HAL Id: hal-01454825
https://hal.archives-ouvertes.fr/hal-01454825
Submitted on 8 Feb 2017
HAL is a multi-disciplinary open access
archive for the deposit and dissemination of
sci-entific research documents, whether they are
pub-lished or not. The documents may come from
teaching and research institutions in France or
abroad, or from public or private research centers.
L’archive ouverte pluridisciplinaire HAL, est
destinée au dépôt et à la diffusion de documents
scientifiques de niveau recherche, publiés ou non,
émanant des établissements d’enseignement et de
recherche français ou étrangers, des laboratoires
publics ou privés.
An Auger and XPS survey of cerium active corrosion
protection for AA2024-T3 aluminum alloy
Arnaud Uhart, Jean-Bernard Ledeuil, Danielle Gonbeau, Jean-Charles Dupin,
Jean-Pierre Bonino, Florence Ansart, Julien Esteban
To cite this version:
Arnaud Uhart, Jean-Bernard Ledeuil, Danielle Gonbeau, Jean-Charles Dupin, Jean-Pierre Bonino,
et al.. An Auger and XPS survey of cerium active corrosion protection for AA2024-T3 aluminum
alloy. Applied Surface Science, Elsevier, 2016, 390, pp.751-759. �10.1016/j.apsusc.2016.08.170�.
�hal-01454825�
O
pen
A
rchive
T
OULOUSE
A
rchive
O
uverte (
OATAO
)
OATAO is an open access repository that collects the work of Toulouse researchers and
makes it freely available over the web where possible.
This is an author-deposited version published in :
http://oatao.univ-toulouse.fr/
Eprints ID : 16639
To link to this article : DOI:10.1016/j.apsusc.2016.08.170
URL :
http://dx.doi.org/10.1016/j.apsusc.2016.08.170
To cite this version :
Uhart, Arnaud and Ledeuil, Jean Bernard and
Gonbeau, Danielle and Dupin, Jean-Charles and Bonino, Jean-Pierre
and Ansart, Florence and Esteban, Julien An Auger and XPS survey
of cerium active corrosion protection for AA2024-T3 aluminum
alloy. (2016) Applied Surface Science, vol. 390. pp. 751-759. ISSN
0169-4332
Any correspondence concerning this service should be sent to the repository
administrator:
staff-oatao@listes-diff.inp-toulouse.fr
An
Auger
and
XPS
survey
of
cerium
active
corrosion
protection
for
AA2024-T3
aluminum
alloy
A.
Uhart
a,
J.B.
Ledeuil
a,b,
D.
Gonbeau
a,
J.C.
Dupin
a,∗,
J.P.
Bonino
b,
F.
Ansart
b,
J.
Esteban
c aIPREM-ECP-UMRCNRS5254,UniversitédePauetdesPaysdel’Adour,TechnopoleHélioparc,2AvenuePrésidentPierreAngot,64053PauCedex09,France bUniversitédeToulouse,UPS-INP-CNRS,InstitutCarnotCIRIMAT,118RoutedeNarbonne,31062ToulouseCedex09,FrancecMessier-Bugatti-Dowty,EtablissementdeMolsheim,3,rueAntoinedeStExupéry,67129Molsheim,France
Keywords: XPS Auger Cerium Conversioncoating AA2024alloy Corrosion 1. Introduction
Aluminumanditsalloysarewidelyusedinaerospaceindustry becauseoftheirlowdensityandmechanicalproperties.Despite the protective film ((hydro) oxide thin film) formed in mois-tureconditions,thehighreactivitycommonlyobserveddrastically increasesincontactwithsolutionscontainingcomplexingagents (e.g.,halides)[1–4].Thecorrosionprocessofaluminuminvolves theadsorptionofaggressiveionsonthe(hydr)oxidealuminum sur-face(e.g.,Cl−),thedissolutionofthisprotectivelayerandthedirect
attackofthemetal[5].Asolutiontoimprovethecorrosion resis-tanceistouseprotectivecoatings.Chromates(CrVI)compoundsare
themostcommonsubstancesusedduetotheirefficiencyinsevere atmosphereandtheirlowcost[6].Chromatescanbeintroduced bythreemainways:directincorporationintoconversioncoatings, onlyaddedinanodizingbathsandaspigmentsinpaintingprimers. However,theuseofchromatesincoatingsinvolvedserious envi-ronmentalproblemsforthesurfacetreatmentindustry,because hexavalentchromatesarecarcinogenicandhighlytoxic[5].Since environmentalregulationsgotstricterinthelastfewyears,withthe comingprohibitionuseofCrVIcompoundsincorrosionprotection
∗ Correspondingauthor.
