Controlled Rejuvenation of Amorphous
Metals with Thermal Processing
The MIT Faculty has made this article openly available.
Please share
how this access benefits you. Your story matters.
Citation
Wakeda, Masato, Junji Saida, Ju Li, and Shigenobu Ogata.
“Controlled Rejuvenation of Amorphous Metals with Thermal
Processing.” Scientific Reports 5 (May 26, 2015): 10545.
As Published
http://dx.doi.org/10.1038/srep10545
Publisher
Nature Publishing Group
Version
Final published version
Citable link
http://hdl.handle.net/1721.1/98444
Terms of Use
Creative Commons Attribution
Amorphous Metals with Thermal
Processing
Masato Wakeda1, Junji Saida2, Ju Li3 & Shigenobu Ogata1,4
Rejuvenation is the configurational excitation of amorphous materials and is one of the more promising approaches for improving the deformability of amorphous metals that usually exhibit macroscopic brittle fracture modes. Here, we propose a method to control the level of rejuvenation through systematic thermal processing and clarify the crucial feasibility conditions by means of molecular dynamics simulations of annealing and quenching. We also experimentally demonstrate rejuvenation level control in Zr55Al10Ni5Cu30 bulk metallic glass. Our local heat-treatment recipe (rising
temperature above 1.1Tg, followed by a temperature quench rate exceeding the previous) opens
avenue to modifying the glass properties after it has been cast and processed into near component shape, where a higher local cooling rate may be afforded by for example transient laser heating, adding spatial control and great flexibility to the processing.
Amorphous metals1–3 have excellent properties such as high corrosion resistance4, high strength5, and
large elastic elongation6. At room temperature, however, plastic deformation localizes into narrow band
region, and sudden fracture occurs exhibiting almost no macroscopic plastic deformation under uniaxial tensile and compressive loadings7–9. This brittle nature is the most serious flaw of amorphous metals
for use as structural materials. Therefore, improvement, and moreover tuning of the deformability has been the largest challenge of the decades in the field of amorphous metals10–13. Rejuvenation14–17 is the
structural excitation of amorphous solids accompanied by an increase in the enthalpy and free volume and is thus the inverse of aging. Since rejuvenation has to change the elastic and plastic deformation behavior17 of amorphous metals, it is a new but promising approach for tuning the deformability of
amorphous metals. It has recently been reported that shot-peening16 and sever plastic deformation17
can mechanically induce the rejuvenation. On the other hand, structural excitation driven by a thermal loading process18–19 also has potential application for practical usage, because the thermal loading
pro-cess realizes a local rejuvenation control, which is more difficult by mechanical propro-cessing. However, the feasibility rejuvenation driven by thermal annealing and quenching is still controversial because of few experimental evidence18 and a lack of clear knowledge of the nonequilibrium glass properties.
In this work, we theoretically propose a recipe to control the level of rejuvenation through systematic thermal processing and clarify the crucial feasibility conditions by means of molecular dynamics (MD) simulations and figure out a rejuvenation map with respect to annealing temperature and quenching rate. Our rejuvenation map provides temperature-cooling rate conditions to achieve desired level of rejuvenation while avoiding crystallization and/or gross shape change. Based on the rejuvenation map we also experimentally demonstrate the realization of rejuvenation level control in Zr55Al10Ni5Cu30 bulk
1Graduate School of Engineering Science, Osaka University, 1-3 Machikaneyama, Toyonaka, Osaka, 560-8531,
Japan. 2Frontier Research Institute for Interdisciplinary Sciences, Tohoku University, Aramaki aza Aoba 6-3,
Aoba-ku, Sendai, Miyagi, 980-8578, Japan. 3Department of Nuclear Science and Engineering and Department of
Materials Science and Engineering, Massachusetts Institute of Technology, 77 Massachusetts Avenue, Cambridge, Massachusetts, 02139, USA. 4Center for Elements Strategy Initiative for Structural Materials (ESISM), Yoshida
Honmachi, Sakyo-ku, Kyoto University, Kyoto, 606-8501, Japan. Correspondence and requests for materials Received: 30 November 2014
Accepted: 17 April 2015 Published: 26 May 2015
www.nature.com/scientificreports/
metallic glass, which evidences our rejuvenation map and recipe do work well even for typical experi-mental cooling rates. Moreover, the internal structural features and mechanical properties of rejuvenated amorphous metals are revealed.
