HAL Id: hal-01692699
https://hal.archives-ouvertes.fr/hal-01692699
Submitted on 25 Jan 2018
HAL is a multi-disciplinary open access
archive for the deposit and dissemination of
sci-entific research documents, whether they are
pub-lished or not. The documents may come from
teaching and research institutions in France or
abroad, or from public or private research centers.
L’archive ouverte pluridisciplinaire HAL, est
destinée au dépôt et à la diffusion de documents
scientifiques de niveau recherche, publiés ou non,
émanant des établissements d’enseignement et de
recherche français ou étrangers, des laboratoires
publics ou privés.
Corrosion behaviour of an assembly between an AA1370
cable and a pure copper connector for car manufacturing
applications
Rosanne Gravina, Nadine Pébère, Adrien Laurino, Christine Blanc
To cite this version:
Rosanne Gravina, Nadine Pébère, Adrien Laurino, Christine Blanc. Corrosion behaviour of an
as-sembly between an AA1370 cable and a pure copper connector for car manufacturing applications.
Corrosion Science, Elsevier, 2017, vol. 119, pp. 79-90. �10.1016/j.corsci.2017.02.022�. �hal-01692699�
O
pen
A
rchive
T
OULOUSE
A
rchive
O
uverte (
OATAO
)
OATAO is an open access repository that collects the work of Toulouse researchers and
makes it freely available over the web where possible.
This is an author-deposited version published in :
http://oatao.univ-toulouse.fr/
Eprints ID : 19370
To link to this article : DOI:
10.1016/j.corsci.2017.02.022
URL :
http://dx.doi.org/10.1016/j.corsci.2017.02.022
To cite this version :
Gravina, Rosanne and Pébère, Nadine and Laurino, Adrien and
Blanc, Christine Corrosion behaviour of an assembly between an
AA1370 cable and a pure copper connector for car manufacturing
applications. (2017) Corrosion Science, vol. 119. pp. 79-90. ISSN
0010-938X
Any correspondence concerning this service should be sent to the repository
administrator:
staff-oatao@listes-diff.inp-toulouse.fr
ContentslistsavailableatScienceDirect
Corrosion
Science
jo u r n al ho m e p a g e :w w w . e l s e v i e r . c o m / l o c a t e / c o r s c i
Corrosion
behaviour
of
an
assembly
between
an
AA1370
cable
and
a
pure
copper
connector
for
car
manufacturing
applications
Rosanne
Gravina
a,b,
Nadine
Pébère
a,
Adrien
Laurino
b,
Christine
Blanc
a,∗aCIRIMAT,UniversitédeToulouse,CNRS,INPT,UPS,ENSIACET,4alléeEmileMonso,CS44362,31030Toulouse,France bLEONIWiringSystemsFrance,5avenuedeNewton,78180Montigny-le-Bretonneux,France
Keywords: A.Aluminium A.Copper B.EIS C.Crevicecorrosion C.Pittingcorrosion
a
b
s
t
r
a
c
t
ThecorrosionbehaviourofanassemblybetweenanAA1370cableandapurecopperconnectorforwiring harnesseswasstudiedinneutralchlorideandsulphatecontainingsolution.Electrochemicalimpedance measurementsshowedthatthecorrosionbehaviourofthecablewascontrolledbytheingressofthe elec-trolyteinsidecablecavities.Further,localimpedancemeasurementswereperformedontwoassembly cross-sections,i.e.withandwithoutcavitiesinthealuminiumcable.Theresultsprovidedevidencefor boththegalvaniccouplingbetweenaluminiumandcopperandthepresenceofcavitiesinthealuminium cableasrelevantexplanationsforthecorrosionbehaviouroftheassembly.
©
1. Introduction
Typicalwiringharnessesintheautomotiveindustryconsistof assemblybetweenCucableandCuconnector.Today,thepressure inenvironmentalregulationsledtosearchforsolutionsgivingrise tofueleconomyandreductioninCO2emission.Inthiscontext,car
manufacturersplannedtoreducebothcostandweightofwiring harnesses.OneinnovativesolutionisthesubstitutionofCubyAl alloys,suchasAA1370,incables.Inthelastyears,severalworks concernedthemanufacturingprocessesofthisnewtypeof assem-bly.Recently,differentmethodshavebeenproposedtoproduceAl strandswithductilityandfatiguecharacteristicsreliablefor auto-motivewiringharnesses[1–4]whileultrasonicweldinghasbeen usedtojoinAlcablewithCuconnector[5,6].However,onemajor problemforwiringharnessesconcernstheircorrosionresistance because,in service,wiring harnesses are exposedto aggressive media,suchasde-icingsalt,whichcangeneratecorrosiondamage. Numerousdataarereportedintheliteratureconcerningthe cor-rosionbehaviourof1xxxAlalloys.Themainresultsconcernedthe influenceofFe-richparticleswhichactascathodicsitesand pro-motefirstdissolutionofthesurroundingmatrixandthen,pitting corrosion[7–9].However,thecorrosionbehaviourofanAlcableis morecomplex.Intheliterature,onlyfewworkshavebeenreported
∗ Correspondingauthor.
E-mailaddress:christine.blanc@ensiacet.fr(C.Blanc).
concerningthecorrosionbehaviourofacable.XuandChen inves-tigatedthecorrosiondamageofwirespecimenspulledfromthe replacedcablesinShimenBridge[10].Theyanalysedthecorroded cablesfromthesheathbreakagetothecablecenterandexplained thattheextentofcorrosionatanypointofacablesectionwas con-trolledbyitsdistancefromthesheathbreakageduetothefluidthat couldpenetrateinsidethecable[10].IshikawaandKawakami[11]
showedthepresenceofcavitiesinsideacableduetothe incom-pletepenetrationofthesheathrubber.Then,duringexposuretoan aggressivemedium,thesolutionpenetrateintothecablecavities leadingtotheformationofaconfinedenvironment.Dependingon theconfigurationoftheharness,e.g.numberofstrandarms con-stitutiveofthecableandspacebetweenthem,andtheintrinsic corrosionbehaviourofthestrand,theconfinedelectrolyte com-positionshouldevolverapidly,e.g.,oxygenconcentration,cations concentrationandpH,andtheninduceseverecorrosion phenom-ena,suchascrevicecorrosion[12–14].ChanelandPeberestudied themechanismsgoverningthedegradationofbrass-coatedsteel cords fortyresin a 0.25M Na2SO4 solution incontact withair
andmaintainedat25◦Cbyusingelectrochemicalimpedance
spec-troscopy(EIS)[15].TheyshowedthatEISwasasuitabletechnique toquantifythecorrosionresistanceofacable[15].Moreover,the assemblybetweenanAlcableandaCuconnectorshould gener-ategalvaniccorrosionduetothedifferenceincorrosionpotential valuesbetweenthetwometalsinaqueoussolution[16–21].Khedr andLashieninvestigatedthecorrosionbehaviourofpureAlinCu2+
richsolutionsandshowedtheacceleratingeffectoftheCu2+cations
http://dx.doi.org/10.1016/j.corsci.2017.02.022
AlcorrosionrateduetoCudepositionandsubsequentgalvanic coupling[17,18].Jorcinetal.[19]showed,foramodelcouple con-stitutedofAlandCu,thechemicaldissolutionofAlduetoapH increaseattheAl/Cuinterface;thisgeneratedanoccludedzonein whichthecompositionoftheelectrolyte(a10−3MNa
2SO4
solu-tionincontactwithairandatroomtemperature)evolvedrapidly. ThegalvaniccouplingbetweenAlandCuwasalsoinvestigatedina 10mMNa2SO4solutionbyusingathin-layercellinordertomodel
theoccludedzoneinthecrevice[20].Jomaetal.performed exper-imentsina0.1MNa2SO4solutionandshowedthatthechemistry
inathin-layercellplaysasignificantroleatleastatalocalscale
[21].