E-mailaddress:dupin@univ-pau.fr(J.C.Dupin).
systems(REACH,2017),differentalternativesubstitution formu-lationshavebeenextensivelystudied.Oneofthemostpromising systemsisbasedonrare-earth(Ce,Nd,La,Y)impregnation solu-tions[7–9].Moreover,Hintonetal.haverevealedthehighpotential of ceriumions inthereduction of thecorrosionrate[7]. Some newcompositeformulations(e.g.,Ce-dopedsol-gelnetwork)offer bothactive(electrochemical)andpassive(barrier)protectionfor theunderlyingalloys[2,4,10,11].TheideaofCeconversion coat-ingsasactiveprotectionfilmsonaluminum alloysisstill akey parameterinthegeneralthoughtandrecentworkseven recog-nizedanenhancedactivitywithaprioracidsurfacepre-treatment
[12].Allthesepreliminarystudiesinspiredanincreasinginterest forresearchersandespeciallyaircraftmanufacturerswhowantto progressintheknowledgeoftheactiveprotectionfilmformation processtooptimizethelifecycleofthecoateddevices.The Cerium-basedconversionprocesshasalreadybeenundertakenwiththe differentceriumsalts[13]and,foreachofthem,withthe addi-tion(ornot)ofdifferentsodiumsalts.Mainobservationswerethe nitrateionprovedtobeastronginhibitorandthesulphateionhad nomarkedeffect(onlyveryslightinhibition).
Thepurposeofthispaperistogivecomplementary informa-tioninthetopicabouttheroleofceriuminhibitorviathestudy oftheconversioncoating(CC)compositionbyadualX-Ray pho-toelectron(XPS)/Augerspectroscopiesanalysis.Foraconvenient protectiveprocessusedintheindustry,suchaconversioncoating
isalwayspre-depositedonto themetalsupportbeforea primer (filler,UVabsorber,...)andtopcoat(againstmechanicalabrasion andenvironmentalerosion)[14].Then,specificpropertiesofthe CeriumCCdiscussedhereafterhavetobeconsideredinaglobal framework.Thecorrosionprotectionofthesomepartsusedinthe aeronauticsisthenthegoodalchemybetweentheactive(CC)and thepassive(primer/paint)assembliesofthewholeprotective sys-tem.Ourshortstudy,onlyfocusedontheactiveprotectiondeposit, presentsanoriginalsurvey,atthesub-micronscale,ofthechemical evolutioninthesurroundingofthecorrodedzones(pits,crevices). Withtheseadaptedtoolsofsurfacecharacterization,migrationof entitiesandchangesintooxidationstatesaredirectlyand simulta-neouslyrecorded.TherealroleoftheactiveCClayerscanbethen wellunderstood.
2. Materialsandmethods 2.1. Materialandtreatments
ThematerialstudiedwasAA2024-T3aluminumalloycomposed by(inwt%)3.8–5.0Cu,0.2–1.2Mg,0.4–1.0Mn,0.5–1.2Si,<0.7Fe, <0.1Cr.Eachsamplesurface(80mm×42mm×1mmpanel)was cleanedandpreparedusingseveralsteps;afteracetonedegreasing, achemicalpretreatmentwasperformedasitfollows:a 20-min immersionina NaOHsolution(pH=9)maintainedat60◦C,
fol-lowedbyarinsingwithdeionizedwater;anda5minneutralization inanaqueoussolutionofNaNO3atpH=0.95atroomtemperature.
Thesampleswerefinallywashedinethanolanddriedinair. Ceriumbasedconversioncoatings(CeCC)weredepositedatroom temperaturebyimmersionofthealloypanelinawatersolution containingCe(NO3)3,6H2O(Fluka, CAS:10294-41-4) ata
differ-entceriumconcentrations(0.01M,0.05M,0.1Mand0.5M).The pHoftheimmersionbathismaintainedto4andnoaccelerating agent(e.g.,H2O2)wasused.Onceimmersedduring300s(duetime
forahomogeneous/coveringfilmandbetterelectrochemical per-formance),sampleswererinsedfourtimeswithdeionizedwater anddriedatroomtemperatureinadesiccator.CeCCwerefinally about2.1±0.5mmthick(examinedbySEM)withyellow/brown colorization,aftera300simmersionintheconversionbath.