Simulation Procedures
For all of the MD simulations described here, we use the Lennard-Jones (L-J) potentials20 developed for
the Cu-Zr amorphous alloy and a 30,000-atom simulation box with three-dimensional (3D) periodic boundary conditions. We consider alloys with atomic compositions of Cu50Zr50, Cu30Zr70, and Cu57Zr43
to ensure composition-independent generality (see Supplementary Information in regard to the latter two). The MD time step was set to 1 fs, and we employed the NPT ensemble for which the temperature and pressure are controlled by the Nose-Hoover21 and Parrinello-Rahman methods22, respectively. An
amorphous model was constructed via a melt-quenching process as follows (Fig. 1). We first arranged the atoms randomly in the simulation box and melted the structure by keeping the model at a high temperature of 3000 K for 20 ps (Fig. 1; process A→B). The melted model was then quenched at a con-stant cooling rate of Rq(= ×1 1012 K/s) from 3000 K to 0 K (process B→D). This quenched product is
referred to as the “as-quenched model” hereafter, and we use a tilde to represent the initial quenching process. By monitorinng the specific volume change during the rapid quenching process with a cooling rate of Rq, the glass transition temperature Tg was determined to be approximately 900 K from the kink
in volume-temperature curve. Since Tg generally depends on the cooling rate23, we also determined a set
of temperatures Ti
g for five different cooling rates Riq ( = −i 1 5): R1q= ×1 1011 K/s, Rq2= ×3 1011
K/s, Rq3 = ×1 1012 K/s, R4q= ×3 1012 K/s, and Rq5= ×1 1013 K/s (see Supplementary Information,
Fig. S1).
We conducted successive thermal loading simulations of the as-quenched model as follows. First, the as-quenched model was reheated at a heating rate of ×1 1013 K/s up to a temperature T
a (process
D→E) and then annealed for a time period ta (= 2000 ps) under constant Ta and zero external pressure
conditions (process E→F). Isothermal annealing processes with ta= 500 and 1000 ps were also conducted
and revealed that the annealing time ta had a small effect on our simulation results (see Supplemental
Information, Fig. S2). We assumed various Ta values ranging from . T0 44 g to . T1 67 g to examine the effect
of the annealing temperature on the level of thermal rejuvenation. After isothermal annealing at Ta for
ta, the model was quenched again at five different cooling rates, Rqi (=Riq) ( = −i 1 5), from Ta to 0 K
(process F→G). This final product is referred to as the “annealed model” hereafter.
Results and Discussion
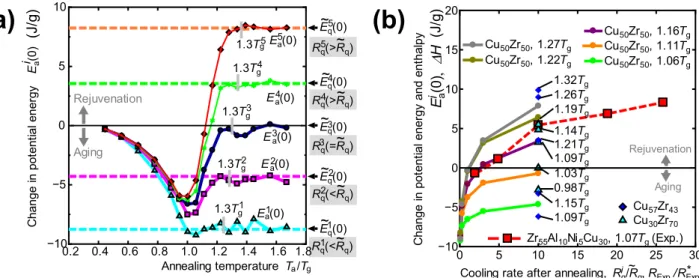
Rejuvenation map. Figure 2(a) shows the generated excess potential energy ( )E 0i
a due to the thermal
loading process from state D to G for different Ta and Rqi values. If any aging occurs during the thermal
loading process, then the excess potential energy value should fall below zero. On the other hand, if thermal rejuvenation occurs, then the excess potential energy value should remain positive. Thus, the excess potential energy can represent the level of rejuvenation or aging depending on the sign. As we see in Fig. 2(a), significant aging is observed whenever the cooling rate is less than Rq, regardless of the Figure 1. Schematic illustration of the initial melt-quenching and subsequent thermal loading process. The
system was initially quenched at a constant cooling rate of Rq (B→D). The instantaneous potential energy at
temperature T in the initial-quenching process is represented as ( )E Tq . The as-quenched model was then
subjected to thermal loading consisting of heating (D→E), isothermal annealing (E→F), and quenching (F→G) at various annealing temperatures Ta and constant cooling rates Rqi. The instantaneous potential
energy at temperature T in the second quenching process (F→G) is represented as ( )E Ti
annealing temperature Ta, whereas thermal rejuvenation is observed in the opposite case. Thus, a higher
cooling rate after isothermal annealing than that of the initial quenching process is a necessary condition for thermal rejuvenation, but this alone is insufficient. Thermal rejuvenation is only realized when the annealing temperature is above a certain critical temperature ≈ .Tc 1 1Tg. Thus, the two conditions,
>
Rqi Rq and >Ta Tc, are crucial conditions for feasible thermal rejuvenation. We also found that a
higher Ri
q and a higher Ta result in a higher level of rejuvenation under these conditions. Figure 2(a) can
then be used as a rejuvenation map to identify conditions for controlling the rejuvenation level. It should be noted that Tc is not an intrinsic material property but depends on both Rq and Rqi as follows: 1) Since
a higher Rq leads to a thermodynamically unstable as-quenched state, further aging proceeds easily
dur-ing later thermal loaddur-ing, and the Tc value is shifted to a higher temperature. 2) Since a higher Rqi does
not allow a more thermodynamically stable state to be found during the final quenching process, limited aging proceeds, and the Tc value is shifted to a lower temperature (Fig. 2(a)).