The present work contributes to get a better insight of the mechanismsgoverningthedegradationofanAA1370cable/pure Cu assembly with a particular attention to the effect of the electrolyteingressinsidethecable.Conventionalelectrochemical measurements(Ecorrmeasurements,polarisationcurvesand
elec-trochemicalimpedancespectroscopy)werefirstcarriedoutforthe AA1370cableinordertoinvestigatethecorrosionbehaviourofthe cablealone.Giventhatthecablewasmadeofalargenumberof strands,theelectrochemicalmeasurementswerealsoperformed forAA1370strandstohaveabetterunderstandingofthe corro-sionmechanisms.Then,toinvestigatethecorrosionbehaviourof theAA1370cable–Cuconnectorassembly,localelectrochemical impedance(LEIS)measurementswereperformedontwo cross-sectionsof theassembly,onecross-section correspondingtoan AA1370cablewithcavitiesandtheotheronewithoutcavities.LEIS isanon-destructiveelectrochemicaltechniquethathasbeenused inrecentyearstostudylocalizedcorrosiononbimetallicsurface
[19,22–26].Itwasusedheretodeterminetheinfluenceofthe
cav-itiesintheAA1370cableafterweldingonthegalvaniccorrosion processesoccurringfortheAA1370cable/Cuconnectorassembly.
2. Experimental 2.1. Materials
TheassemblywasobtainedbyultrasonicweldingusingaDS 20-IIapparatusbetweenanAA1370(99.7%Al,0.072%Fe,0.0045%Mg, 0.045%Si;wt.%)cableandapureCu(99.90wt.%Cu;200–400ppm O2)connector.TheCuconnectorwasahotstampedCusheet.In
thefollowing,theAA1370cableintheassemblywillbereferred toas‘theAlpart’;theAA1370cable/Cuconnectorassemblywillbe referredtoasAl/Cuassembly.Duringtheweldingprocess,thecable (withoutthepolymershell)waspositionedontheCuconnectorand apressurewasexerted.Then,ultrasonicvibrationswereappliedso thatthecableandtheCuconnectormovedrelativelytooneanother withanoscillatingmovement.Thisledtoafrictionbetweenthetwo metalsandthentotheformationofaweldingzone.Thetotallength oftheweldedzonewasabout12mm.Thequalityofthewelding dependsmainlyontheappliedpressureandontheamplitudeof thevibrations.Therewasashortzone(8mm)wherethestrands constitutiveofthecablewereentirelyweldedtogethersothatthe cabledidnotrevealanycavitiesafterthewelding,thiszonebeing calledasthe‘effectiveweldedzone’.
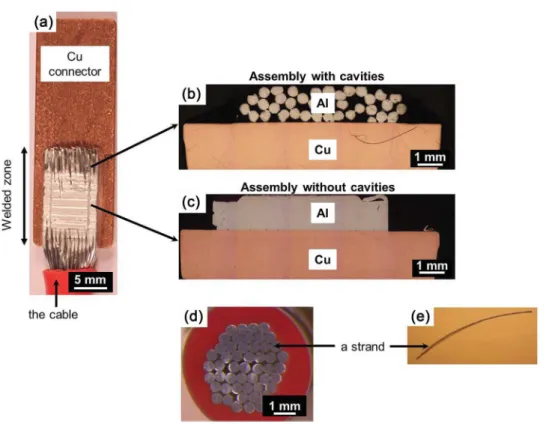
Thegalvanic couplingat theAl/Cu interfacewas studiedby electrochemicalmeasurementsperformedontwocross-sections oftheassembly.Fig.1ashows aglobalpictureoftheassembly. Thetwoselectedcross-sectionsareshowninFig.1bandc.Oneof thecross-sectionswasremovedfromaporouszoneofthecable due tothe fact that thestrands constitutive of the cablewere not entirelyweldedtogether (henceforthcalled ‘assemblywith cavities’)(Fig.1b).Anothercross-sectionwasremovedfromthe effectiveweldedzonewherenocavitieswereobservedbetween theweldedstrands(henceforthcalled‘assemblywithoutcavities’)
(Fig.1c).Beforecorrosiontests,thecross-sectionswereembedded inanepoxy-resinwithoutfillingthecavities.Thiswasachievedby embeddingthecross-sectionsinaparafilmshell.
Fora betterunderstandingofthecorrosionbehaviourofthe assembly, electrochemical measurements were also performed forcross-sectionsofboththeAA1370cablebeforewelding and AA1370strandsconstitutiveofthecable.Thecross-sectionswere perpendiculartothecable/strandaxis.Thecablewasanassembly of50strands(0.52mmdiameter)protectedbyaplastic insulat-ingsleeve.Fig.1dshowsanopticalobservationofacross-section ofacablewhere the50strandscanbeobserved.Itcanbeseen that thespacebetweenthestrands isnot uniform. Toperform theelectrochemicalmeasurements,thecablewasembeddedin anepoxy-resinwithoutfillingthespacebetweenthestrands(see theprocedurefortheassembly).Ifonlythesurface perpendicu-lartothecableaxiswastakenintoaccount,thesurfaceareaof thecross-sectionofthecablewith50strandsexposedtothe elec-trolyteduringtheelectrochemicalexperimentswas0.11cm2.To
studythecorrosionbehaviourofastrand,thedifficultywasits diameter,i.e.0.52mm.Threeelectrodeswerepreparedwiththe cross-sectionsof1strand(S=0.002cm2),4strands(S=0.008cm2)
and22 strands(S=0.047cm2)respectivelyexposed tothe
elec-trolyte.Forthe3electrodes,thestrandswereorganizedtoobtaina regulararrangementoftheircross-sections(theaxisofeach indi-vidualstrandbeingparalleltotheothers)andembeddedtogether inanepoxy-resin.Allthecavitiesbetweenthestrandswerefilled bytheepoxy-resin.Preliminaryexperimentsshowedthatsimilar resultswereobtainedforthe3electrodes.However,the repro-ducibilityofthemeasurementswasbetterfortheelectrodewith 22strandsduetothefactthatthesurfacewasmorerepresentative ofthealloymicrostructure.Inthefollowing,forbrevity,onlythe resultsfortheelectrodewith22strandsarepresented.This elec-trodewasreferredtoas‘strandsample’.Itcouldbenotedherethat thecorrosionbehaviourofthestrandsamplecouldbeconsidered representativeofthebehaviourofacableforwhichthecavities wouldbesealed.Electrochemical measurementswerealso per-formedforaCusampletakenoffaconnectorusedfortheassembly; thesurfaceexposedtotheelectrolytewas0.11cm2.
Beforeallelectrochemicaltests,thesurfaceofthespecimens wasmechanicallyabradedwithsuccessivegritSiCpapers(1200, 2400)andthenpolishedfrom6to1mmgradewithdiamondpaste anddeionisedwateraslubricant.
2.2. Electrochemicalmeasurements
Allelectrochemicalmeasurementswereperformedinsulphate and/orchloride-containingsolutions;theyareassumedtobe rep-resentativeoftheautomotiveenvironments.Severalauthorshave usedsulphateand/orchloride-containingsolutionstostudyAl/Cu galvaniccouplingallowingtheresultsobtainedinthisworktobe comparedwiththeirresults[20,21].Fortheconventional electro-chemicalmeasurements(Ecorrmeasurements,polarizationcurves
andimpedancemeasurements),thecorrosivemedium(pH=6.5) waspreparedfromdeionisedwaterbyadding0.1MNa2SO4 and
asmallconcentrationofchloride(0.001MNaCl).ForLEIS experi-ments,a0.001MNaClsolution(pH=6.5)preparedfromdeionised water was chosen to maintain a low conductivity required to optimize the measurements in the low-frequency range [27]. Theelectrolyte wasmaintained ata temperatureof 25◦C±1◦C
exceptfortheLEISmeasurementsperformedatroomtemperature (22◦C±1◦C).
