Disruption of the US pre-exposure effect and latent inhibition in two-way active avoidance by systemic amphetamine in C57BL/6 mice
11
0
0
Texte intégral
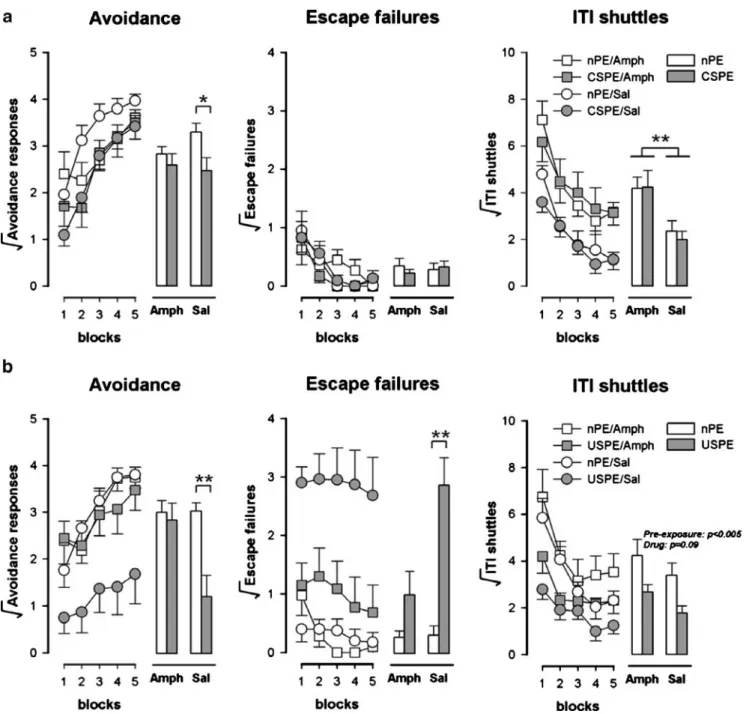
Figure
Documents relatifs