The levels of aging and rejuvenation are relative quantities and depend on the choice of reference amorphous state. Thus, if we choose a perfectly rejuvenated amorphous state as the reference amorphous state, which is supposedly constructed through melt quenching at an infinitely fast cooling rate, then fur-ther rejuvenation will not occur. In reality, however, it is impossible to realize such a perfectly rejuvenated state; even though a near-perfectly rejuvenated state can be realized, we would not be able to observe it because of the extremely short lifetime of the highly excited state. For this reason, the discussions above are valid for actual glass systems in general, in which both aging and rejuvenation can be realized.
It is worth noting that evaluating the level of aging and rejuvenation based on the volume change (i.e., mass density change) yields qualitatively similar results to those in Fig. 2(a) (see Supplementary Information, Fig. S3).
Based on the above findings, we demonstrate thermal rejuvenation experimentally in a Zr55Al10Ni5Cu30
bulk glassy alloy ( =Tg 684 K at a heating rate of 0.33 K/s). The as-quenched Zr55Al10Ni5Cu30 alloy rod
was sliced into 0.5-mm-thick disc samples that were initially annealed at 685 K for 120 s at heating and cooling rates of 0.17 K/s, denoted as R∗
Exp. The fully relaxed samples were then annealed again at 735 K
(= . T1 07 g) for 120 s at a heating rate of 0.33 K/s and then quenched at cooling rates of RExp. = 0.33, 0.83,
1.7, 3.2, or 4.4 K/s (Ref. 24). The excess enthalpy generated during the second annealing process ∆H is calculated as ∆ =H
∫
RT723KCp s, −C dTp q, , where RT is the room temperature. Cp s, and Cp q, are thespe-cific heats of the sample reheated to 723 K and the as-secondary annealed state, respectively. The spespe-cific heat measurement was conducted with a heating and cooling rates of 0.33 K/s using DSC. Figure 2(b) shows the change in the potential energy calculated by the MD simulation compared to the change in the enthalpy obtained from the experiment. The cooling rates after annealing employed in the MD sim-ulation and experiment are normalized by the typical cooling rates, Rq (= ×1 1012 K/s) and R∗Exp.
Figure 2. Rejuvenation map. (a) Change in the potential energy caused by the thermal loading process
(D→G), where ( )E 0i
a represents the potential energy of the annealed model with a cooling rate of Rqi. The
potential energy of the as-quenched model with a cooling rate Riq is shown by the dashed lines. The grey
rectangles represent . T1 3 gi, where Tgi is the estimated glass transition temperature in the melt-quenching
process with a cooling rate Riq(=Rqi). (b) Change in the potential energy calculated by MD and the change in
the enthalpy obtained from the experiment. For the cooling rate after annealing, Ri
q is normalized by Rq in
www.nature.com/scientificreports/
curves in Fig. 2(b) are not directly comparable; yet the basic trend and the order of magnitude roughly match. The enthalpy obtained from the experiment increases with increasing cooling rate after annealing, and the positive enthalpy of the plots for RExp.= 0.83, 1.7, 3.2, and 4.4 K/s indicate that thermal
rejuve-nation was achieved. This is clear evidence of the feasibility of thermal rejuverejuve-nation via thermal loading, provided the necessary conditions are satisfied. The rough matching between MD and experimental curves in Fig. 2(b) implies that the level of rejuvenation is dominated by a cooling rate ratio between initial melt-quenching and final quenching, rather than the absolute magnitude of the cooling rate.