Fortheconventionalelectrochemicalmeasurements,the exper-imentalset-upconsistedofathree-electrodecell,connectedtoa BiologicVSPapparatus,withalargeplatinumelectrodeusedas counterelectrodeandasaturatedcalomelelectrode(SCE)as ref-erenceelectrode.Specimensusedastheworkingelectrodewere
Fig.1.(a)GlobalviewoftheassemblybetweenAA1370cableandpureCuconnector;cross-sectionsoftheassemblies(b)withand(c)withoutcavities;(d)theAA1370cable and(e)astrand.
thestrandsampleandthecable.Forthetwocross-sectionsofthe assembly,onlyEcorrwasmeasuredforexposuretimestothe
elec-trolyterangingfrom1hto168h.In thefigures,meanvaluesof Ecorr aregivenonthebasisof atleasttenexperimentsforeach
experimentalcondition.Forthepolarizationcurves,thesamples werefirstexposedtotheelectrolyteatEcorrfor3handthen,the
anodicandcathodicpartswereobtainedindependentlyfromEcorr
atapotentialsweeprateof0.07mVs−1.Foreachsample,atleast
threecurveswereplottedtocheckthereproducibility.Impedance measurementswereperformedunderpotentiostaticconditionsat Ecorrwitha15mVpeak-to-peaksinusoidalperturbation.Frequency
wassweptdownwardsfrom65kHzto3mHzwith9pointsper decade.Severalimpedancediagramswererecordedasafunction oftimerangingfrom3hto168h.Allimpedancemeasurements wererepeatedthreetimestocheckthereproducibility.
The corrosion behaviourof the Al/Cu assembly wasstudied bylocalelectrochemicalimpedancespectroscopy(LEIS).The mea-surementswerecarriedoutwithaSolartron1287Electrochemical Interface,a Solartron1255B frequencyanalyser anda Scanning Electrochemical WorkstationModel 370(Uniscan Instruments). Thismethodusedafive-electrodeconfiguration.Theprobe(i.e., abi-electrodeallowinglocalcurrentdensitymeasurements)was steppedacrossaselectedareaofthesample.Theanalysed part had anareaof 8mm×14mmandthestepsizewas500mmin thexandydirections.Mapswereobtainedatafixedfrequency, choseninthepresentcaseat10Hz,andadmittancewasplotted ratherthanimpedancetoimprovethevisualizationoftheresult
[28].Localimpedancediagramswererecordedoverafrequency rangeof65kHz–3Hzwith8pointsperdecade.Spectrawere plot-tedforthe2cross-sectionsoftheassemblyshowninFig.1bandc fromtheCuparttotheAlpartwiththeoriginofaxisbeingtheAl/Cu interface.Thetimetorecordallthelocalimpedancediagramswas lowerthan60min.ForalltheLEISmeasurements,thespatial reso-lutionwasabout1mm2,i.e.theanalysedsurfacewasabout1mm2
whenalocalimpedancediagramwasplotted[27].Fortheassembly
withcavities(Fig.1b),thecavitiescorrespondedto20%ofthe ana-lysedsurfacesothattheeffectivemetallicsurfacewasonly80%of theanalysedsurface(analysisperformedonthecross-section per-pendiculartothecableaxis).Inthefollowing,forLEISresults,the impedancevalues(incm2)arenotcorrected.Therefore,forthe
assemblywithcavities,theimpedancevaluesareoverestimated withanerrorofabout20%ifonlythesurfaceexposedtothebulk solutionistakenintoaccount.
2.3. Surfacecharacterization
AA1370surfaceswereobservedbeforeandafterthe electro-chemicaltestsbyusingaNikonEclipseMA200opticalmicroscope (OM).ALeo435VPscanningelectronmicroscope(SEM)wasused tovisualizetheAl/Cuassemblyafterdifferentexposuretimesto theelectrolyticsolutionandtoobtainabetterdescriptionofthe corrosionmorphology,particularlyattheAl/Cuinterface.Forall theobservations,thesampleswereremovedfromtheelectrolyte aftertheelectrochemicaltests,rinsedwithdeionised waterand thenair-dried.
3. Experimentalresultsanddiscussion
First, the corrosion behaviour of the cable, as compared to the strand sample, was studied by combining stationary and impedancemeasurements.Then,attentionwaspaidtothe corro-sionbehaviouroftheAl/Cuassembly.
3.1. ElectrochemicalbehaviouroftheAA1370cable
Fig.2showsOMobservationsofastrandandofthecableafter twoexposuretimes(3hand168h)totheaggressivesolutionat theircorrosionpotential.Forbothsamples,aweakdissolutionof thematrixaroundtheFe-richparticles,identifiedasAl3Fe
Fig.2.Opticalmicroscope(OM)observationsofthestrandandofthecableafter:(a,b)3hofimmersionand(c,d)168hofimmersionatEcorrin0.1MNa2SO4+0.001MNaCl. (a)and(c)correspondtothestrand,(b)and(d)tothecable.(e)SEMobservationofcorrosionphenomenaoccurringattheAl3Feintermetallicparticles.(f)close-uponthe corrosionfilamentsobserved.
bshowedthatafter3hthematrixdissolutionaroundthe inter-metallicparticleswasmoreextendedforthecablethanforthe strand.After168hofimmersion(Fig.2candd),asignificant dis-solutionofthematrixaroundtheFe-richparticles(Fig.2e)canbe seenonthewholesurfaceforbothsamples,andinaddition,for thecable,largepitssurroundedbyfinefilaments(Fig.2f)werealso observedattheperipheryofthestrands.Thesecorrosionfeatures, inagreementwithliteraturedata,accountedforthepenetration oftheelectrolyteinsidethecableleadingtoaconfinedmedium andcrevicecorrosion phenomenon[10,11].OMobservations of cross-sections parallel to the cable axisafter various exposure timestothechloride-containingsulphatesolutioncorroborated thisassumptionandallowedthedepthofthecorrodedzoneto bemeasured(Fig.3a).Itwasassumedtobeagoodindicationof theelectrolytepenetrationdepth.Inordertomeasurethedepthof thecorrodedzone,thecorrosiondamagewasenhancedby polar-izingat−700mV/SCEfor5minthesamplesaftertheexposureat Ecorr(Fig.3aandb).Theelectrolytepenetrationdepthwasfoundto
becomerapidlyindependentoftheexposuretime,reachingabout 800mmafter3hofexposure(Fig.3c).
Fig.4 reports Ecorr values versus exposuretime tothe
elec-trolyteforthestrandsampleandthecable.Forthestrandsample, Ecorr stabilized rapidly and an almost stationary value(around
−600mV/SCE)wasobtainedafter3hofimmersion.Onthe con-trary, for the cable, Ecorr decreased significantly towards more
negativevaluesduringimmersionwhichwasinagreementwith thedifferencesincorrosionmorphologyobservedbetweenthetwo samples(Fig.2).Fig.5showsthepolarizationcurvesforthestrand sampleandthecableobtainedafter3hofexposuretothe chloride-containingsulphatesolution.Forthecable,thecurrentdensities werecalculatedbytakingintoaccount(i)onlythecross-sections ofthe50 strands constitutiveof thecable,i.e.only thesurface exposedtothebulksolution(S=0.11cm2,nocorrectedsurfacein
Fig.5)and(ii)boththecross-sectionsandthelateralsurfaceof the50strandsona800mmdepth,i.e.boththesurfacesexposed tothebulkand toaconfinedelectrolyte(S=0.76cm2,corrected
Fig.3.(a)OMobservationofacablecross-section(paralleltothecableaxis)after3hofexposuretothe0.1MNa2SO4+0.001MNaClsolutionfollowedbypolarizationfor 5minat−700mV/SCE;(b)close-uponacorrodedstrandinsidethecable;(c)electrolytepenetrationdepthversustheexposuretime.
