HAL Id: inserm-00396259
https://www.hal.inserm.fr/inserm-00396259
Submitted on 20 Dec 2017
HAL is a multi-disciplinary open access
archive for the deposit and dissemination of
sci-entific research documents, whether they are
pub-lished or not. The documents may come from
teaching and research institutions in France or
abroad, or from public or private research centers.
L’archive ouverte pluridisciplinaire HAL, est
destinée au dépôt et à la diffusion de documents
scientifiques de niveau recherche, publiés ou non,
émanant des établissements d’enseignement et de
recherche français ou étrangers, des laboratoires
publics ou privés.
The p.Asp216His TOR1A allele effect is not found in the
French population.
Mélissa Yana Frédéric, Fabienne Clot, Arnaud Blanchard, Claire-Marie
Dhaenens, Gaetan Lesca, Laura Cif, Alexandra Dürr, Marie Vidailhet,
Bernard Sablonniere, Alain Calender, et al.
To cite this version:
Mélissa Yana Frédéric, Fabienne Clot, Arnaud Blanchard, Claire-Marie Dhaenens, Gaetan Lesca, et
al.. The p.Asp216His TOR1A allele effect is not found in the French population.. Movement Disorders,
Wiley, 2009, 24 (6), pp.919-21. �10.1002/mds.22407�. �inserm-00396259�
5. Krauss JK, Pohle T, Borremans JJ. Hemichorea and hemiballism associated with contralateral hemiparesis and ipsilateral basal ganglia lesions. Mov Disord 1999;14:497 501.
6. Shan DE, Ho DM, Chang C, Pan HC, Teng MM. Hemichorea-hemiballismus: an explanation for MR signal changes. AJNR Am J Neuroradiol 1998;19:863 870.
7. Chang MH, Chiang HT, Lai PH, Sy CG, Lee SS, Lo YY. Puta-minal petechial haemorrhage as the cause of chorea: a neuroi-maging study. J Neurol Neurosurg Psychiatry 1997;63:300 303. 8. Chu K, Kang DW, Kim DE, Park SH, Roh JK.
Diffusion-weighted and gradient echo magnetic resonance findings of hemi-chorea-hemiballismus associated with diabetic hyperglycemia: a hyperviscosity syndrome? Arch Neurol 2002;59:448 452. 9. Midi I, Dib H, Ko¨seoglu M, Afsar N, Gu¨nal DI. Hemichorea
asso-ciated with polycythaemia vera. Neurol Sci 2006;27:439 441. 10. Pisani A, Diomedi M, Rum A, et al. Acanthocytosis as a
predis-posing factor for non-ketotic hyperglycaemia induced chorea-ballism. J Neurol Neurosurg Psychiatry 2005;76:1717 1719. 11. Oh SH, Lee KY, Im JH, Lee MS. Chorea associated with
non-ketotic hyperglycemia and hyperintensity basal ganglia lesion on T1-weighted brain MRI study: a meta-analysis of 53 cases including four present cases. J Neurol Sci 2002;200:57 62. 12. Mestre TA, Ferreira JJ, Pimentel J. Putaminal petechial
haemor-rhage as the cause of non-ketotic hyperglycaemic chorea: a neuropathological case correlated with MRI findings. J Neurol Neurosurg Psychiatry 2007;78:549 550.
13. Suto Y, Mori M, Kagimoto H, Saito J. [A case of hemichorea with hyperglycemia presenting with low signal intensity in the striatum on T2*-weighted gradient-echo magnetic resonance imaging.] Rinsho Shinkeigaku 2004;44:86 90 (in Japanese). 14. Chung SJ, Im JH, Lee MC, Kim JS. Hemichorea after stroke:
clinical-radiological correlation. J Neurol 2004;251:725 729. 15. Ohara S, Nakagawa S, Tabata K, Hashimoto T. Hemiballism
with hyperglycemia and striatal T1-MRI hyperintensity: an au-topsy report. Mov Disord 2001;16:521 525.