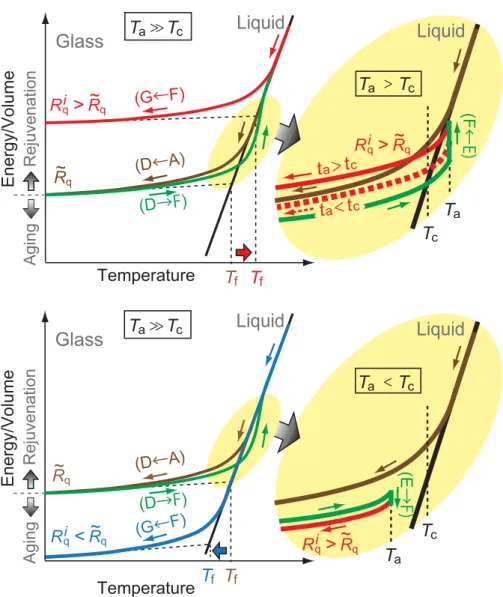
Figure 3 shows a schematic of the energy/volume change during the initial melt-quenching (A→D) and subsequent thermal loading processes (D→G). (1) If the annealing temperature Ta is much higher than
Tc ( Ta Tc), then an amorphous metal may quickly “relax” to an equilibrium liquid state within the
heating stage (green curve in Fig. 3, top and bottom left) before the annealing process starts. As the equi-librium liquid is cooled (F→G), sooner or later the liquid deviates from the equiequi-librium liquid state and eventually the structure is frozen into a glass. A cooling rate Ri
q higher than Rq induces an earlier deviation
from the equilibrium liquid and leads to a rejuvenated glass with a higher energy/volume (red curve in Fig. 3, top left), while a cooling rate lower than Rq leads to an aged glass (blue curve in Fig. 3, bottom left). Figure 3. Schematic illustration of the energy/volume change during the initial melt-quenching (A→D) and
subsequent thermal loading processes (D→G) under conditions of Ta Tc and > Rqi Rq (top left); Ta Tc
and Ri< R
q q (bottom left); >Ta Tc and > Rqi Rq (top right); and <Ta Tc and > Rqi Rq (bottom right). The
equilibrium liquid is represented as a solid black line. In the top right figure, the solid red curve represents the energy/volume change under >ta tc conditions, while the dotted red curve represents that under <ta tc conditions. In the top and bottom left figures, the vertical dotted lines represent the fictive temperature Tf, at which the extrapolated glass line intersects the equilibrium liquid line. The aging decreases and rejuvenation increases Tf by changing the energy/volume versus temperature curve of the glass.
(2) If Ta is slightly above Tc (Ta>Tc), then the annealing time ta becomes a key factor in addition to the
cooling rate Ri
q because the relaxation time to the equilibrium liquid is considerable compared with the
heating and annealing times. Thus, during isothermal annealing the energy/volume increases to approach that of the equilibrium liquid. We actually observed the energy increase during the isothermal annealing process under >Ta Tc conditions (see Supplementary Information, Fig. S4). As shown in Fig. 3 (top
right), a longer annealing time leads to a larger energy/volume increase during isothermal annealing and eventually to a rejuvenated glass after cooling (solid red curve), while a shorter annealing time may lead to an aged glass (dotted red curve). Therefore, there is a critical minimum annealing time for rejuvenation tc; rejuvenation is realized only if >ta tc. (3) If Ta is below Tc ( <Ta Tc), then the annealing-cooling
process always leads to aged glass as shown in Fig. 3 (bottom right). In Fig. 3 (top and bottom left), the fictive temperature Tf (Ref. 25) is also indicated, at which the extrapolated glass line intersects the
equi-librium liquid line. In a similar manner to Tg, glass produced at a higher cooling rate from a high
tem-perature liquid has a higher fictive temtem-perature and vice versa26.
Change in the topological order by rejuvenation. We investigated the change in the topological order induced by the thermal loading process. The internal structures of amorphous metals, such as the short-range order (SRO) and medium-range order (MRO) have been attracted much attention, because they are correlated with the intrinsic properties of amorphous metals. Figure 4(a) shows the change in the fraction of icosahedral SRO, which has an energetically stable, high packing structure and is known to be a core amorphous metal structure. The figure shows clearly that aging increases and rejuvenation decreases icosahedral SRO.
