..-u-
.*m..*- Aye, 0.- ~1.1:s.,
-.. .- r------.---..~4->--,~~..,r-.~~--lmrlr-r.,.l-lo.r.--.-.wI-.----------*--~~~------i . 2A
3
1
253.*225Qwt3
O,/I O r O G d e O j 2-
252.0251,.7
3 , 3
0,OT 9,02>
250,8 250.6 0.2 0.03 <) ,,02
-..-.a*- .,.-,,.- I:,ire, 0,05 A - ~ Q . O,.il5Pio'w?. !3n:i\j?l0
Or:i.t.:!
nu:!.
I?:;. Y~;-L:I- M .'. \I f:~:c.;:lo!;:;.<;n ];:!.tf3!,qt
)To,
I ) o ~ ~ ~ . ~ ; : r ~ ~ ~ 5 l o n R t ~ . ~ i r b o r t:?t I I4lI.a Lor~.i; ~:l.%a/Pr;or.
_.--.-.----
-.*- CCCCCC.C.CCCC,_I , .-, A.-.-...-..c...
-..w.c.*..--
r.-8.-u---I--...--*--..---
2
A 5 '
3.109.1
3 - 3 9 o,Z Q. 03 0, !322
195.5
1.95 4 20.3
0 ; . 03 0,023
1E794
1.87eO 0.4. 0.06 0,O.j-I-" 0-
.----,
A v o , 0 , 0 5
0.02
A
4-
3.256,
G235=6
3..0 O o l . 6 G,Og2
236,'t 2:55,80,,9
(3 ,, ?. J!. 0.03'3
236-0
25409
1.,1---.
0
*,1.'7
..-
0 .c i
Avo,
0e15
A783 0.05A
5
1
195.3
3.94'e2ltl
1). 1.7 O e 0 32 !-800
6
1 1 3 7 o 5 1.1.Om17
0.033
394.4193.4
1.0
0.l.G 0 , 0 3..,.-.-.PI -.a-
--
.,-. r.l".~-...L.-.~~--.~,C...-.-I-V,..I.-..~",-,,,- ..~.
.-.
-. ..-*. ."..- lr.--1".1-...*1.- U,.'"l,-"-. . A."..
..I.-
' .--- C-l.-
...
--,.---
Lot
i t ~ .
M3tri1 p I . r . \J-L ~ ; . r ~?:?;or: ion
r!n'iriD o : : ~ : ~ . G ~ I s . ~ S O It'u!i\bol* W.t 1 ;I -: I; ~:01;;3
-
i?j.2.,7 LO:J~G~.I~.~-D/JLCC*
Avo. O,.CO Avo. 0.49
A 4
I
237.7
232,rl.5 . 3
0.03 0, $22 2j7,.O 233,O 4.0 0 . 6 3 0 , .
31
3
2 3 2 - 6 22fl-O 4.0 0,75 0. j ZC W C W I
---
9IIE
NO,
8 TlhiIL -. ~ !.!? 26 'I., 27 3. ";:
2...
I..,..
J, L ~ T ~ c , , ?.F,:i:5 :I- ,. 2 :.; ,j ??a,;.:
-...*...-
Aye,,
3. ,-, . ;!.C
ar:s
o xi..
oil R~riici
s ~ ~ L I : . ~ / Y
o a r
=
$ & s r $ w e ; ; ~ ; ~I)