Aqueous corrosion solutions—Corrosion attack wasconducted on the samples in a 3.5wt.% NaCl solution at 25◦C. Time of
immersionwassetto1haccordingtotheappearanceoffirst cor-rosionpits. Asingleandspecific testof ioniccrosssectionwas achievedontheAA2024substratecoveredwithCe-basedcoating ([Ce]=0.1M)withaJEOLIB-0901crosspolisher.Analytical condi-tionsarereportedelsewhereinarecentwork[15].After96hunder airexposure,samplessurfaceandcrosssectionwereanalyzedwith XPSandAuger; this operationwasa firsttrytoinvestigatethe corrosionpropagationandtheeffectoftheinhibitordeepinthe material.
Theelectrochemicalbehaviorofthesystemswasevaluatedby electrochemicalimpedance spectroscopy (EIS)in a 0.05MNaCl staticsolution(pH=6.0)forthedifferentpanelsoriginalygotfrom severalimmersiontimes(1s,60sand300s)intheconversionbath. Fortheelectrochemicalmeasurements,athree-electrode electro-chemicalcellwasused,consistingofaplatinumcounterelectrode, asaturatedcalomelreferenceelectrodeandthesamplewasused asaworkingelectrode,withanexposedareaequalto15cm2.The
experimentalapparatususedfortheelectrochemicalinvestigation wasapotentiostat(AUTOLABPGSTAT30)andafrequencyresponse analyzer(FRA). EISmeasurementswere performedin potentio-staticmodeattheOCP,obtainedaftera1hstabilizationofthe potentialintheelectrolyte.TheamplitudeoftheEISperturbation signalwas10mV,andthefrequencystudiedrangedfrom100kHz to10mHz.Onlyvaluesofthechargestransferresistance(R)are
reportedelsewhereinthetextforanyconcentrationsofcerium testedintheconversionbath.
2.2. Surfaceanalysis
2.2.1. Morphologyofmaterialswithscanningelectron microscopy(SEM)
Themorphologyandthemicrostructureofthecoatings,before andaftertheimmersiontestsincorrosionsolutions,wereobserved by Scanning Electron Microscopy (SEM). High resolution high-energy images were obtained with a JEOL JAMP-9500F Auger spectrometer(JEOLLtd,Tokyo,Japan)workingunderprecise condi-tions(30keV,2nA,workingdistance=23mm,pressure<2.10−7Pa)
fittedwithaSchottkyfieldemissionelectrongunusinga conven-tionalsecondaryelectrondetector(SED)intheanalysischamber. Thepresentanalysismodepermitsahighdepthfieldfor nanopar-ticlesvisualization.
2.2.2. SurfacechemicalanalysiswithX-rayphotoelectron spectroscopy(XPS)
IntheaimtocharacterizetheCeCCcomposition,X-Ray Photo-electronSpectrometry(XPS)measurementswereperformedona ThermoK-alphaspectrometerwithahemisphericalanalyzerand amicrofocussed(analysis areawasc.a.400mm2) monochroma-tizedradiation(AlKa,1486.6eV)operatingat72Wunderaresidual pressureof1×10−9mbar.Thepassenergywassetto20eV.Charge
effectswerecompensatedbytheuseofa chargeneutralisation system(low energy electrons)which had the uniqueability to provideconsistentchargecompensation.Alltheneutraliser param-etersremainedconstantduringanalysisandallowonestofinda 285.0eVC1sbindingenergyforadventitiouscarbon.Spectrawere mathematicallyfittedwithCasaXPSsoftwareusingaleastsquares algorithmandanon-linearbaseline.Thefittingpeaksofthe exper-imentalcurvesweredefinedbyacombinationofGaussian(70%) andLorentzian(30%)distributions.Onlycorelevelsspectraforthe elementswithhighestphotoionizationcrosssectionwererecorded inordertoextractmorereliableinformation.