Figure 4(b) shows the topological MRO composed of icosahedra. To characterize the MRO network structure, we employ the MRO clustering coefficient parameter X, which was employed in our previous work27. For the center atom of the icosahedron α, the clustering coefficient parameter X can be defined α
as, Xα =2Lα/ n nα( α− )
{ B B 1 }, where nBα is the icosahedral bond number, and L is the number of trian-α
gular geometries. As X is the average clustering coefficient parameter over all icosahedra in an amor-phous model, a large X value implies a highly developed MRO network structure. As shown in Fig. 4(b), X increases during the thermal loading process (D→G) when the annealing temperature Ta is below
. T
1 1 g or the cooling rate is below Rq. In contrast, although some fluctuation is observed, X decreases
when Ta is above . T1 1 g and the cooling rate is above Rq.
Together with Fig. 2(a) and Fig. 4, we conclude that aging increases and rejuvenation decreases the topological SRO and MRO, which are typical characteristics of glass structures. The clear correspondence between the potential energy profile in Fig. 2(a) (and the volume profile in Supplementary Information,
0.2 0.4 0.6 0.8 1.0 1.2 1.4 1.6 1.8 −0.030 Annealing temperature Ta/Tg Ch ange Rq 0.2 0.4 0.6 0.8 1.0 1.2 1.4 1.6 1.8 −0.080 Annealing temperature Ta/Tg Rq Rq5 ,1.3Tg Figure 4. The change in the topological order induced by the thermal loading process. (a) Change in the
fraction of the icosahedral atomic cluster through thermal loading with different final cooling rates Ri
q. The
as-quenched model has an icosahedral cluster fraction of 0.081. (b) Change in the average cluster coefficient of the icosahedral MRO X during the thermal loading process (D→G) with different final cooling rates Ri
q.
The X value of the as-quenched model is 0.327. The four figures to the right of the graph represent the MRO network structure27 of the annealed models. The final cooling rate and annealing temperature are
www.nature.com/scientificreports/
Fig. S3) and the SRO and MRO in Fig. 4 indicates that aging and rejuvenation change the macroscopic structural properties of amorphous metals, such as the free volume, by changing the internal topological order.
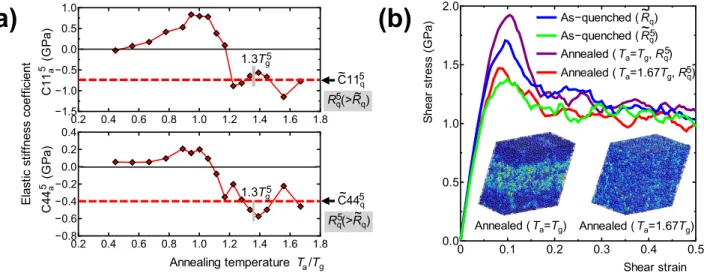
Change in the mechanical properties by rejuvenation. To demonstrate the possibility of con-trolling the mechanical properties via aging and rejuvenation, the change in elastic stiffness due to ther-mal loading (process D→G) was investigated. The elastic stiffness coefficients of the as-quenched model with a cooling rate Rq (state D in Fig. 1), C11q and C44q, were calculated from the second spatial
deriv-ative of the total potential energy ( )E 0q ; these C11q and C44q values are taken as the reference. The
elastic stiffness coefficients of the models annealed at Ta and with a final cooling rate Rq5 (state G) were
also calculated, and the change in the elastic stiffness coefficients due to thermal loading (process D→ G), C11a5 and C44a5, are shown Fig. 5(a). Both C11a5 and C44a5 increase in the case of aging, while they
decrease in the case of rejuvenation. Moreover, both values saturate in the high Ta region and take values
of C11q5 and C44q5, respectively, which are the elastic stiffness coefficients of the as-quenched model with
a cooling rate of Rq5 (=Rq5). The clear correspondence between the potential energy profile ( =i 5) in
Fig. 2 and the elastic stiffness coefficient profile in Fig. 5(a) shows the possibility of controlling the elas-tic properties of amorphous metals via thermally induced rejuvenation and aging. The correlation between SRO in Fig. 4(a) and elastic stiffness in Fig. 5(a) in this study agrees with that found in our previous studies27, 28, where the elastic constant increases with increasing icosahedral SRO.
