READ THESE TERMS AND CONDITIONS CAREFULLY BEFORE USING THIS WEBSITE. https://nrc-publications.canada.ca/eng/copyright
Vous avez des questions? Nous pouvons vous aider. Pour communiquer directement avec un auteur, consultez la première page de la revue dans laquelle son article a été publié afin de trouver ses coordonnées. Si vous n’arrivez pas à les repérer, communiquez avec nous à PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca.
Questions? Contact the NRC Publications Archive team at
PublicationsArchive-ArchivesPublications@nrc-cnrc.gc.ca. If you wish to email the authors directly, please see the first page of the publication for their contact information.
NRC Publications Archive
Archives des publications du CNRC
This publication could be one of several versions: author’s original, accepted manuscript or the publisher’s version. / La version de cette publication peut être l’une des suivantes : la version prépublication de l’auteur, la version acceptée du manuscrit ou la version de l’éditeur.
Access and use of this website and the material on it are subject to the Terms and Conditions set forth at
Dependence of degree of silica polymerization and intrinsic mechanical
properties of C-S-H on C/S ratio = Influence du rapport C/S sur le degré
de polymérisation de la silice et les propriétés mécaniques
intrinsèques des C-S-H
Beaudoin, J. J.; Feldman, R. F.; Baron, J.; Conjeaud, M.
https://publications-cnrc.canada.ca/fra/droits
L’accès à ce site Web et l’utilisation de son contenu sont assujettis aux conditions présentées dans le site
LISEZ CES CONDITIONS ATTENTIVEMENT AVANT D’UTILISER CE SITE WEB.
NRC Publications Record / Notice d'Archives des publications de CNRC:
https://nrc-publications.canada.ca/eng/view/object/?id=035c8f42-b5b3-478b-bfaa-6b69079d6947
https://publications-cnrc.canada.ca/fra/voir/objet/?id=035c8f42-b5b3-478b-bfaa-6b69079d6947
'YHl
Natlonal Research
Conseil natlonal
"la
1
+
1
Coundl Canada
de
recherches
Canada
no.
1510
c .
2
I
BLDG
. :
Institute for
lnstitut de
Research in
recherche en
Construction
construction
Dependence of Degree of Silica
Polymerization and Intrinsic Mechanical
Properties of
C-S-H-
on
C/S
Ratio
by J.J. Beaudoin and R.F. Feldman
Reprinted from
8th International Congress
on the Chemistry of Cement
Rio de Janeiro, Brazil, September 22
-
27,1986
p. 1-6
(IRC Paper No. 1510)
Price $3.00
NRCC 28689
-
& s m
Lics
a o z m
ant&tudi&
Ies
propri&€s
physiques
et mhnigucs
de six
pr@mtions
de
C-S-H
(hydrates de dicate calciquc)
aymtdes
rapporrs
CIS
de
0,68 B
1,48
et desrapports
HIS
de
I.91 & 669.
La
caracthisacion dcs
rnat&nauxm p r e n a i t
les msms
de la densite.
de la
surface et
du
degd
de palym6risauon
de
la
silice.
Us
ant
acquis
une cataine connaissance
des drfftrences
de
nature
enae les
six
pdpmtions
de
C-S-H
partir
de
donn&esobrenues
sur les mdcarions
de
la
masse
er
de la longueur par rapport
I'humiditk rcianvc.Une
certaine d€pendance du de@ de polymkrisatian
s u rle rapport
US
a 6tt etablie.
Des
6chantiIlons comprirnts de p o d
de
C-S-H
out
ttt
atilists pour
mesurer
Its
propridt6s
mkaniques.
La reIation
enmle module
d'€WcitE
IE)ou
la midmtC
(H) etla
pornsit6
a kt6
dCrermin6e. Des
estimations
des
valeurs inainstqucs
(ou
porosite
z h )dc
ces
propriktks
(Eget
Hg)
ont
tt15faites
P
pa&
d'analysts
utilisant divers
modklcs Gel
asticite
pour les corps pareux. L'influcnct du rapport
US
ctdu
degd
de polymCrisation sur les
propriEt&
rnkaniques intrinstques
ctles
a u wp m h c s des mod&les
(factem
"forme",
etc.) a tgalement
6t15dhterminet.
h
s
facteurs "fonnt" associks h
dcuxdes
modtles
dbpendent
du rapport
US.