Fig.4.VariationofEcorrduringimmersionin0.1MNa2SO4+0.001MNaClforthe strandsampleandthecable.
Fig.5.Polarizationcurvesofthestrandsampleandthecableobtainedafter3h ofimmersionatEcorrin0.1MNa2SO4+0.001MNaCl.Scanrate=0.07mVs−1.For thecable,thecurve‘nocorrectedsurface’onlytakesintoaccountthecablesurface exposedtothebulk(S=0.11cm2);the‘correctedsurface’curvetakesintoaccount boththesurfaceexposedtothebulkandthatexposedtotheconfinedmedium insidethecavities(S=0.76cm2).
surfaceinFig.5).Forboththestrandsampleandthecable,the cathodicbranch,correspondingmainlytotheoxygenreduction, wassimilar.Forthestrandsampleandthecable,Ecorrvalueswere
−580mV/SCEand−730mV/SCErespectively.Theanodicdomain wascharacterizedforthestrandsample bya passivityplateau, withpassivecurrentdensitiesofabout4.10−4mAcm−2,followed
byabreakdownpotentialat500mV/SCEassociatedwith,first,a slowincreaseoftheanodiccurrentdensitiesandthenasharper one.Thiscouldbeassociatedto,first,anincreaseofthe dissolu-tionrateofthematrixsurroundingtheFe-richparticlesandthen topittingcorrosion.Besides,thesignificantcurrentfluctuations observedafterthesharpincreaseofthecurrentdensities consti-tutesacharacteristicfeatureofthepittingcorrosion.Forthecable, ashortpseudo-plateauwasobservedandtheanodiccurrent densi-tieswereabout2.10−3mAcm−2(correctedsurface).Whateverthe
surfacetakenintoaccount,thecurrentdensitieswerehigherforthe cable,ascomparedtothestrandsample;thiscouldbeassignedto adissolutionprocessratherthanapassivityprocess.Abreakdown potentialwasthenobservedat−550mV/SCEfollowedbyasharp increaseoftheanodiccurrentdensities.Alltheresultsshoweda lowercorrosionresistanceforthecableascomparedtothestrand sample.Thedifferencesincorrosionbehaviourbetweenthetwo sampleswererelatedtothepenetrationoftheelectrolyteinsidethe cablecavitiesleadingtotheformationofaconfinedandthenmore aggressiveelectrolyte.Thisconfirmed,forthecable,theoccurrence ofbothpittingcorrosiononthesurfaceexposedtothebulk solu-tionandcrevicecorrosiononthesurfaceexposedtotheconfined mediuminside thecavities. Thehigheranodiccurrentdensities observedforthecablewererelatedtotheprogressive modifica-tionoftheelectrolytetrappedinsidethecavitiesandsubsequent dissolutionphenomenon.
Impedance measurements were performed for the strand sampleandthecableafterdifferentexposuretimestothe chloride-containingsulphatesolution.Fig.6showsthediagramsinBode (Fig.6a) and Nyquist (Fig. 6b) coordinates for thestrand sam-ple. After3hand 24h of immersion, the impedance diagrams arecharacterizedbytwotimeconstants.Thefirsttimeconstant (60kHz–1Hz)wasassociatedtotheresponseofthepassivefilm, whilethesecondtimeconstant(1Hz–100mHz)wasattributedto thechargetransferprocessand totheoxygenreductiononthe passivefilminagreementwithliterature[27].Impedance mea-surementsperformedatEcorrinadeaeratedelectrolyte(resultsnot
Fig.6.Electrochemicalimpedancediagramsobtainedforthestrandsample–(a) Bodeand(b)Nyquistcoordinates–afterdifferentexposuretimesatEcorrto0.1M Na2SO4+0.001MNaCl.
at low frequencies and no modification in the high frequency domain.Thisconfirmedtheattributionofthelowfrequencytime constanttotheoxygenreduction.After168hofimmersion,the impedancediagramshowedonlyonetimeconstantwithalower resistancewhichcouldbeassociatedtothecorrosionofthealloy, i.e.tothechargetransferprocess.Suchanevolutionofthediagrams wasinagreementwiththeOMobservations(Fig.2c),i.e.the disso-lutionofthematrixsurroundingtheFe-richparticlesduetooxygen reductionontheintermetallicsand subsequentalcalinizationof theelectrolyte for increasingimmersion times. The impedance diagramsobtainedforthecable(Fig.7)wereclearlymodifiedas compared tothose obtainedfor the strandsample. Theresults arepresented by takinginto account thetotal surface exposed totheelectrolyte(the surfaceexposedtothebulksolutionand thatexposedtotheconfinedmediumtrappedinsidethecavities). TheNyquistdiagram(Fig.7b)wasconstitutedbyastraightlinein thehigh-frequencydomainfollowedbyasemicircle.Thistypical shapecanbeassociatedtotheoxygendiffusioninaporoussystem
[30–33]inagreementwiththepenetrationoftheelectrolyteinside
thecablecavities.Theflattenedsemicirclemayoriginatefromthe cavityshape [34].Theimpedancevalues forthecablewereten timessmallerthanthoseobtainedforthestrandsamplein agree-mentwiththedifferencesincorrosiondamagebetweenthetwo sampleswhichhighlightedtheinfluenceoftheelectrolyte pene-trationthroughthecavitiesofthecable.For168hofimmersion,a decreaseoftheimpedancewasobservedand,inthehigh-frequency domain,atimeconstantappearedwhichcouldbeassociatedtothe corrosionofthealloyintheconfinedmedium[35,36].Additional experiments(results notshown) wereperformedwitha cross-sectionofthecableforwhichthecavitieshadbeensealedwitha resinbyforcingtheresintopenetrateintothecavitieswitha vac-uumsystem.Theimpedancediagramobtainedforthecablewith sealedcavitiesafterimmersionintheaggressivesolutionandthat ofthestrandsampleweresuperimposed.Thiswasinagreement withcommentsintheexperimentalpartandconfirmedthatthe corrosionbehaviourofthecablewascontrolledbythepenetration oftheelectrolyteinsidethecable.
Fig.7.Electrochemicalimpedancediagramsobtainedforthecable–(a)Bode and(b)Nyquistcoordinates–after differentexposuretimesatEcorr to0.1M Na2SO4+0.001MNaCl.Theimpedancevaluestakeintoaccountboththesurface exposedtothebulkandthatexposedtotheconfinedmediuminsidethecavities (S=0.76cm2).
Equivalent electrical circuits are frequently used to extract parametersassociatedwiththeimpedancediagrams.Inthepresent study,thedifferentsetsofimpedancemeasurementsperformed allowedtheinterpretationofthedifferenttimeconstantsin agree-mentwithliteraturedataandrelevantparametersweredefined. Forthestrandsample,relevantparametersweretheoxidefilm resistance(Rox)andthechargetransferresistance(Rt).Theywere
directlymeasuredontheimpedancespectra.Further,among rel-evantparameters,aconstant phaseelement(CPE)isoftenused insteadofa capacitance totakethenon-ideal behaviourofthe interfaceintoaccount.TheCPEisgivenby:
ZCPE=
1
Q (jω)˛ (1)
where ais related to theangle ofrotation of a purely capaci-tivelineonthecomplexplaneplotsandQisin−1cm−2s˛.In
thepresentstudy,aandQweredeterminedusingthegraphical methodproposedbyOrazemetal.[37].Forthecable, consider-ingthecomplexityofaporoussystem,thepolarizationresistance (Rp)wasassumedtoallowthecorrosionresistanceofthecableto
becomparedtothatofthestrandsample.Fig.7ashowedthatthe impedancemoduluswasquitestableforfrequencieslowerthan 10mHz;therefore,theimpedanceatlowfrequency(3mHz)was measuredandusedtoevaluatethecorrosionresistanceofthecable. TheimpedanceparametersarereportedinTable1.