Plastic deformation behavior of the “aged model” and “rejuvenated model” was also investigated by simple shear MD deformation tests, in which the models were applied incremental affine shear strain at 0 K. At each strain step, the atomic structure and simulation box were relaxed using the conjugate gradi-ent method. Figure 5(b) shows the shear stress change during the shear deformation tests for four mod-els; two as-quenched models (state D) with different cooling rates, Rq and Rq5(>Rq), and two annealed
models (state G) with different annealing temperatures, Tg (aged model) and . T1 67 g (rejuvenated model)
with the same cooling rate of after annealing, Rq5(=Rq5). All the model shows a maximum shear stress at
around γ = .0 1 engineering shear strain, and the peak shear stress increases by the thermal loading process D→G with the annealing at =Ta Tg and decreases with the annealing at Ta = .1 67Tg. The two
figures of atomic structure in Fig. 5(b) represent snapshots of the annealed models at γ = .0 4 engineer-ing shear strain, in which atoms are colored by Mises strain29. The aged model shows an inhomogeneous
deformation, in which Mises strain is localized into band region parallel to shear direction, while the rejuvenated model shows more homogeneous deformation. The clear difference in deformation mode 0 0.1 0.2 0.3 0.4 0.5 0.0 0.5 1.0 1.5 2.0 Shear strain Shear st re ss (G Pa ) As−quenched ( Rq) As−quenched ( Rq) Annealed ( Ta=Tg, Rq) Annealed ( Ta=1.67Tg, Rq) Annealed ( Ta=Tg) Annealed ( Ta=1.67Tg)
(b)
~ ~5 5 5 0.2 0.4 0.6 0.8 1.0 1.2 1.4 1.6 1.8 −1.5 −1.0 −0.5 0.0 0.5 1.0 0.2 0.4 0.6 0.8 1.0 1.2 1.4 1.6 1.8 −0.8 −0.6 −0.4 −0.2 0.0 0.2 0.4 Annealing temperature Ta/Tg C4 4a (G Pa ) Elas tic st iff nes s coef fici en t C1 1a (G Pa ) C115q Rq5(>Rq) C445q Rq5(>Rq) ~ ~ ~ ~ 5 5 1.3Tg5 1.3Tg5(a)
Figure 5. The change in the mechanical properties induced by the thermal loading process. (a) Change in
the elastic stiffness coefficients C115a and C44 a
5 during thermal loading (process D→G) at a final cooling rate
of Rq5 and various annealing temperatures Ta. The elastic stiffness coefficients of the as-quenched model with
a cooling rate Rq (state G) are C11q =171 9. GPa and C44q=57 5. GPa; these are adopted as reference
values. The elastic stiffness coefficient of the as-quenched model with a cooling rate Rq5 are shown by the
dashed lines. (b) The shear stress change during the shear deformation tests for four models; two as-quenched models (state D) with different cooling rates, Rq and R5q (>Rq), and two annealed models (state G)
with different annealing temperatures, Tg (aged model) and . T1 67 g (rejuvenated model) with the same
cooling rate of after annealing, Rq5(=R5q). Inset figures represent snapshots of the two annealed models
of catastrophic shear localization behavior such as shear banding. We actually demonstrated that rejuve-nation enhances more homogenous deformation in Fig. 5(b), which supports a fictive-temperature-based discussion of ductility in amorphous metals.
Conclusion
In summary, we have conducted thermal loading MD simulations of an already-formed amorphous metals subjected to heating, isothermal annealing, and second quenching and have produced a rejuve-nation map with respect to the annealing temperature and second quenching rate that allows conditions to be found to control the level of rejuvenation. We found that thermal rejuvenation occurs via a thermal processing of isothermal annealing at temperatures above . T1 1 g and subsequent quenching at a cooling
rate that is higher than that of the initial quenching process. The level of rejuvenation increases with increasing annealing temperature and quenching rate. We also experimentally demonstrated control of the level of thermal rejuvenation in Zr55Al10Ni5Cu30 bulk glassy alloy. The above results are qualitatively
consistent with analytical models of fictive temperature evolution in quenching and heating glass DSC analysis26, but now adding a constructive notion of applying the heating-quenching process multiple
times to engineer glass properties, with quantitative proofs that the scheme actually works.