Le
module
d'dlasticitk intrinstquc
etlcs
v d e m
dela
rnicroduretd
nc dwndcnt pas du
r
a
m
CrS,
Lts
auteursdiscutent
dm
dsultats
entenant
cmptc
dcs ddTQtncesdans la
microshucture
dw mrttkrkuxde
C-S-A.
- - -
- - - - - - -.
_
_
- - .-
-- -
J.J. Henudoin and R.F. Peldman, Oivision of Building &search. National Research Cnl~ncll Canatla; J. baron and M. Gmjeaud, lafarge Coppee Recherche, Prance
DBhl:NI)I~~CG OF I)I<(;KKE 01: SLI.ICA POLYMIXLZATION AND INTRINSIC MECHANICAL PROPERTIES OF C-S-H ON CIS RATIO INPLUSNC~! IW MPPORT C/S SUR LF,
IIKGHE
UE P O L Y ~ ~ H I S A T I O N DE LA SILICE ET LESPRIIPH
.kr?s
MRCAN
[QURS Z N T U I N S ~ ~ U E S U K S C-S-HSUMMARY: Physico-~echanical properties of six C-S-H preparationslhaving C/S ratios of
0.68
to 1.49 and HIS ratios of 1.91 to6.69
were studied. Characterization of materials included density, surface area and silica polymerization measurements. Some insight as to differences in the nature of the six C-S-H preparations was also obtained from weight a~ld length change versus relative humidity data.A
dependence of degree of polymerization on CIS ratio was established. Compacted samples of C-S-H powders vere used for mechanical property measurements. Modulus of elasttcity (E) or microhardness (H) versue porosity relations were determined. Estimates of the intrinsic (or zero porosity) values of these properties (E,, and Ho) were made from analyses using different elastic models for porous bodies. The dependence of the intrinsic mechanical properties And other model parameters (shape factors, etc.) on CIS ratio and degree of polymerization were determined. Shape factors associated with two of the models are dependent on C/S ratio. Intrinsic modulus of elasticity and microhardness values are independent of CIS ratio. Reeults are discussed with respect to possible differences in the microstruct~~re of the C-S-H materials.Foram estudadas as propriedades fisico-quimicas de
6preparacdes de C-S-H tendo rela
cdes de C/S de 0,68 a 1,49, relacdes de Il/S de 1,91 a
6 , 6 9 . Acaracterizacdo doe materiais
incluiu dcnsidadc, Srea de superficie especifica e medic6es de polimerizaclo da silica. Tam
bim se obtcve algum discernimento quanto 5s diferen~as
na natureza das seis preparacdes
de
C-S-H, a partir de mudan~a
de peso e comprimento versus dados de umidade relativa. Foi estg
belecida uma dependencia do grau de polimeriza~do
para corn a relacdo C/S. Foram usadas
smog
tras compactadas de C-S-H para medicdes da propriedade mecbnica. Determinaram-se m6dulos de
elasticidade
(E)ou microdureza (H) versus relacdes de porosidade. Foram feitas estimativas
dos valores intrinsecos (ou porosidade zero) destas propriedades (Eo e Ho) a partir de an514
ses usando diferentes modelos elisticos para corpos porosos. Foram determinados a depend&-
cia das propriedades meclnicas intrinsecas e outros parlmetros-modelo (fatores de
forma,
etc.) sobre a relacgo C/S e grau de polimerizaclo. 0 s fatores de forma associados com dois
dos modelos dependem da relacso
C / S . 0m a u l 0 de elasticidade
eos valores de microdureza
sHo independentes
d arelacdo C/S. 0 s resultados.sdo
discutidos sob o aspect0 de
possiveis
diferen~as
na microestrutura dos materiais de C-S-H.
J.J. Beaudoin and R.F. Peldman, Division uf Building Rerearch, National Rcrearch
Council Canada; J. Baron and M. Conjeaud, Lafarge Qpp6e Becherche, France
INTRODUCTION
Calcium silicate hydrates (C-S-H)* form the principal cementing phases in binders for such building materials as portland cement mortar, concrete, certain typea of .brick and other lime-silica products. The silicate minerals in portland cement (C.S and C2S) hydrate to form C-S-H and calcium hydroxide (CHI. It ie generally held that C-S-H plays an important role in the mechanical behavior of cement systems. Several factors, however, make a quantitative assessment of thia role difficult: (i) Hydrated cement contains several other cement phases (aluminates, ferrites, etc.) including CH; (ii) the role of CH has been investigated but is not fully understood (1);
(iii) the C/S ratio varies over a wide range, from 0.8 to nearly 3.0 depending on waterfeolid ratio,
temperature and time of curing (2); (iv) the
dependence of strength on degree of polymerization of the silicatea is not clearly established; (v) the extent to which mechanical properties of the C-S-H solids contribute to the the porous system
properties cannot be determined directly in hydrated cenents.