Forthestrandsample,duringthefirst24hofimmersion,aox
remainedconstantandequal to0.95. ThevaluesofQox slightly
increasedfrom7.310−7−1cm−2s0.95to8.610−7−1cm−2s0.95.
Theaox andQox valuesaccountedforthepresenceofapassive
layer.ThevariationofQoxwithincreasingimmersiontimecould
berelatedtotheevolutionofthethicknessand/orofthe chemi-calcompositionofthepassivefilminrelationwiththedissolution ofthematrixaroundtheintermetallics.Duringthefirst24h,the
85
Table1
Parametersobtainedfromtheimpedancediagramsforthestrandsampleandforthecableafterdifferentimmersiontimesin0.1MNa2SO4+0.001MNaClsolution.Forthe cable,theimpedancevaluestakeintoaccountboththesurfaceexposedtothebulkandthatexposedtotheconfinedmediuminsidethecavities(S=0.76cm2).
ImmersionTimes(h) 3 24 168 Strand Rox(kcm2) 201±5 220±5 / aox 0.95±0.01 0.95±0.01 / Qox(−1cm−2)sa 7.310−7±110−7 8.610−7±110−7 / Rt(kcm2) 1010±20 821±10 324±5 a / / 0.94±0.01 Q(−1cm−2)sa / / 4.2510−6±110−6 Cable Rp(kcm2) 106±5 63±5 27±2
Fig.8. PolarizationcurvesoftheAA1370strandsample,theAA1370cableandthe pureCuconnectorafter3hofimmersionatEcorrin0.001MNaClsolution.
corrosionbehaviourofthestrandsamplewascontrolledbythe presenceof theoxidefilmwithhighvaluesof bothRox andRt
while,for168hofimmersion,theelectrochemicalbehaviourwas controlledbythecorrosionprocesses(decreaseoftheRtvalues).
Forthecable,thelowRpvalueswereinagreementwiththelow
corrosionresistanceofthecableascomparedtothestrand sam-ple;moreover,thedecreaseofRpwithincreasingimmersiontime
wasinagreementwiththeextentofthecorrosiondamagerelated tothepenetrationoftheelectrolyteinsidethecableandtothe crevicecorrosionphenomenaonthesurfaceexposedtothe con-finedmediumwhilepittingoccurredonthesurfaceexposedtothe bulk.
3.2. Electrochemicalbehaviouroftheassembly 3.2.1. Preliminaryexperimentsandobservations
Apreliminarystudywasperformedin0.001MNaClto charac-terizeseparatelythecorrosionbehaviourofAl(i.e.AA1370)andCu inthesamesolutionasthatusedfortheLEISmeasurements.First, theanodicbranchofthepolarizationcurvesfor theAlsamples (strandsampleandcable)andthecathodicandanodicbranches forpureCuwereobtained(Fig.8).ComparisonofFigs.5 and8
showedthattheglobalshapeofthecurvesfortheAlsampleswas thesameindependentlyoftheelectrolyte.Forthestrandsample, a600mV-longplateaucorrespondingtoapassivityplateauwith lowanodiccurrentdensities(3.10−4mAcm−2)wasobservedand
followedbyanabruptincreaseofthecurrentassociatedtopitting corrosion.Forthecable,aplateauwasalsoobserved;itwas bet-terdefinedthaninsulphateandchloride-containingsolutionsbut, again,correspondedtohigheranodiccurrentdensitiesthanforthe strandsampleandwasconsideredasapseudo-passivityplateau. Itwasrapidlyfollowedbyasharpincreaseoftheanodiccurrent densitiesassociatedtobothpittingandcrevicecorrosionas
previ-Fig.9. VariationofEcorrduringimmersionin0.001MNaClsolutionfortheAl/Cu assemblieswithandwithoutcavities.
ouslyexplained.Asobservedinthechloride-containingsulphate solution,thebreakdownpotentialassociatedtotheincreaseofthe anodiccurrentdensitieswasmorenegativeforthecable(pitting andcrevicecorrosion)thanforthestrandsample(pitting corro-sion) whichshowedthelowercorrosionresistanceof thecable ascomparedtothestrandsample.Inthe0.001MNaClsolution, thebreakdownpotentialwas10mV/SCEand−500mV/SCEforthe strandsampleandthecable,respectively.Then,itcanbeseenfrom
Fig.8thatthecorrosionpotentialofpureCuwasmorepositive (200mV/SCE)thanthoseofthestrandsample(−700mV/SCE)and thecable(−850mV/SCE)showingasexpectedthat,intheAl/Cu assembly,pureCuwillbethecathodeandtheAlsampleswillbe theanode[20,21,38–40].FromtheanodiccurvesoftheAlsamples andthecathodiccurveofCu,thecommonpotentialmeasured,in thecaseofagalvaniccouplingbetweenthetwometalswiththe samesurfacearea,wouldbeabout−400mV/SCEforanAlstrand sample/Cuassembly,and−500mV/SCEforanAlcable/Cu assem-bly.Itwasworthnoticingthat,fortheassembliesinvestigatedhere andasshowninFig.1,theratioSCu/SAl(SCuandSAlarethesurface
areasexposedtotheelectrolyteintheassemblyrespectivelyfor CuandAl)wassignificantlyhigherthan1.Therefore,thecommon potentialshouldbeshiftedtowardsmorepositivevaluesthanthose previouslyestimated(withthesamesurfacearea).FromFig.8,it canbeclearlyseenthat,in0.001MNaCl,thecommonpotentialwas inthelocalizedcorrosion(pittingandcrevicecorrosion)regimefor thecable,whereas,dependingontheratioSCu/SAl,itcouldbeinthe
passiveregimeforthestrandsample.Thismeantthatasevere cor-rosiondamagewasexpectedforthecablewhencoupledwithCu. Thissuggestedalsothatdifferencesshouldbeobservedbetween thetwotypesofassembly,i.e.withandwithout(allthestrands con-stitutiveofthecablebeingweldedtogether)cavities,asdescribed inFig.1.
Fig.10.OpticalmicroscopeobservationsofthecorrosiondamagefortheAl/Cuassembliesafter(a,b)3hand(c,d)168hofimmersionatEcorrina0.001MNaClsolution: assembly(a,c)withoutand(b,d)withcavities.(e)SEMmicrographoftheAl/Cuinterfaceoftheassemblywithoutcavitiesafter24hofimmersionin0.001MNaClsolution (tiltof70◦).
Fig.9showedEcorr valuesversusimmersiontime in0.001M
NaClforthetwotypesofassembly.Asexpected,Ecorrvalueswere
betweenthoseoftheAlcableandCu.Forbothassemblies,Ecorr
valuesdecreasedwithincreasingimmersiontimewhichcouldbe relatedtocorrosiondamage.Duringthefirst24h,Ecorrvalueswere
thesameforthetwotypesof assembly;theydecreasedslowly from−500mV/SCEto−600mV/SCE.After24hofimmersion,Ecorr
oftheassemblywithoutcavitiesstabilizedaround−600mV/SCE, whereas,fortheassemblywithcavities,Ecorrmovedtowardsmore
negativevaluesandreached−770mV/SCEafter168hof immer-sion.ThisvariationofEcorrsuggestedthat,foranimmersiontime
longerthan24h, thecorrosiondamage wasmoreextended for theassemblywithcavitiesandunderlined,asalreadydiscussed, thatthepresenceofcavitiesinthecablemodifiedthecorrosion processes.