The fact that rejuvenation is realized via annealing even slightly above Tg and subsequent quenching
is important for modifying the glass properties after the material has been cast and processed into near component shape because amorphous metals are easy to handle at temperatures slightly above Tg because
of their high resistance to shape changes and/or crystallization.
One would have the flexibility of applying the thermal rejuvenation very locally, using for example transient laser heating; if the laser-heated spot is very small, the local cooling rate afterwards could be much higher than with the entire glassy block cooling down from melt. In practical thermal loading, the achieved level of rejuvenation will vary across the sample because heterogeneous heat transfer and heat conduction over time and space are expected. If we need to demonstrate the details of the heterogeneity in an actual heat treatment process, then we could employ a coarse-grained heat transfer and tempera-ture distribution analysis such as FEM in combination with our rejuvenation map. This analysis frame-work could be a useful engineering tool for predicting heterogeneous rejuvenation and engineering the desired rejuvenation distribution while avoiding crystallization and/or gross shape changes with certain boundary conditions. Recently, nanoimprinting of metallic glass35 has been developed using a
thermo-plastic forming process slightly above Tg, which allows the realization of low-cost fabrication of micro-
and nanodevices. Our study proposes that a thermoplastic forming process consisting of annealing slightly above Tc and then rapid cooling before the glass relaxes into equilibrium liquid would avoid
embrittlement by aging in the thermoplastic forming process.
References
1. Greer, A. L. Metallic Glasses. Science 267, 1947–1953 (1995).
2. Inoue, A. Stabilization of metallic supercooled liquid and bulk amorphous alloys. Acta Mater. 48, 279–306 (2000).
3. Schuh, C. A., Hufnagel, T. C. & Ramamurty, U. Mechanical behavior of amorphous alloys. Acta Mater. 55, 4067–4109 (2007). 4. Asami, K., Kawashima, A. & Hashimoto, K. Chemical Properties and Applications of Some Amorphous Alloys. Mater. Sci. Eng.
99, 475–481 (1988).
5. Masumoto, T. & Maddin, R. The mechanical properties of palladium 20 a/o silicon alloy quenched from the liquid state. Acta
Metall. 19, 725–741 (1971).
6. Ashby, M. F. & Greer, A. L. Metallic glasses as structural materials. Scr. Mater. 54, 321–326 (2006).
7. Lewandowski, J. J. & Lowhaphandu, P. Effects of hydrostatic pressure on the flow and fracture of a bulk amorphous metal. Phil.
Mag. A 82, 3427–3441 (2002).
8. Zhang, Z. F., Eckert, J. & Schultz, L. Fatigue and Fracture Behavior of Bulk Metallic Glass. Metall. Mater. Trans. A 35, 3489–3498 (2004).
9. Conner, R. D., Yim, H. C. & Johnson, W. L. Mechanical properties of Zr57Nb5Al10Cu15.4Ni12.6 metallic glass matrix particulate
composites. J. Mater. Res. 14, 3292–3297 (1999).
10. Sarac, B. & Schroers, J. Designing tensile ductility in metallic glasses. Nat. Comm. 4, 2158 (2013). 11. Das, J. et al. “Work-Hardenable” Ductile Bulk Metallic Glass. Phys. Rev. Lett. 94, 205501 (2005).
www.nature.com/scientificreports/
12. Zheng, N. et al. Design of ductile bulk metallic glasses by adding “soft” atoms. Appl. Phys. Lett. 100, 141901 (2012).
13. Kumar, G., Neibecker, P., Liu, Y. H. & Schroers, J. Critical fictive temperature for plasticity in metallic glasses. Nat. Comm. 4, 1536 (2013).
14. Hodge, I. M. Physical Aging in Polymer Glasses. Science 267, 1945–1947 (1995).
15. Utz, M., Debenedetti, P. G. & Stillinger, F. H. Atomistic Simulation of Aging and Rejuvenation in Glasses. Phys. Rev. Lett. 84, 1471–1474 (2000).
16. Concustell, A. et al. Structural relaxation and rejuvenation in a metallic glass induced by shot-peening. Phil. Mag. Lett. 89, 831–840 (2009).
17. Meng, F., Tsuchiya, K. S. II. & Yokoyama, Y. Reversible transition of deformation mode by structural rejuvenation and relaxation in bulk metallic glass. Appl. Phys. Lett. 101, 121914 (2012).