16. Chang MH, Li JY, Lee SR, Men CY. Non-ketotic hyperglycae-mic chorea: a SPECT study. J Neurol Neurosurg Psychiatry 1996;60:428 430.
17. Kuroiwa T, Yamada I, Katsumata N, Endo S, Ohno K. Ex vivo measurement of brain tissue viscoelasticity in postischemic brain edema. Acta Neurochir Suppl 2006;96:254 257.
18. Kreis R, Ross BD. Cerebral metabolic disturbances in patients with subacute and chronic diabetes mellitus: detection with MR spectroscopy. Radiology 1992;184:123 130.
19. Graybiel AM. Neurotransmitters and neuromodulators in the basal ganglia. Trends Neurosci 1990;13:244 254.
20. Guyot LL, Diaz FG, O’Rgan MH, Song D, Phillis JW. The effect of streptozocin-induced diabetes on release of excitotoxic and other amino acids from the ischemic rat cerebral cortex. Neuro-surgery 2001;48:385 390.
The p.Asp216His
TOR1A Allele
Effect Is Not Found in the
French Population
Me´lissa Yana Fre´de´ric, PhD,
1,2Fabienne Clot, PhD,
3,4,5,6Arnaud Blanchard, MS,
1,2Claire-Marie Dhaenens, PharmD, PhD,
7,8Gae¨tan Lesca, MD, PhD,
9,10Laura Cif, MD,
11Alexandra Du¨rr, MD, PhD,
3,4,5,6Marie Vidailhet, MD,
PhD,
3,4,5,12Bernard Sablonniere, MD, PhD,
7,8Alain Calender, MD, PhD,
9,10Maria Martinez, PhD,
13Nicolas Molinari, PhD,
14Alexis Brice, MD,
3,4,5,6,12Mireille Claustres, MD, PhD,
1,2,15Sylvie Tuffery-Giraud, PhD,
1,2and Gwenae¨lle Collod-Beroud, PhD
1,2*
1
INSERM, U827, Montpellier, F-34000, France;
2Universite´
MONTPELLIER1, UFR Me´decine, Montpellier, F-34000
France;
3INSERM, UMR_S679 Neurologie & The´rapeutique
Expe´rimentale, Paris, F-75013, France;
4UPMC Univ Paris
06, UMR_S679, Paris, F-75005, France;
5Institut Fe´de´ratif
des Neurosciences (IFR70), Hoˆpital Pitie´-Salpeˆtrie`re, Paris,
F-75013, France;
6AP-HP, Hoˆpital Pitie´-Salpeˆtrie`re,
De´partement de Ge´ne´tique et Cytoge´ne´tique, Paris, F-75013,
France;
7CHRU de Lille, UF de Neurobiologie, Centre de
Biologie-Pathologie, Lille, F-59037, France;
8INSERM,
U837, Institut de Me´decine Pre´dictive et de Recherche
The´rapeutique, Lille, F-59045, France;
9Hoˆpital Edouard
Herriot, Service de Ge´ne´tique Mole´culaire et Clinique, Lyon,
F-69437, France;
10Universite´ Claude Bernard Lyon 1,
Villeurbanne, F-69622, France;
11CHU Montpellier, Hoˆpital
Guy de Chauliac, Service de Neurochirurgie, Montpellier,
F-34000, France;
12AP-HP, Hoˆpital Pitie´-Salpeˆtrie`re,
Fe´de´ration des Maladies du Syste`me Nerveux, Paris,
F-75013, France;
13INSERM, U563, Toulouse, F-31024,
France;
14CHU Nıˆmes, De´partement d’Information Me´dicale,
Nıˆmes, F-30025, France;
15CHU Montpellier, Hoˆpital Arnaud de
Villeneuve, Laboratoire de Ge´ne´tique Mole´culaire,
Montpellier, F-34000, France
Abstract: DYT1 dystonia are one of the exceptions in
human genetics with its unique and recurrent mutation
(c.907delGAG). In this rare movement disorder, the
mutation is associated with incomplete penetrance as well
as great clinical variability, making this disease a
bench-mark to search for genetic modifiers. Recently, Risch
et al. have demonstrated the implication of the rs1801968
SNP in disease penetrance. We attempted to replicate this
*Correspondence to: Gwenae¨lle Collod-Be´roud, INSERM U827, Institut Universitaire de Recherche Clinique, 641 av du doyen Gaston Giraud, 34093 Montpellier Cedex 05, France.