Several authors have attributed strength
dif ferencea of cement products to dif ferences in CIS
ratio of the C-S-H present in thoae products. For example, low strength of autoclaved cement products has been attributed to the preeence of aC2SH
(CIS
-
2.0) ( 3 ) .In order to investlgate the contribution of the C-S-H phaee alone, it was decided to syntheeize C-S-H having a wide range of CIS ratioa and to conduct mechanical terte on the material. In addition to mechanical behavior, the importance of the C-S-H phase for volume change and durability ie recognized. There ie, however, a paucity of data on there characteristice for the C-S-8 phaee itself. The objective of this inveetigation wae to determine the dependence of the degree of polymerization, adeorption characterietice and intrinsic mechanical propertiee of C-S-H preparations on CIS ratioe.
EXPERIMENTAL Haterial8 and Preparation
Six C-S-E compoeitionr were produced in aqueous rolutione containing variable proportions of sodium mtarilicate and calcium oxide. They were dried by
WarNng in acetone and ether.
In
order to removethe acetone and ether they were firet expoeed to
a
rtream of dry nitrogen (500 a 3 / d n ) for four daye. Then t h y wmre dried in nitrogen atmasphere at 40%
for 24 hour.. Thir wu the starting condition foz
the experirntal work.
The C/S and H/S ratioe are given in Tnble 1. The C I S ratio vu obtainad w i n g X-ray fluorwcence
and flame ctrophotowtry. The H/S ratio wam
calculated% 1088 on ignition at 1000°C.
I.
spite of the drying t r e a t m t e , the tvo C-S+i preparatiow b v i n g tha highart C/S ratio bad
-at chedrtry notation: C
-
CaQ; S-
Si02;a
-
50.abnormally high ignition loss. The HIS ratioe for these preparations are unexpectedly high when
compared to published values (2). There is no
obvious explanation for thid.
No carbonation was detected in the powdered samples. Calcite and vaterite were found in the
compact with CIS = 1.26. Aragonite and vaterite
were detected in the compact with CIS
-
1.17. Nounreacted C or S wee detected by X-ray diffraction or infrared spectroscopic methods.
Morphology, as revealed by SEM, is broadly divided into two groups according to CIS ratio:
CIS
-
0.99-
1.49 and 0.68-
0.87. C-S-H appears ae50
urn
clusters of 1urn
thin foil subparticlee in thefirst group and is representative of Type I1 cement
paste morphology (4).
In
the eecond group thesubparticlea are much smaller and difficult to resolve'at the 20nm limit of the SEM. At
CIS
-
0.68, the subparti,clea appear as e m 1 1 grains.It could not be determined if the grains were agglomerates of thin foils.
Density value8 were determined ueing mass and solid volume measurements determined by helium
pyknometric techniques (5). Surface area8 of the
C-S-H eamplea were determined ueing nitrogen adsorption techniquee with Numinco-Orr
instrumentation. Density and surface area values
are given in Table 2.
Silica Polymerization Hcaeurements
The molybdate complexing method developed by Parrott and Taylor was used in this inveetigation
(6). The molybdate complexlng reaction curves were
obtained on the following:
1) mono-, di- and polyeilicic acidr. The
aonosilicic acid was obtained by diesolving 83 mg
of anhydrous portland cement in 100 a1 of
methanolic EC1 (0.15 N). The diailicic acid was
prepared with 115 mg of hemimorphite instead of 82 mg of anhydrous cement. The polysilicic acid
was prepared by reacting 317 mg of anhydrour
cerent in 15 ml of ~lethanolic HCl (2.0 8 ) for 20
minutee. ,
2) powdered and coapacted C-S-8 eaapler. The collpactr vere prepared at preesurer rangin# from 510 to 1360 HPa.
ltro eete of meaaurewnte vere conducted on powdered
C-S-H eamples and rilicic acidr. The second set included mearuremnts on compacts.