2.2.3. Surfacechemicalanalysiswithaugerspectroscopy(XPS) andchemicalmappingwithscanningaugermicroscopy(SAM)
TheAugeranalyseswerecarriedout withthepreviousJEOL JAMP9500FAugerspectrometerworkingunderUHVconditions (pressure<2.10−7Pa).The UHVequipmentwasa Schottky field
emissionAugerelectronspectrometer(FE-AES)dedicatedtovery highspatialresolution analysis andhigh brightness.The hemi-sphericalelectronanalyzercoupledwithamultichanneldetector (7channeltrons)offeredidealsettingsforenergyresolvedAuger analysis.
3. Resultsanddiscussion 3.1. AA2024-T3surfacepreparation
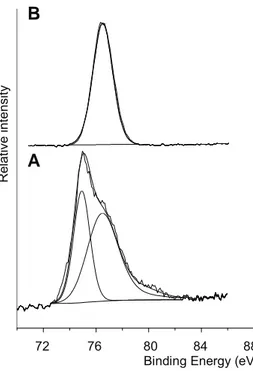
TheAA2024-T3alloysurfacewasinitiallycleanedbeforeCeCC asreportedintheMaterialsandtreatmentsection.Thechemical surfacestateiscontrolledwiththeXPSandresultsshowahigh initialcontentofhydrocarbonentitiesattheAAl2024-T3surface (Fig.1A).
Thesurfaceisfullcontaminatedasnoalloyelementsarevisible. Oncechemicallytreatedwithacidandalkalinebaths(Fig.1B),the nearalloysurfaceexhibitsacleardecreaseofthecarboncomponent (Table1)andanaluminumsignalwithtwodistinctcomponents: –at71.8eVassociatedwiththemetallicAl◦oxidationstate,–and
at74.6eVcorrespondingtoAl-O(H)environment;thethinnative (hydro)oxideoverlayerisapotentialanchorageareaforthefuture conversioncoating.CopperCu2psignalattestsofthebaremetal
80 78 76 74 72 70 68
Binding Energy (eV)
1000 800 600 400 200 0
N
o
rm
a
liz
e
d
I
n
te
n
s
it
y
Binding Ene
rgy (eV)
B
A
C1s O1s OAuger Al2s Al2p F1s Cu2p FAuger Al° Alox/hydrox Cu3pFig.1. LargescaleXPSspectrumofAA2024-T3alloy:(A)beforeand(B)aftersurfacepreparation(degreasing/acidcleaning).
Table1
SurfacecompositionoftheAA2024-T3substrate:beforeandafterdegreasing/acid treatmentandcoveredwithCeCC([Ce]=0.1M)after300sofimmersioninthe conversionbath.
Element(at.%) C O Al Ce N Cu F
Beforesurfacepreparation 96.5 3.5 – – – –
Aftersurfacepreparation 27.1 46.3 25.5 – 0.2 0.9
AA2024-T3/CeCC([Ce]=0.1M) 31.0 46.8 3.8 12.2 6.2 – –
detectionandtheefficiencyofthecleaningtreatment. Effective cleaningpre-treatmentsareoftendemonstratedtobeassociated withthegenerationofsurfacedefaults,grainedtopologyfavoring redoxreactionsnecessarytotheCeCCformation[16].
Fluorine components oflow intensityobserved in theclean samplespectrumcomefromaresidualpollutionintheanalysis chamberintheperiodoftheexperiment.Itdoesnothaveanyeffect ontothesurfacechemistryasithasbeenmainlyfound,inverylow concentration(around0.9at.%),bondedtoresidualhydrocarbon entities.
3.2. Ce-basedconversioncoating(CeCC)overAA2024-T3before corrosion
TheCeCCcoveringisexpectedtobequitehomogeneouswhich isfundamentalforthefutureindustrialapplications.SEMimagesof theCeCCpanelssurfaceweresystematicallyrecordedinthe differ-entimmersionconditions([Ce],immersiontime).Beforecorrosion, theappearanceofthesurfacecoatingisquitehomogeneousfor ceriumconcentrationsrangingfrom0.01Mto0.1Mwhateverthe immersiontime.OnfigureESI1,onecanobserveinthecaseofthe Ce(0.1M)CCcoating(300sofimmersion)asurfacerarelyaffected bytopographydefects(holes,excrescences,...).Whenincreasing theceriumconcentration(e.g.,[Ce]=0.5M),rightfromthefirst sec-ondsofimmersion,thecoatingiscrackedandcanpeeloffforlonger stayintheconversionbath.Thenthehigherthecerium concentra-tionis,themoredefectivethecoatingsurfacebecomes.