The corrosiondamages for thetwo types of assembly were observedbyOMandSEMaftershortimmersiontimes(3hand24h) andlongimmersiontime(168h)in0.001MNaCl(Fig.10;results areonlygivenfor3hand168h).Independentlyoftheimmersion time,AlpartwascorrodedwhiletheCusurface exposedtothe bulksolutionremainedsafe.Forashortimmersiontime,no signif-icantdifferencewasobservedbetweenthetwotypesofassembly (Fig.10aandb).After168hofimmersion,significantdifferences wereobservedfortheAlpartdependingonthepresenceornot ofcavities(Fig.10candd).Fortheassemblywithoutcavities,OM observationsshowedthepresenceofpitsandnumerousfine fila-mentsaroundthepitsfortheAlpart(Fig.10c).Fortheassembly withcavities,aseverecorrosiondamageoftheAlpartwasobserved withnot onlypitsobservedonthesurfaceexposedtothebulk solutionbutalsosomestrandsconstitutiveofthecablecompletely dissolvedoverseveralhundredsofmicrometersindepth.These
corrosionfeatureswereinagreementwithpittingcorrosion phe-nomenaoccurringonthesurfaceexposedtothebulksolutionwhile crevicecorrosionoccurredinthecavities[38].Moredetailed atten-tionwaspaidtotheAl-Cuinterface.Forbothassemblies,corrosion products(blackareasinFig.10candd)wereclearlyobservedatthe Al/CuinterfaceandspreadallovertheAlpartfortheassemblywith cavities(Fig.10d).Fig.10eshowsaSEMmicrographoftheAl/Cu interfaceafter24hofimmersionfortheassemblywithout cavi-ties.AttheAl/Cuinterface,acrevicewasclearlyvisibleandalsothe Aldissolution.Suchacrevicewasalsoobservedfortheassembly withcavities:itwasevenmorevisible.Similarfeaturewasalready observedforaAl/Cumodelcouple[19].ThisshowedthatforCu, eventhoughnocorrosiondamagewasobservedonthesurface exposedtothebulksolution,corrosionphenomenaoccurredon theverticalwallinsidethecrevice,referringtocrevicecorrosion processes[20,21].Thus,forbothassemblies,theinitialstepofthe corrosionprocesswasthedissolutionoftheAlpartattheAl/Cu interfaceduetothealkalinisationlinkedtotheoxygenreduction reactionontheCupart[19].Theelectrochemicalreactionsthat justifythealuminiumcorrosionwereasfollows[38,40–42]:
Cathodic(ontheCusurfaceexposedtothebulksolution):
O2+2H2O+4e−→4OH− (2)
Anodic:
Al→ Al3++3e− (3)
2Al+6H20 →2Al(OH)3+3H2 (4)
Al(OH)3+OH−→ [Al(OH)4]− (5)
With increasing immersion time, in the crevice, hydrolysis of cationscan beassumed and asa consequencethe chemical
Fig.11. Admittancemapsobtainedafter24hofimmersionatEcorrin0.001MNaCl solutionat10HzabovetheAl/Cuassemblies:(a)withoutcavitiesand(b)with cav-ities.Admittancevaluesarecalculatedtakingintoaccountananalyzedsurfaceof 1mm2.
compositionoftheelectrolyteattheAl/Cuinterfaceshouldvary significantlywithadecreaseofpHasshownbyShietal.[40].This canbeascribedtothereactionofAldissolutionandhydrolysis[40]: Al+nH2O→Al(OH)n3−n+nH++3e−,n=1–3 (6)
Therefore,inthecreviceformedattheAl/Cuinterface,thepH gotmore acidicwhichshouldpromoteacrevicecorrosion phe-nomenonforCu.Ontheopposite,farfromtheAl/Cuinterface,Cu wasincontactwiththebulksolutionwhereoxygenispresent[21]
leadingtoanon-corrodedsurface.FortheAlpart,fortheassembly withoutcavities,pittingcorrosionwasobserved.Concerningthe assemblywithcavities,pittingcorrosionwasobservedontheAl surfaceexposedtothebulksolutionandcrevicecorrosionoccurred inthecavitiesexistingbetweenthestrandsduetothe penetra-tionoftheelectrolyteinsidethecavities.Therefore,thecorrosion productsobservedshouldincludebothcopperoxides,aluminium oxidesandhydroxides[20,38,40,42].Håkanssonetal.showedthat themaincorrosionproductsformedduringgalvaniccorrosionof aluminium/carbonsystemsin0.6MNaClwasAl(OH)3,presented
asagelatinoussubstanceinthecrevicesbetweentheAlwires[43]. TheseauthorsalsoshowedthatAl(OH)3shouldcontroltherateof
thecorrosionprocesseswhichshouldexplaintheplateauobserved onthepolarizationcurvesforthecableafterthebreakdown poten-tial(Fig.8).
3.2.2. LEISstudyoftheassemblies
LEISwasfirstusedinmappingmode.Mapsobtainedat10Hzfor theassemblieswithandwithoutcavities,after24hofexposurein a0.001MNaClsolutionareshowninFig.11.Forbothsamples,the admittancevaluesarehigher(i.e.,lowerresistance)ontheAlpart whichwasduetothegalvaniccouplingbetweenAl(anode)and Cu(cathode).Moreover,heterogeneousvaluesoftheadmittance
wereobservedontheAlpartinaccordancewithpittingcorrosion processes,aspreviouslyshown.ComparisonofFig.11aandbdid notshowsignificantdifferencesbetweenthetwotypesofassembly inagreementwithEcorrmeasurements(Fig.9).
Then, localimpedance spectrawerecollected after24hand 168hofimmersionforthetwotypesofassembly(Fig.12,Nyquist coordinates).
First,itcanbeseenthatimpedancevaluesontheAlpartfor theassemblywithcavitiesarehigherthanthosefortheassembly withoutcavities.Asexplainedintheexperimentalpart,impedance valuesontheAlpartareoverestimatedfortheassemblywith cavi-ties.Theerroronthemeasurementswasestimatedtobeabout20% byconsideringonlythesurfaceexposedtothebulksolution.Ifthe surfaceexposedtotheconfinedelectrolyteinthecavities (consid-eringthesurfaceintheplaneparalleltothecableaxis)istakeninto account,thiserrorshouldbereducedbecausetheexposedsurface wouldbeincreased.Nevertheless,despitetheseinaccurate mea-surements,itwashelpful,forbothAl/Cuassemblies,tostudythe changesobservedforthelocalimpedancediagramsversus immer-siontimeandtocomparetheelectrochemicalresponseoftheAl partandtheCuparttodescribetheinfluenceofthecavitiesonthe corrosionbehaviouroftheAl/Cuassembly.
Then,forbothassemblies,theimpedancevaluesafter24hof immersion (Fig. 12a and b) were higher on the Cu part, then decreasedwhentheprobemovedtowardstheAl/Cuinterface,and decreasedagainwhentheprobewasovertheAlpart.Suchan evo-lutionoftheimpedanceasafunctionofthepositionoftheprobe wasinagreementwiththegalvaniccouplingbetweenAlandCu. After24hofimmersion,pittingcorrosionoccurredontheAlpart (Fig.10).Fortheassemblywithcavities,thediagramsplottedfor theCupart(redcurves inFig.12)aresignificantlymodified in thelowfrequencyrange ascomparedtothoseobtainedforthe assemblywithoutcavities.Theirglobalshapesuggesteda diffusion-controlledelectrochemicalprocess inrelationwiththecathodic reactionofoxygenreductionontheCupart.After168hof immer-sion,thediffusion-controlledprocesswasevenmoremarkedon thediagramsfortheassemblywithcavities(Fig.12d)andcouldbe alsonotedfortheassemblywithoutcavities(Fig.12c).This conclu-sioncouldberelatedtothoseofHåkanssonetal.whoshowedthat, foraAl/carboncouple,theratecontrollingmechanismwassolely themasstransportofoxygenthroughthegelatinousAl(OH)3inside
thecrevice[43].