E-mail: gwenaelle.collod-beroud@inserm.fr Potential conflict of interest: None reported.
Received 3 April 2008; Revised 16 October 2008; Accepted 5 Novem-ber 2008
Published online 3 March 2009 in Wiley InterScience (www. interscience.wiley.com). DOI: 10.1002/mds.22407
919
p.Asp216His TOR1A ALLELE EFFECT IN THE FRENCH POPULATION
result in an exhaustive DYT1 French population with no
success. Our results argue that the rs1801968 H allele
effect is not part of the modifiers in the French
popula-tion of DYT1 patients and that others have to be
identi-fied in our population.
Ó 2009 Movement Disorder Society
Key words: TOR1A; DYT1; population studies; genetic
modifiers; movement disorders; dystonia
In a recent American report (published in June 2007),
Risch and colleagues looked for both
trans and cis
effects of rs1801968
TOR1A SNP in DYT1 dystonia.
1This study is based on previous experiments of Kock
et al.
2on torsinA, the protein encoded by the
TOR1A
gene and supposed to have a role in nuclear envelope
and endoplasmic reticulum organization.
3Kock et al.
have demonstrated that cells overexpressing torsinA
with the rs1801968 H allele (called 216H allele)
devel-oped inclusions that were reduced when the
c.907del-GAG mutation was coexpressed (216H allele was
intro-duced in
cis of the mutation). In their study, Risch
et al. analyzed 22 index cases and their family members
(population corresponding to 17 independent families of
European descent including 2/3 with Ashkenazi origin
as well as 3 Asian, 1 African American, and 1 Mexican
families). They showed that the rs1801968 H allele
(216H allele) was more frequent in
trans in
asymptom-atic
TOR1A mutation carriers (Non Manifesting
Car-riers, NMC) (Table 1, *) and less frequent in
sympto-matic carriers (Manifesting Carriers, MC) (Table 1,
$),
when compared to controls (Normal Controls, NC)
(Ta-ble 1,
€). They deduced from these results a highly
pro-tective effect of the H allele when in
trans with the
c.907delGAG deletion. Furthermore, as they found the
rs1801968 D allele (216D allele) in the 21/22
independ-ent c.907delGAG carrier chromosomes studied (one
individual was not typed: ‘‘D or H ?’’), they suspected
that allele D was required in
cis for disease penetrance.
This article opened a new avenue with the first
descrip-tion of a clinically relevant modifier of DYT1 dystonia.
We have started a large and exhaustive study of
TOR1A mutation carriers in France
4after the surprising
demonstration of the scarcity of the mutation in a
pop-ulation of 12,000 French newborns.