XPSanalysisforthecoatedpanelshasshownwell-identified chemical entities from the coating but also from the AA2024 substrate.For instance, after300sof immersionin the conver-sionbath,thelargescalesurvey (Fig.2A) ofAA 2024-T3/CeCC (e.g.,[Ce]=0.1M)systemdisplaysemergenceofthemain differ-entorbitals(Al2p,Al2s,C1s,O1sandCe3d)withintensesignals (Table1).Thecoating,veryfewmicronsthick,appearstobequite homogeneousandleveling(Fig.2B)buttheobservationofsome substrateelementswiththeXPStechniquewouldattestofa cer-tainporosityofthedeposit.After1sofimmersion,TheXPSdata give,atthesubstratesurface,aCe/Alratiobetween0.4and0.5for [Ce]=0.1Mwhenitis6timeshigherforthe[Ce]=0.5Min rela-tionwitharealdifferenceofdepositthickness.Forlongertimes ofimmersion(e.g.300s),theCe/Alratioisquitestablefor0.1M whereasitdrasticallydropsfor0.5M.Thiscouldconfirmthe dam-agingincreaseofthecoatingathighceriumconcentrationwiththe immersiontime.
Differentareasofanalysiswerepointedoutandnodifference wasfoundforthechemicalcompositionoftheCeCCcoating traduc-ingagoodhomogeneity.Theceriumcorrosioninhibitorhasbeen recordedinquiteimportantat%between2.6%(for[Ce]=0.01M) to13.7%for[Ce]=0.5M).Forallthecoatings,thechemicalsurvey attestsofamixtureofceriumspecies(Ce+IIIandCe+IV,seefigure ESI2)asreportedelsewhere[17].Histogramsshowamain propor-tionofCe+IVforpanelscoveredupatlowceriumconcentration whenCe+IIIisinthemajorityforhigherceriumconcentration.
In the high resolution Ce3d spectrum, a same structured experimentalprofileisrecorededwhateverthedifferentcerium concentrationoftheCCbathandimmersiontimes.Onlyintensities varyaccordingtothecontentofCe+IIIandCe+IVspecies.InFig.3A andtheTable2(e.g.,Ce(0.1M)CCimmersed300s),fourmainpeaks correspondingtothepairsofspin-orbitdoubletscanbeidentified, ingoodagreementwithotherauthors[20,21]and thepresence ofa+IIIoxidationstate.Thehighestbindingenergypeaks,u1and
u’1respectivelylocatedatabout885.8±0.2eVand904.3±0.2eV
aretheresultofaCe3d94f1O2p6finalstate.Thelowestbinding
energystatesu0andu’0respectivelylocatedat881.8±0.2eVand
Fig.2.Cerium-basedconversioncoating(with[Ce]=0.1M;300sofimmersionintheconversionbath)overAA2024-T3alloy:(A)largescaleXPSspectrumand(B)SEM micrographofthecrosssection.
R e la ti v e i n te n s it y 870 880 890 900 910 920 930
Binding Energy (eV)
A
u0 u’0 u1 u’1 v’2 v2 v1 v’0 v0 v’1B
u0 u1 u’1 u’0 870 880 890 900 910 920 930Binding Energy (eV)
Fig.3.XPSCe3dspectrumoftheCe(0.1M)CC(300simmersiontimeinthe con-versionbath)/AA2024-T3:(A)as-preparedand(B)after180sofcompucentricAr+
erosion(rastersize:0.8mm,IE:3000eV).
Table2
Labeling,meanpositionofthe10componentsofamixedCe(III)/Ce(IV)sample accordingto(*)meanpositionofpeaksforthedifferentCe(0.1M)CC(300s immer-siontime)ontoAA2024-T3substrate.