In order to quantitatively analyse the LEIS diagrams, three parametersweregraphicallyextracted:theCPEvalues(aandQ) andthevaluesoftheimpedancemodulusat1Hz(|Z|1Hz).Fig.13
showsthevariationoftheseparametersforeachdiagramobtained overtheAlandCuparts.Independentlyoftheassemblyandof theimmersiontime,|Z|1HzvaluesfortheCupartwerehigherthan
thosefortheAlpart,whichconfirmedthedifferencesin reactiv-ity betweenthetwo metals whentheywerecoupled(Fig.13a, d).On theCupart,thelow frequency rangewasrelated tothe diffusionprocesslinkedtotheoxygenreduction(Fig.12). What-evertheassembly,theincreaseof|Z|1Hzbetween24hand168hof
immersionwasingoodagreementwiththeshiftofthecorrosion potentialsmeasuredfortheassembliestowardsmoreandmore negativevaluesfrom24hto168hofimmersion(Fig.9):thiswas linkedtoamoreandmoresignificantcathodicpolarizationforthe Cupart.Whentheimmersiontimeincreased,inagreementwith thecorrosionpotentialvalues measuredforthetwoassemblies, thedifferencesin|Z|1HzvaluesbetweentheAlandCupartswere
evenmoremarkedfortheassemblywithcavities.After168hof immersion,|Z|1HzvaluesmeasuredontheAlpartfortheassembly
withcavitieswerelowerthanthoseoftheAlpartforthe assem-blywithoutcavities.Thiswasexplainedbytakingintoaccountthe increaseoftheelectrochemicalprocessesina confinedmedium previouslyshowedforthecable:theeffectofthegalvaniccoupling
Fig.12.Localimpedancespectraobtainedafter(a,b)24hand(c,d)168hofimmersionatEcorrin0.001MNaClsolutionfordifferentpositionsofthebi-electrodeabovethe Al/Cuassemblies:(a,c)withoutcavitiesand(b,d)withcavities.Impedancevaluesarecalculatedtakingintoaccountananalyzedsurfaceof1mm2.
Fig.13. Variationoftheparameters((a)and(d)|Z|1Hz,(b)and(e)aand(c)and(f)Q)obtainedfromthelocalimpedancespectraafterdifferentexposuretimesin0.001M NaClandfordifferentpositionsofthebi-electrodeovertheAl/Cuassemblies:(d)withoutcavitiesand( )withcavities.Impedancevaluesarecalculatedtakingintoaccount ananalyzedsurfaceof1mm2.
wasmoreandmoremarkedwithincreasingimmersiontimefor theassemblywithcavities.
ThedifferencesinreactivitybetweenCuandAlfromonepart, andbetweenthetwoassembliesfortheotherpart,wereconfirmed bythevariationofaandQwithincreasingimmersiontime.After 24hofimmersion,thestrongreactivityattheAl/Cuinterfacewas
showedwithboththedecreaseofaandtheincreaseofQinthezone whereSEMmicrographies(Fig.10e)showedacreviceontheAlpart. Thisevolutionwasmoremarkedforanassemblywithcavitiesin relationwithalowercorrosionresistanceoftheAlpartduetothe penetrationoftheelectrolyteinsidethecavitiesofthecable.After 168hofimmersion,aandQvaluesconfirmedtheincreaseofthe
89
galvaniccouplingeffect.Between24hand168hofimmersion,a valuessignificantlydecreasedinagreementwithanincreaseofthe diffusionprocessesontheCupartandastrongercorrosionforthe Alpart.Moreover,itcanbeseenthatafter168hofimmersion, avaluesarelowerforboththeCuandAlpartsfortheassembly withcavitiesascomparedtotheassemblywithoutcavities.The resultcouldbelinkedtoastrongerincreaseofQvaluesattheAl/Cu interfaceobservedfortheassemblywithcavitiesascomparedto theassemblywithoutcavities.Theresultswereingoodagreement withthepresenceoftheconfinedelectrolytewhichamplifiedthe effectofthegalvaniccouplingwhentheimmersiontimeincreased. Therefore,theLEISmeasurementsprovidedevidencethatthe corrosionbehaviouroftheAl/Cuassemblywasexplainedbyboth thegalvaniccouplingbetweentheAlandCuparts,whichenhanced thecorrosionoftheAlpart,andthepenetrationoftheelectrolyte insidethecavitiesobservedintheAlpartoftheassembly,which ledtoamoreandmoreconfinedandaggressiveelectrolytewith increasingimmersiontimesothatitenhancedthereactivityofthe AlpartandthusthegalvaniccouplingwithCu.
4. Conclusions
ThestudywasfirstfocussedonthecorrosionbehaviourofanAl (AA1370)cableascomparedtoastrand.
1.OMobservationsafterexposuretoachloride-containing sul-phatesolutionshowedamoreextendedcorrosiondamagefor thecablethanforthestrand.
2.Electrochemical measurements showed for the cable: (i) a strongerdecreaseofthecorrosionpotentialwithtimethanfor thestrand,(ii)anincreaseoftheanodiccurrentdensity associ-atedwithapseudo-passivityplateauandamorenegativepitting potentialascomparedtothestrandand(iii)impedancediagrams characteristicofaporoussystem.
3.ThecorrosionbehaviouroftheAlcablewascontrolledbythe penetrationoftheelectrolytebetweenthestrandsconstitutive ofthecable.Suchaphenomenonledtoamoreaggressive con-finedmediumwhichexplainedthelowercorrosionresistance ofthecableascomparedtothestrand.
4.Then,thecorrosionbehaviouroftheassemblybetweenanAl cableandaCuconnectorwasinvestigatedbyelectrochemical techniques,andinparticularlocalimpedancemeasurements. 5.Aldissolution occurreddue togalvaniccouplingwiththeCu
connectorandcrevicecorrosionwasobservedontheCuwalls perpendicularlytotheAl/Cuinterfaceexposedtotheelectrolyte. 6.LocalimpedancedatashowedthatthecavitiesoftheAlcable significantlyinfluencedthecorrosionbehaviourofthe assem-bly:thereactivityattheAl/Cuinterfaceandtheextentofthe corrosiondamageontheAlpartwerestrongerfortheassembly withcavities.
References
[1]Y.Yamano,T.Hosokawa,Developmentofaluminumwiringharness,SEITech. Rev.73(2011)73–80.
[2]A.Laurino,E.Andrieu,J.P.Harouard,J.Lacaze,M.C.Lafont,G.Odemer,C. Blanc,Corrosionbehaviorof6101aluminumalloystrandforautomotive wires,J.Electrochem.Soc.160(2013)5069–5575.
[3]K.Yoshida,K.Doi,Improvementofductilityofaluminumwireforautomotive wiringharnessbyalternatedrawing,ProcediaEng.81(2014)706–711.
[4]S.Koch,H.Antrekowitsch,AluminumAlloysforwireharnessesinautomotive engineering,BergHuettenmaennMonatsh152(2007)62–67.
[5]S.-I.Matsuaka,H.Imai,Directweldingofdifferentmetalsusedultrasonic vibration,J.Mater.Process.Technol.209(2009)954–960.
[6]J.W.Yang,B.Cao,X.C.He,H.S.Luo,Microstructureevolutionandmechanical propertiesofCu–Aljointsbyultrasonicwelding,Sci.Technol.Weld.Join.19 (2014)500–504.
[7]R.Rambat,A.J.Davenport,G.M.Scamans,A.Afseth,Effectofiron-containing intermetallicparticlesonthecorrosionbehaviourofaluminum,Corros.Sci. 48(2006)3455–3471.
[8]J.O.Park,C.H.Paik,R.C.Alkire,ScanningMicrosensorsformeasurementof localpHdistributionsatthemicroscale,J.Electrochem.Soc.143(1996) 174–176.
[9]O.Seri,M.Imaizumi,ThedissolutionofFeAl3Intermetalliccompoundand
depositiononaluminuminAlCl3solution,Corros.Sci.30(1990)1121–1133.
[10]J.Xu,W.Chen,Behaviorofwireinparallelwirestayedcableundergeneral corrosioneffects,J.Constr.SteelRes.85(2013)40–47.