5Fifty-three index
cases carrying the c.907delGAG mutation have been
identified. Most of the families are Caucasian and of
European ancestry. Ashkenazi Jew origins are reported
in eight families. Four additional families are of
Magh-rebian origin and one of Caribbean origin. We have
confirmed that
TOR1A-linked dystonia clinical
mani-festations probably vary according to different
ethnic-ities, suggesting population-specific disease-modifying
factors. We then attempted to replicate the results from
Risch and colleagues in our population. Haplotypes
linked to the mutation (cis) or inherited from the
non-carrier parent (trans) were determined from 53
inde-pendent families. Seventy-five
TOR1A symptomatic
mutation carriers and 20 asymptomatic mutation
car-riers were included. The rs1801968 allele frequency
was also analyzed in a control population of
compara-ble size (227 nonrelated individuals versus 197 in
Risch et al.). No difference in the allele distribution
was observed between the two control populations. We
first found that all carriers of the
TOR1A mutation also
had the rs1801968 D allele in
cis, as described in the
American study (Table 2). However, when looking at
trans effect of this specific allele, we did not find any
H allele in asymptomatic carriers of the mutation. This
H allele distribution was not related to a particularly
TABLE 1. Allele frequencies for rs1801968 SNP in trans (non c.907delGAG carrier chromosomes)
Symptomatic carriers (manifesting carriers) Asymptomatic carriers (nonmanifesting carriers) Control individuals (normal controls)
D allele H allele D allele H allele D allele H allele
French study 75 MC’s chromosomes, 20 NMC’s chromosomes, 454 NC chromosomes 73 (97%) 2 (3%) 20 (100%) 0 404 (89%) 50 (11%) American study 119 MC’s chromosomes, 113 NMC’s chromosomes, 394 NC chromosomes 117 (98.3%) 2 (1.7%)$ 89 (78.8%) 24 (21.2%)* 337 (85.6%) 57 (14.4%)€
German and Italian population studies 42 MC’s chromosomes,
24 NMC’s chromosomes
42 (100%) 0 Likely:
18 (75%) 6 (25%)
Not done
Note that in the American and the French studies, the specific allele associated intrans of the mutation has been determined, whereas it was not specified in the German and Italian population studies.
Movement Disorders, Vol. 24, No. 6, 2009
low frequency in the French population as no difference
in the allele distribution was observed between the two
control populations. Even if the population studied is
smaller than the one of Risch et al. (20 versus 113,
respectively),
the
difference
in
the
distribution
of
rs1801968 alleles between these two populations is
statis-tically significant (Fisher’s Exact Test
P
5 0.0237).
Very recently, the American study has been
con-firmed in a mixed German and Italian population of 42
symptomatic individuals carrying the
TOR1A
c.907del-GAG deletion and 24 asymptomatic mutation carriers
from 35 families.
6Unfortunately, haplotypes linked to
the mutation (cis) or inherited from the noncarrier
par-ent (trans) could not be determined (C. Kamm,
per-sonal communication). They identified the 216H allele
in asymptomatic patients in 6/48 chromosomes (trans
or
cis). Nevertheless, as all the symptomatic carriers of
the
TOR1A mutation have been demonstrated to be
homozygous for the 216D allele, it is likely that the
asymptomatic carriers from the same families have
also a 216D allele in
cis. The six 216H alleles would
then be localized in
trans of the mutation.
In the present study, we did not observe the high
frequency of rs1801968 H allele in nonmanifesting
car-riers of the c.907delGAG mutation, reported by others,
suggesting that other modifiers should explain the
incomplete penetrance of the c.907delGAG mutation in
the French population.
Acknowledgments: M.Y.F. is supported by a grant from
AFM (Association Franc¸aise contre les Myopathies). This
work was financially supported by the LFCD-AMADYS and
INSERM Dystonia National Network and Gis Maladies
Rares. We thank all patients and family members for the
par-ticipation in this study.
Author Contributions: M.Y. Fre´de´ric: Conception and
design, Acquisition of data, Analysis and interpretation of
data, Critical revision of all or part of the submitted
publica-tion material for important intellectual content,
Administra-tive, technical or material support. F. Clot: Acquisition of
data, Drafting of all or part of the submitted publication
ma-terial, Administrative, technical or material support. A.
Blan-chard: Acquisition of data, Analysis and interpretation of
data, Critical revision of all or part of the submitted
publica-tion material for important intellectual content,
Administra-tive, technical or material support. C.-M. Dhaenens:
Acquisi-tion of data, Drafting of all or part of the submitted
publica-tion material, Administrative, technical or material support.
