Timeline representation of clinical data usability and added value for pharmacovigilance
Texte intégral
Figure
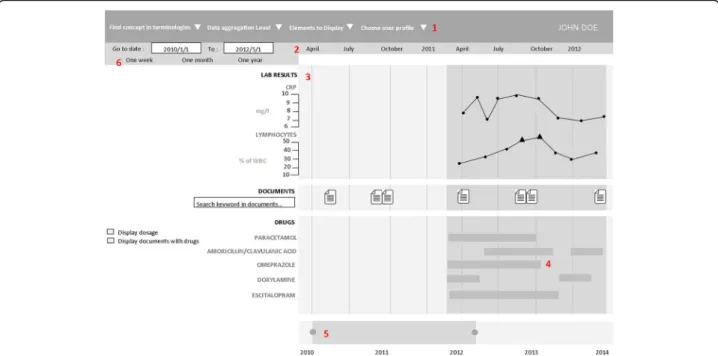
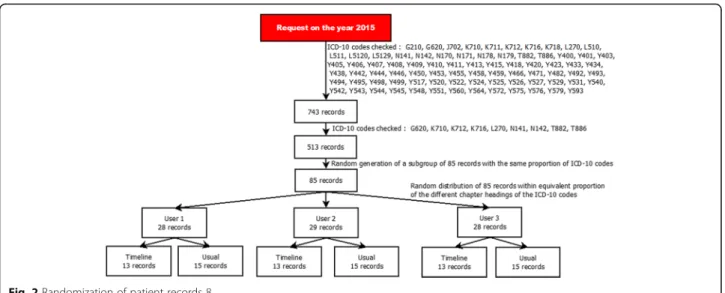

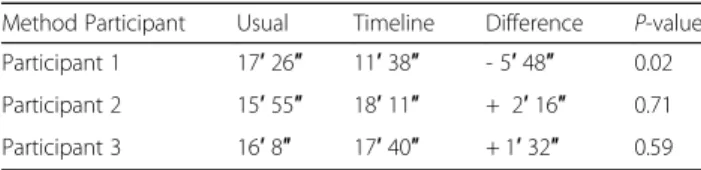
Documents relatifs
The following ATC codes and DDDs were agreed at the meeting of the WHO International Working Group for Drug Statistics Methodology in October 2018.. Comments or objections to
The following ATC codes and DDDs were agreed at the meeting of the WHO International Working Group for Drug Statistics Methodology in March 2019.. Comments or objections to
amoxicillin and enzyme inhibitor amoxicillin and beta-lactamase inhibitor J01CR02 ampicillin and enzyme inhibitor ampicillin and beta-lactamase inhibitor J01CR01 Bile
The following ATC codes and DDDs were agreed at the meeting of the WHO International Working Group for Drug Statistics Methodology in October 2017.. Comments or objections to
Comments or objections to the decisions from the meeting should be forwarded to the WHO Collaborating Centre for Drug Statistics Methodology (whocc@fhi.no) before 1 September 2015.
The following ATC codes and DDDs were agreed at the meeting of the WHO International Working Group for Drug Statistics Methodology in March 2014.. Comments or objections to
The following ATC codes, DDDs and alterations were agreed at the meeting of the WHO International Working Group for Drug Statistics Methodology in October 2013.. Comments
where kcal is the kilocalorie for Period of a day, is ration for , and AL is activity level category of patient. The periodic kilocalorie intake is further shared to three macro