Spin-orbit component electronicstate attribution (*)peakposition (eV)±0.5eV Ce(III) u0 3d5/2 Ce(III)3d94f1 O2p6 880.7 u0′ 3d3/2 901.1 u1 3d5/2 Ce(III)3d94f2 O2p5 885.6 u1′ 3d3/2 904.6 Ce(IV) v0 3d5/2 Ce(IV)3d94f2 O2p4 882.5 v0′ 3d3/2 903.4 v1 3d5/2 Ce(IV)3d94f1 O2p5 886.8 v1′ 3d3/2 907.5 v2 satellite Ce(IV)3d94f0 O2p6 898.8 v2′ satellite 916.7
Someminorcomponentsowingtohybridizationwithligand orbitalsand partialoccupancyofthevalence 4forbitalarealso observed.Sixnewpeakslabeledv0,v1,v2(for3d5/2)andv0′,v1′,
v2′(for3d3/2)referringtothethreepairsofspinorbitdoubletsare
characteristicofCe4+3dfinalstates[22,23].Thelowestbinding
energystatesv0(v’0),v1(andv’1)respectivelylocatedat882.5eV
(903.4eV),886.8(and907.5)aretheresultofCe3d94f2O2p4and
Ce3d94f1O2p5finalstates.Thehighestbindingenergypeaks,v2
andv’2respectivelylocatedatabout916.7and898.8±0.2eVare theresultofaCe3d94f0O2p6finalstate.Thesatellitepeakv2
asso-ciatedtotheCe3d3/2ischaracteristicofthepresenceoftetravalent
Ce(+IVoxidationstate)duetoapartialoxidationofceriuminthe referencematerial.Thisfactisprettyinterestingasthis compo-nentisisolatedfromtheothersatapprox.916–917eV,asamatter offact,onecanusethispeakasaquantitativeprobeoftheamount ofCe(IV)[18,24–26].
InsuchacoatingtheminorCe+IVspeciesseemtobelocated inthetoplayerpartasdemonstratedbysoftAr+ionerosionwhich
revealsonlyaCe+IIIstateafter180sofsurfaceerosion(Fig.3B). SuchobservationswereevenreportedinafurnishedworkofL.S. Kastenandcoll.[18].Ce+IV(depositionofCeO2-2H2OorCe(OH)4
layer)isfoundtobepredominantunderdifferentexperimental conditions:whenthecerium-basedconversioncoatingisachieved using,forinstance,chlorideceriumsaltbasedconversionbathin presenceofhydrogenperoxide[19].AtlowCeconcentration,the smallerthicknessofthedepositinvolvestheoxidationofmost dis-persedCe+IIIspecieswhichislessevidentforthickercoating(high Ceconcentration)forwhichonlytoplayersofCe+IIIareoxidized. TheoxidationeffectismorepronouncedforCe(0.1M)CCcompare toCe(0.5M)CCcertainlyduetoamoreimportantporosityforthis lastone.
Indeed,ithasbeendemonstratedthattheintegralareaofv2′
peakwithrespecttothetotalCe3dareacouldbetranslatesinto percentageofCe4+[24,27].Shyuandcoll.[28]havedemonstrated
thattheintegralareaofthev2′peakwithrespecttothetotalCe3d
areacouldbetranslatedintopercentageofCe4+withtherelative
errorofbeingintherangeof10%.InthecaseofpureCe4+,the
v2′peakshouldconstitutearound14%oftotalintegralintensity.
A
B
Rel a ti v e i n te n s it y 72 76 80 84 88Binding Energy (eV)
72 76 80 84 88 Binding Energy (eV)
Fig.4.XPSAl2pspectrumof:(A)Ce(0.1M)CC(300simmersiontimeinthe con-versionbath)/AA2024-T3and(B)AlCeO3referencematerial.
Ce4+reportedintheliterature,percentageofCe4+wascalculated
by: Ce4+=
′ 2%
14 ×100 (1)
wherev2′%isthepercentageofv2′peakareawithrespecttothe
totalCe3darea.
For alltheanalyzed panelsunder differentpreparation con-ditions, the Al2p experimental is quite the same with a two componentsconvolution.Fig.4AofCe(0.1M)CCimmersed300s
showstwochemicalenvironments:thecomponentat74.8eVcan beassociatedwiththenativeoxidelayerontopAA2024-T3 sub-stratewhiletheoneonhighenergyside(76.9eV)isattributedtoa Al/O/Cemixedchemicalenvironmentasdetectedinacommercial stoichiometricAlCeO3material(CAS12014-44-7,Sigma-Aldrich)
analyzedinsameexperimentalconditions(Fig.4B).Thenpartofthe ceriumentitiesseemstobelocatedinthenearsurfaceofthealloy (neartheAloxide/hydroxidetoplayer)whichcouldbeakeypoint inthecorrosionprotection,intermsofproximitywiththemetal surface.Thefactthatonecanobservethepresenceofaluminum, evenifthecoatingisaround2.0mmthick,would alsosignifya certainporositydegreeofthedeposit.