G. Lesca: Acquisition of data, Critical revision of all or part
of the submitted publication material for important
intellec-tual content. L. Cif: Acquisition of data, Critical revision of
all or part of the submitted publication material for important
intellectual content. A. Du¨rr: Acquisition of data, Drafting of
all or part of the submitted publication material, Supervision.
M. Vidailhet: Acquisition of data, Critical revision of all or
part of the submitted publication material for important
intel-lectual content, Supervision. B. Sablonniere: Acquisition of
data, Drafting of all or part of the submitted publication
ma-terial, Administrative, technical or material support. A.
Cal-ender: Conception and design, Drafting of all or part of the
submitted publication material. M. Martinez: Analysis and
interpretation of data, Drafting of all or part of the submitted
publication material, Statistical expertise. N. Molinari:
Analy-sis and interpretation of data, Drafting of all or part of the
submitted publication material, Statistical expertise. A. Brice:
Acquisition of data, Drafting of all or part of the submitted
publication material, Supervision. M. Claustres: Acquisition
of data, Critical revision of all or part of the submitted
publi-cation material for important intellectual content,
Administra-tive, technical or material support, Obtaining funding. S.
Tuffery-Giraud: Analysis and interpretation of data, Critical
revision of all or part of the submitted publication material
for important intellectual content, Administrative, technical
or material support. G. Collod-Beroud: Conception and
design, Acquisition of data, Analysis and interpretation of
data, Critical revision of all or part of the submitted publication
material for important intellectual content, Obtaining funding,
Administrative, technical or material support, Supervision.
REFERENCES
1. Risch NJ, Bressman SB, Senthil G, Ozelius LJ. Intragenic cis and trans modification of genetic susceptibility in DYT1 torsion dystonia. Am J Hum Genet 2007;80:1188 1193.
2. Kock N, Naismith TV, Boston HE, et al. Effects of genetic varia-tions in the dystonia protein torsinA: identification of polymor-phism at residue 216 as protein modifier. Hum Mol Genet 2006; 15:1355 1364.
3. Kamm C, Ozelius L, Breakefield X. TorsinA and DYT1 early-onset dystonia. Future Med 2008;3:61 72.
4. Frederic MY, Clot F, Cif L, et al. Is the early-onset torsion dys-tonia (EOTD) linked to TOR1A gene as frequent as expected in France? Neurogenetics 2008;9:143 150.
5. Frederic M, Lucarz E, Monino C, et al. First determination of the incidence of the unique TOR1A gene mutation, c.907del-GAG, in a Mediterranean population. Mov Disord 2007;22: 884 888.
6. Kamm C, Fischer H, Garavaglia B, et al. Susceptibility to DYT1 dystonia in European patients is modified by the D216H poly-morphism. Neurology 2008;70:2261 2262.
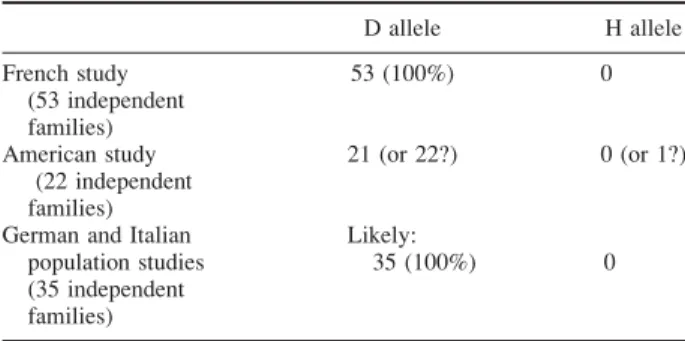
TABLE 2. Allele frequencies for rs1801968 SNP in cis
(c.907delGAG carrier chromosomes)
D allele H allele French study (53 independent families) 53 (100%) 0 American study (22 independent families) 21 (or 22?) 0 (or 1?)
German and Italian population studies (35 independent families)
Likely:
35 (100%) 0
Note that in the American and the French studies, the specific al-lele associated incis of the mutation has been determined, whereas it was not specified in the German and Italian population studies.