18. Kumar, G., Rector, D., Conner, R. D. & Schroers, J. Embrittlement of Zr-based bulk metallic glasses. Acta Mater. 57, 3572–3583 (2009).
19. Kumar, G., Prades-Rodel, S., Blatter, A. & Schroers J. Unusual brittle behavior of Pd-based bulk metallic glass. Scr. Mater. 65, 585–587 (2011).
20. Kobayashi, S., Maeda, K. & Takeuchi, S. Computer Simulation of Deformation of Amorphous Cu57Zr43. Acta Metall. 28, 1641–
1652 (1980).
21. Nòse, S. A unified formulation of the constant temperature molecular dynamics methods. J. Chem. Phys. 81, 511–519 (1984). 22. Parrinello, M. & Rahman, A. Polymorphic transitions in single crystals: A new molecular dynamics method. J. Appl. Phys. 52,
7182–7190 (1981).
23. Moynihan, C. T., Easteal, A. J., Wilder, J. & Tucker, J. Dependence of the glass transition temperature on heating and cooling rate. J. Phys. Chem. 78, 2673–2677 (1974).
24. Saida, J., Yamada, R. & Wakeda, M. Recovery of less relaxed state in Zr-Al-Ni-Cu bulk metallic glass annealed above glass transition temperature. Appl. Phys. Lett. 103, 221910 (2013).
25. Tool, A. Q., Relation between inelastic deformability and thermal expansion of glass in its annealing range. J. Am. Ceram. Soc.
29, 240–253 (1946).
26. Moynihan, C. T., Easteal, A. J. & Debolt, M. A., Tucker J. Dependence of the Fictive Temperature of Glass on Cooling Rate. J.
Am. Ceram. Soc. 59, 12–16 (1976).
27. Wakeda, M. & Shibutani, Y. Icosahedral clustering with medium-range order and local elastic properties of amorphous metals.
Acta Mater. 58, 3963–3969 (2010).
28. Wakeda, M., Shibutani, Y., Ogata, S. & Park J. Relationship between local geometrical factors and mechanical properties for Cu-Zr amorphous alloys. Intermetallics 15, 139–144 (2007).
29. Li, J. AtomEye: an efficient atomistic configuration viewer. Modelling Simul. Mater. Sci. Eng. 11, 173–177 (2003).
30. Wada, T., Inoue, A. & Greer, A. L. Enhancement of room-temperature plasticity in a bulk metallic glass by finely dispersed porosity. Appl. Phys. Lett. 86, 251907 (2005).
31. Sergueeva, A. V. et al. Shear band formation and ductility of metallic glasses. Mater. Sci. Eng. A 383, 219–223 (2004). 32. Shimizu, F., Ogata, S. & Li, J. Yield point of metallic glass. Acta Mater. 54, 4293–4298 (2006).
33. Shimizu, F., Ogata, S. & Li, J. Theory of Shear Banding in Metallic Glasses and Molecular Dynamics Calculations. Mater. Trans.
48, 2923–2927 (2007).
34. Wang, Q. et al. Superior Tensile Ductility in Bulk Metallic Glass with Gradient Amorphous Structure. Sci. Rep. 4, 4757 (2014). 35. Kumar, G., Tang, H. X. & Schroers, J. Nanomoulding with amorphous metals. Nature 457, 868–872 (2009).
Acknowledgments
This work was supported by Grants-in-Aid for Challenging Exploratory Research (Nos. 23656418 and 25630013), Scientific Research (A) (No. 23246025), and the Elements Strategy Initiative for Structural Materials (ESISM). J.L. acknowledges support by NSF DMR-1240933 and DMR-1120901.
Author Contributions
M.W. performed the molecular dynamics simulations and J.S. conducted the experiments. J.L. and S.O. assisted with data interpretation. M.W. and S.O. designed, and M.W., J.L. and S.O. wrote the paper. All authors discussed the results and participated in manuscript editing.
Additional Information
Supplementary information accompanies this paper at http://www.nature.com/srep Competing financial interests: The authors declare no competing financial interests.
How to cite this article: Wakeda, M. et al. Controlled Rejuvenation of Amorphous Metals with Thermal Processing. Sci. Rep. 5, 10545; doi: 10.1038/srep10545 (2015).
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Com-mons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/