[11]Y.Ishikawa,S.Kawakami,Effectsofsaltcorrosionontheadhesionofbrass platedsteelcordtorubber,RubberChem.Technol.59(1986)1–15.
[12]J.J.Perdomo,I.Song,Chemicalandelectrochemicalconditionsonsteelunder disbondedcoatings:theeffectofappliedpotential,solutionresistivity, crevicethicknessandholidaysize,Corros.Sci.42(2000)1389–1415.
[13]S.H.Zhang,S.B.Lyon,Anodicprocessesonironcoveredbythin:dilute electrolytelayers(I)-anodicpolarisation,Corros.Sci.36(1994)1289–1307.
[14]R.Oltra,B.Malki,F.Rechou,Influenceofaerationonthelocalizedtrenching onaluminumalloys,Electrochim.Acta55(2010)4536–4542.
[15]S.Chanel,N.Pébère,Aninvestigationonthecorrosionofbrass-coatedsteel cordsfortyresbyelectrochemicaltechniques,Corros.Sci.43(2001)413–427.
[16]D.Wong,L.Swette,Aluminumcorrosioninuninhibitedethyleneglycol-water solutions,J.Electrochem.Soc.126(1979)11–15.
[17]M.G.A.Khedr,A.M.S.Lashien,Theroleofmetalcationsinthecorrosionand corrosioninhibitionofaluminuminaqueoussolutions,Corros.Sci.33(1992) 137–151.
[18]M.G.A.Khedr,A.M.S.Lashien,Corrosionbehaviorofaluminuminthepresence ofacceleratingmetalcationsandinhibition,J.Electrochem.Soc.136(1989) 968–972.
[19]J.-B.Jorcin,C.Blanc,N.Pébère,B.tribollet,V.Vivier,Galvaniccoupling betweenpurecopperandpurealuminum,J.Electrochem.Soc.155(2008) C46–C51.
[20]C.Blanc,N.Pébère,B.Tribollet,V.Vivier,Galvaniccouplingbetweencopper andaluminuminathin-layercell,Corros.Sci.52(2010)991–995.
[21]S.Joma,M.Sancy,E.Sutter,T.T.M.Tran,B.Tribollet,Incongruentdissolution ofcopperinAl-Cualloys:influenceoflocalpHchanges,Surf.InterfaceAnal. 45(2013)1590–1596.
[22]P.DeLima-Neto,J.P.Farias,L.F.G.Herculano,H.C.DeMiranda,Determination ofthesensitizedzoneextensioninweldedAISI304stainlesssteelusing non-destructiveelectrochemicaltechniques,Corros.Sci.50(2008) 1149–1155.
[23]G.Baril,C.Blanc,M.Keddam,N.Pébère,Localelectrochemicalimpedance spectroscopyappliedtothecorrosionbehaviorofanAZ91magnesiumalloy, J.Electrochem.Soc.150(2003)488–493.
[24]S.Marcelin,N.Pébère,Synergisticeffectbetween8-hydroxyquinolineand benzotriazoleforthecorrosionprotectionof2024aluminiumalloy:alocal electrochemicalimpedanceapproach,Corros.Sci.101(2015)66–74.
[25]M.Mouanga,M.Puiggali,B.tribollet,V.Vivier,N.Pébère,O.Devos,Galvanic corrosionbetweenzincandcarbonsteelinvestigatedbylocalelectrochemical impedancespectroscopy,Electrochim.Acta88(2013)6–14.
[26]D.Sidane,E.Bousquet,O.Devos,M.Puiggali,M.Touzet,V.Vivier,A. Poulon-Quintin,Localelectrochemicalstudyoffrictionstirweldedaluminum alloyassembly,J.Electroanal.Chem.737(2015)206–211.
[27]J.-B.Jorcin,M.E.Orazem,N.Pébère,B.Tribollet,CPEanalysisbylocal electrochemicalimpedancespectroscopy,Electrochim.Acta51(2006) 1473–1479.
[28]J.-B.Jorcin,E.Aragon,C.Merlatti,N.Pébère,Delaminatedareasbeneath organiccoating:alocalelectrochemicalimpedanceapproach,Corros.Sci.48 (2006)1779–1790.
[29]N.A.Belov,D.G.Eskin,A.A.Aksenov,MulticomponentPhaseDiagrams: ApplicationsforCommercialAluminiumAlloys,firstedition,ElsevierLtd, Oxford,2005.
[30]D.D.Macdonald,Reflectionsonthehistoryofelectrochemicalimpedance spectroscopy,Electrochim.Acta51(2006)1376–1388.
[31]C.Gabrielli,M.Keddam,N.Portail,P.Rousseau,H.Takenouti,V.Vivier, Electrochemicalimpedancespectroscopyinvestigationsofamicroelectrode behaviorinathin-layercell:experimentalandtheoreticalstudies,J.Phys. Chem.110(2006)20478–20485.
[32]H.-K.Song,H.-Y.Hwang,K.-H.Lee,L.H.Dao,Theeffectofporesize distributiononthefrequencydispersionofporouselectrodes,Electrochim. Acta45(2000)2241–2257.
[33]A.Lasia,Impedanceofporouselectrodes,J.Electroanal.Chem.397(1995) 27–33.
[34]C.Hitz,A.Lasia,Experimentalstudyandmodellingofimpedanceoftheheron porousNielectrodes,J.Electroanal.Chem.51(2001)213–222.
[35]S.Marcelin,N.Pébère,S.Régnier,Electrochemicalinvestigationsoncrevice corrosionofamartensiticstainlesssteelinathinlayercell,J.Electroanal. Chem.737(2015)198–205.
[36]M.Keddam,A.Hugot-Le-Goff,H.Takenouti,D.Thierry,M.C.Arevalo,The influenceofathinelectrolytelayeronthecorrosionprocessofzincin chloride-containingsolutions,Corros.Sci.33(1992)1243–1252.
[37]M.E.Orazem,N.Pébère,B.Tribollet,Enhancedgraphicalrepresentationof electrochemicalimpedancedata,J.Electrochem.Soc.153(2006)B129–B136.
[38]R.Vera,P.Verdugo,M.Orellana,E.Mu ˜noz,Corrosionofaluminiumin copper–aluminiumcouplesunderamarineenvironment:influenceof polyanilinedepositedontocopper,Corros.Sci.52(2010)3803–3810.
R.Gravinaetal./CorrosionScience119(2017)79–90
[39]L.B.Coelho,M.Mouanga,M.-E.Druart,I.Recloux,D.Cossement,M.-G.Olivier, ASVETstudyoftheinhibitiveeffectsofbenzotriazoleandceriumchloride solelyandcombinedonanaluminium/coppergalvaniccouplingmodel, Corros.Sci.110(2016)143–156.
[40]H.Shi,E.-H.Han,F.Liu,T.Wei,Z.Zhu,D.Xu,Studyofcorrosioninhibitionof coupledAl2Cu–AlandAl3Fe–Albyceriumcinnamateusingscanningvibrating
electrodetechniqueandscanningion-selectiveelectrodetechnique,Corros. Sci.98(2015)150–162.
[41]N.Murer,R.Oltra,B.Vuillemin,O.Néel,Numericalmodellingofthegalvanic couplinginaluminiumalloys:adiscussionontheapplicationoflocalprobe techniques,Corros.Sci.52(2010)130–139.
[42]J.Bernard,M.Chatenet,F.Dalard,Understandingaluminumbehaviorin aqueousalkalinesolutionusingcoupledtechniques:partI.Rotatingring-disk study,Electrochim.Acta52(2006),86–93.
[43]E.Håkansson,J.Hoffman,P.Predecki,M.Kumosa,Theroleofcorrosion productdepositioningalvaniccorrosionofaluminum/carbonsystems, Corros.Sci.114(2017)10–16.