Weight a d Length Change I r o t h e ~
C-S-8 specireno vere compacted at 510 E(Pa in
the shape of dirce 31.8 diameter and 1.27 a
thick. Weight change isotherma vere determined using a daiccator method after K.n&ro et a1 (7).
Equilibration time. were at least 30 day8 for each
humidity. Two compacts were used for each C-S-kI
preparation. For longtb change wasurerents
specimenr we- mounted on modified 'hckormann
optical extenromterr ( 8 ) . This determined the dimenrional change. to a sensitivity of
4 x 10'~ m/m. The exteneaters were placed in
J.J. Beaudoin and R.F. Feldman, Division of Building Research, National Rese.arch Council Canada; J. Baron and M. Conjeaud, Iafarge Copp6e Recherche, France
Mechanical Properties Sample Preparation
Specimens were prepared by compacting the various C-S-H powders at pressures up to 1020 MPa. A11 specimens were circular discs 31.8 mm diameter and 1.27 mm thick.
Porosity Measurement
Poroaity was calculated uaing the apparent volame of the sample and the aolid volume directly earnure$ using helium pyknometric tqchniquea ( 5 ) .
Porosity wae aleo datermined on ~ o m e compacted sampLes by mercury intrusion e t h o d s at pteeeures up
to 408 MPa.
Modulus of Elasticity Measurement
The method of measurement consists essentially of measuiing the center point deflection of a centrally loaded circular disc supported at three equally spaced edge supports (9). At least three specimens were used for each test condition. All measurements were made in a glove box conditioned at
11% RH. 1
Microhardness Heaeurement
Microhardness was determined on compacted C-S-H samples using a leitz microhardness apparatus with a Vickers indentor (10). The equipment was placed in a conditioned glove box at 11% RH. Up to ten measurements were made on each disc.
RESULTS AND DISCUSSSLON Silica Polymerization
In the molybdate complexing method the analysis of results is based upon the assumption that an individual silicic acid in a mixture of silicic acids is complexed at a rate which is solely dependent upon its degree of condensation, and that the reaction for a silicic acid with a degree of condensation '1' takes the form:
where Y is the proportion of silica complexed, bi is the rate constant and ti the time shift.
The reaction for a mixture of n eilicic acids can be then represented by the equation:
where ai is the proportion of silica with the degree of condensation 'in in the mixture.
In general, the reaction curves (log(1-Y) ve. ti, not shown) for compacts and povdered eamples ~ndice'.o. that compaction led to very little
Eerence in the degree of polymerization of the C-S-H, despite large differences in the HIS. The degree of polymerization of the eilicates. meaeured by bp (the elope of the polymer reaction curve.
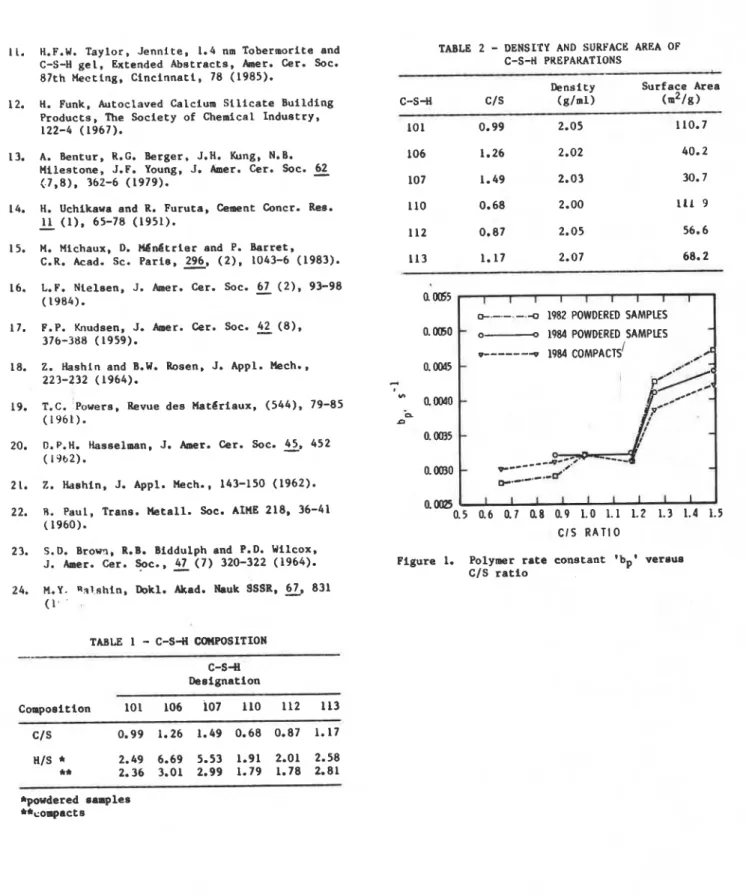
which increases when degree of polymerization decreasen; the polymer fraction refers to silicate chains longer than dimere), -is plotted against CIS ratio in Figure 1. The curves in the figure are plotted for data obtained two years apart. Thia was intended to assess possible aging effects. The degree of polymerization dxreases non-linearly with an increase in C/S ratio. The term (bp) increases slowly, quickly and slightly for the range of C/S ratios, 0.66-1.17, 1.17-1.26, and 1.26-1.49. The decrease in the degree of polymerization can be explained by both the Taylor and the Feldman models, which postulate the omission of silica tetrahedra in the silicate chain and possible replacement by hydroxyl groups (11,8). Omission of tetrahedra increases the effective CIS ratio of the polymer. Thia is in agreement with results obtained on cement pastes (12,13) and synthetic C-S-H of CIS ratio 0.8 to 1.1 (14). but was not observed on other synthetic C-S-H of C/S ratio 1.14 to 1.55 (15).
Weight Change and Length Change Isotherms Weight change isotherms are plotted in
Figure 2. All isotherms exhibit primary hysteresis. All isotherms except the one for the C-S-H
preparation having C/S
-
1.26 exhibit secondary hysteresis at lower humidities. Scanning loops start st 11% RH on the desorption branch and rejoin the adsorption branch at 80% RH. Secondaryhysteresis is probably due to intercalation of hydrate water and/or aging effects. The preparation having CIS
-
1.26 (no secondary hysteresis) has thelowest amount of water held at 11X RH (6%) and has a low N2 surface area (40 m2/g). Adsorption on rewetting to 80% RH is reversible. Initiation and completion of intercalation and dissociation
processes associated with hydrate water may occur at low humidities (below 11% RH). Thus,
irreversibility may not be observed. The C-S-H preparation with lowest C/S ratio loses the largest amount of water at high humidities on desorption, e.g., for C/S = 0.68 there is a 23% weight loss at
80% RH compared to a weight loss of 3-52 for the other preparations. This is an indication that the pore structure is much coarser for the C/S
-
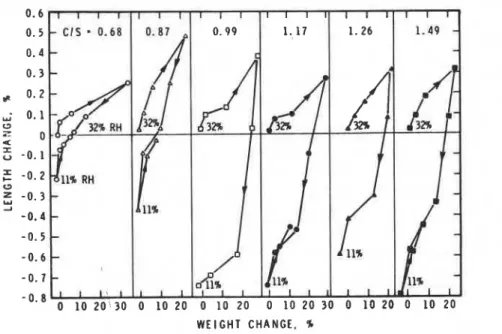
0.68 material. These factors will be considered when examining the dependence of mechanical properties on C/S ratio.Length change veraue w i g h t change curves are plotted in Figure 3. The curves are qualitatively similar to the curve for bottle hydrated cement obtained by Peldman (8). The curve for C/S = 0.87
is quantitatively similar, Measurements on drying could not be made below 11% RH as the specimens fractured. The starting condition was 32% RH. The slopes of the length change
-
m i g h t change curvenon deeorption (100
-
BOX RH) for C-S-H in order of increarring C/S ratio are 7.1, 25.3, 123.3, 38.0,110.0 and 55.0 n
lr3,
renpectively. The t r opreparations with the lowast C/S ratio have the l e s t mlopen. Thin may Indicate that they have a coarser pore etructure than the other preparations, if one ignores possible aging factors. 'Ihis is supported by pore m i + & distribution data (not
presented). Pore size maauremente on compacts
(510 MPa) indicate that in the fine pore range (pore
radii 0.0022-0.0015 urn, the lLmiting size of the
poroaiwter) the pcreantqe o f pore voLum war
J.J. Beaudoin and R.F. Feldman, Division of building Research, National Research Guncil Canada; J. Baron and M. Conjeaud, hfarge Copp6e Recherche, France
CIS ratio. The high surface area for the CIS
-
0.68 material may be related to the presence ofmicropores not detected by mercury porosimetry. All the C-S-H samples have large, irreversible shrinkages. Irreversibile shrinkage at 32% RH in order of increasing CIS ratio is as follows: 0.05. 0.11, 0.72, 0.60, 0.42 and 0.58%. The two lowest CIS ratio preparations have the lowest irreversible shrinkages. This may be due to lower amounts of interlayer Ca++ and closer proximity of silicate sheets in the C-S-H structure.
Intrinsic Mechanical Properties
Modulus of elasticity (E) and d.crohardneas (H) values were determined on compacted C-S-H samples prepared at different porosities. Mechanical property
-
porosity data were fitted to several different models, which have been used to describe the dependence of mechanical properties on porosity for a variety of porous system8 (16-23). Estimates of intrinsic values of E and H (i.e.. zero porosity values, Eo and Ho) were obtained by extrapolation methods. All the models fit the data reasonably well with on8 exception. Modulus of elasticity dALa for the Nielsen model have low values of correlation coefficient for four preparations. Ho and Eo, regardless of the model used for their estimation, do not correlate well with CIS ratio. The highest values of correlation coefficient for Ho vs. CIS ratio are 67.4 and 62.7, obtained using the Nielsen and the Knudsen models, respectively. The shapefactors "a" (Nielsen mo~el) and "b" (Knudsen model) do depend on CIS ratio. The shape factor, "a", for example, increases to a maximum at CIS = 1.0 and then decreases. This dependence may, however, be a consequence of the effects of compaction on pore shape.
In another approach the modulus of elasticity data for all C/S ratios were treated collectively. .ne data were fitted to the Balshin model (24) i.e.
B
-
E 1-p)". The regression equation is E-
34 1 181 (l-p)2-.1, which is very similar tothe expression derived from the Powers d e l (19). Suitable regression curves can be obtained using other models (18-23) in a collective treatment of all the data. The application, however, of the Nielsen and Knudsen models (16.17) does not result in a single curve which describes all the data satisfactorily. bnce, for these models, there is a dependence of the shape factors on CIS ratio. The main conclusion arrived at by treating the data in this way is the same as that obtained with separate data aets for each CIS ratio, 1.e. Eo is independent of CIS ratio and degree of polymeriZation. The Nielsen and Knudsen modele do not predict, as do the others, that the relation E = (p) is independent of
CIS ratio.
On the basis of the statistical analysis of the data for all C-S-H preparations, Eo and Ho appear to be independent of C/S ratio. However, examination of the data (all, models) reveals, for example, that Eo is nearly always higheat for CIS
-
1.26. This C-S-H preparation had the highest HIS ratio and reveraible characteristics in the adsorption region of the weight change isotherm. The presence of interlayer water in the region (11-100% RH) may contribute to rigidity of the C-S-H if. the eilicate sheets are held rigidly by this water.Preparations having CIS = 0.68 also have high E
,
, values. This may be due to the absence of interlayer ~ a + + and cloeer proximity of ailicata sheete. It therefore appears that the degrea of polymerization has little effect on EO. The C-S-H preparation having the highest value of H depends on the predictive model used. The largee? value of Ho (1337 UPa) was estimated by the Nielsen model and was for the preparation having CIS
-
1.26.CONCLUSIONS
I
1. Degree of polymerization of C-S-H depends on CIS
ratio. It decreases as CIS ratio increases.
I
2. Synthetic C-S-H materials prepared using sodiummetasilicate solutions are generally layered systems. Irreversible length and weight change characteristics on exposure to water vapor are due to intercalation processes and aging phenomena.
3. Zero porosity values of modulus of elasticity and microhardness for the C-S-H preparations studied appear to be independent of C/S ratio.
As microhardness generally correlates with strength, values for intrinsic strength would appear to be independent of CIS ratio over the range studied.
4. Highest values of modulus of elasticity were obtained for the preparation (CIS = 1.261, which
exhibited reversible sorption characteristics in the low pressure region of the water isotherm. Intercalation processes and their effects on the proximity of silicate sheets may have % greater effect on intrinsic mechanical properties than composition and/or degree of polymerization.
REFERENCES
I
1. J.J. Beaudoin, Cern. Concr. Res.2
(3). 319-324(1983).
2. H.F.W. Taylor, "The Calcium Silicate Hydrates", The Chemistry of Cements, Chapter 5, 167-232 (1964). Academic Press, Ed. H.F.W. Taylor. 3. J.J. Beaudoin and R.F. Feldman, Cem. Concr.
Res.
5
(2). 103-118 (1975).4. S. Diamond, Proc. Conf. Hydraulic Cement Pastes
-
Their Structure and Properties, hiv. Sheffield. UK, 2 (1976).5. R.F. Feldman, Cem. Technol.
2,
3 (1972). 6. L.J. Parrott and M.G. Taylor, Cem. Concr. Ree.9 483-488 (1979).
-'
7. D.L. Kantro, S. Brunauer and C.H. Weire, M.vances in Chemistry, Series 33, 199 (1962).
1
8. R.P. Feldman, Proc. V. Int. Symp. Chem.
am.,
Tokyo, Part 111, Vol. 111, 53-66 (1968). 9. P.J. Sereda, R.F. Feldman and E.G. Swanson,
High. Res. Bd. Spec. Rpt. 90, 58 (1966). 10. P.J. Sereda, Cem. Concr. Res.
2,
717 (1972)..l.J. Beaudoin and R.F. Feldman, D i v i s i o n of B u i l d i n g Research, N a t i o n a l Reeearch C o i ~ n c i l Canada; J. Baron and M. Conjeaud, b f a r g e Gappi5e Becherche, France
H.F.W. T a y l o r , J e n n i t e , 1.4 nm Tobermorite and C-S-H g e l , Extended A b s t r a c t s , h e r . Cer. Soc. 8 7 t h Meeting, C i n c i n n a t i , 78 (1985).
H. Funk, Autoclaved Calcium S i l i c a t e B u i l d i n g P r o d u c t s , The S o c i e t y of Chemical I n d u s t r y , 122-4 (1967).
A. Bentur, R.G. Berger, J.H. Kung, N.B.
Milestone. J.F. Young, J. Amer. Cer. Soc.
2
C7.8). 362-6 (1979).
H. Uchikawa and R. F u r u t a , Cement Concr. Ree. 11 ( I ) , 65-78 (1951).
-
M. Michaux, D. M e d t r i e r and P. B a r r e t , C.R. Acad. Sc. P a r i s ,
296,
( 2 ) . 1043-6 (1983). L.F. Nieleen, J. h e r . Cer. Soc.67
( 2 ) . 93-98 (1984).F.P. Knudsen, J. h e r . Cer. Soc.
62 ( 8 ) .
376-388 ( 1959).
2. Hashin and B.W. Rosen, J. Appl. Mech., 223-232 (1964).
T.C. Powers, Revue d e s Mat€riaux. (544). 79-85 (1961).
O.P.H. Hasselman, J. Amer. Cer. Soc.
2.
452 (19b2).2. Hashin, J. Appl. Mech., 143-150 (1962).
R. P a u l , Trans. Pletall. Soc. AIME 218, 36-41 (1960).
S.D. Brown, R.B. Biddulph and P.D. Wilcox,
J. Amer. Cer. Spc.,
67
( 7 ) 320-322 (1964).M.Y. a a l s h i n , Dokl. Akad. Nauk SSSR,
z,
831( 1 ' ,
TABLE 1
-
C-S-H CONPOSITION C-S-H D e s i g n a t i o nCompoeition 101 106 107 110 112 113
TABLE 2
-
DENSITY AND SURFACE AREA OFC-S-H PKEPAKATIONS Density S u r f a c e Area C-S-H CIS ( g l m l ) (m2/g)
C
0--
.-
. -n 1982 POWDERED SAMPLES 0.0060 -0 1984 POWDERED SAMPLESO
.
m
~
L
J
0.5 0.6 0.7 (18 0.9 1.0 1.1 1.2 1.3 1.4 1.5 CIS RATIO F i g u r e 1. Polymer rate c o n s t a n t 'bpl v e r s u s CIS r a t i o *powdered samples **compactsJ.J. Beaudoin and R.F. Feldman, M v i s i o n of Building Research, National Research Council Canada; J. Baron and M. Conjeaud, Lafarge Copp6e Recherche, Prance
R E L A T I V E H U M I D I T Y , %
Figure 2. Water adsorption isotherms of C-S-H preparations having d i f f e r e n t C I S r a t i o s
W E I G H T C H A N G E . %