Whatevertheconditionsofimmersiontime,acomplementary surveyofthesurfacesamplewassystematicallydonewithanAuger mapping(SAM)analysistocontrolthespatialchemical homogene-ityofchemicalelements.
InFig.5,thecaseoftheCe(0.1M)CCcoatingimmersed300sin theconversionbathispresentedandfewsurfacedefects(cracks, holes,...)arenoticedasdiscussedelsewhere,attestingofagood levelingdeposit inthepresent conditionsofdeposit.TheAuger mappingallowsonetoconsideragooddispersalofceriumwiththe experimentaldepositprocessusedinthiswork.Theobservationof thealuminumandoxygeninquitegoodrelativecontentconfirms thenativealuminum(hydro)oxideoverlayerintheproximityof thecerium.
3.3. Ce-basedconversioncoating(CeCC)overAA2024-T3after corrosion
3.3.1. Corrosionresistance
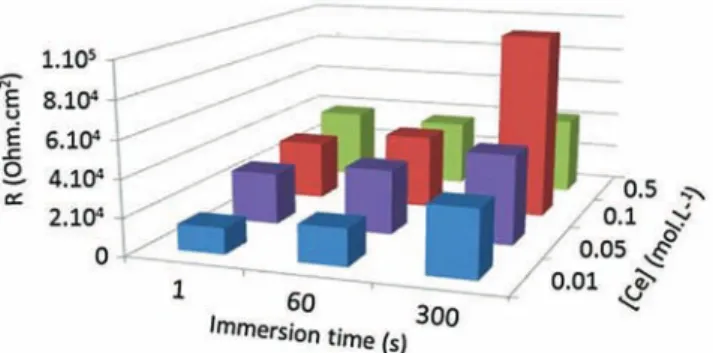
Theelectrochemicalresults(Fig.6),indicate,afteronesecondof immersionina0.05MNaClstaticsolution(pH=6.0),thatin con-trolledproportions,theadditionofceriumintheconversionbath leadstotheincreaseof theanti-corrosionproperties.However, itseemsthereisacerium concentrationthreshold(0.1M)over whichthecorrosionresistancefails.Thecoatingdopedwith0.5M Ce(NO3)3showsthelowestresistanceandthehighestcapacitances
Fig.5.Ce(0.1M)CCcoating(immersiontime=300sintheconversionbath)as-prepared:SAMimagesforCe,AlandOelements(RVBscale)andSEMmicrographofthesame area(bottomright).
Fig.6.ChargestransferresistanceevolutionofAA2024T3panelsimmersedin someconversionbathsofdifferentceriumconcentrationsandfordifferenttimes ofimmersion.
Fig.7.(A)SEMmicrographofthecorrodedCeCC(Ce=0.1M)appliedontoAA 2024-T3(300simmersiontime).
attheendofthetestperiod,suggestingthattheincreaseofthe ceriumconcentrationpromotestheformationofmoredefective coatings.Moreover,thecorrosionresistanceisclearly enhanced after300sofimmersion.
Theseeventsareprobablyduetothephysico-chemical struc-turationoftheCeCCwhichcouldchangewiththeceriumcontent (internalconstraints).Actually,inrelationwiththeeffectiverole ofCe+IIIionsextensivelyreportedfortheactiveprotectionofAA 2024-T3alloy[6,29],thepresentstudywillonlyfocusonto cerium-basedconversioncoating(with[Ce]=0.1M)forwhichthebetter corrosionresistanceisreachedafter300sofsubstrateimmersion intheconversionbath.
3.3.2. Chemicalanalysisofthecorrosionattack
OnthecoatedCeCCpanels,whatevertheirinitialimmersion timeandCeconcentrationproceededintheconversionbath,first corrosionpitsappearedbefore1hofimmersionintothe3.5wt.% NaClsolution.After96hunderairexposuredrying,panelswere thenanalyzedwithXPS,SEMandSAM.AglobalSEMmicrograph forthetreatedsubstrateinthe3.5wt.%NaClsolutionispresented inFig.7.Itdisplaysthepresenceofholesrandomlydispersedatthe surfaceandsomezoneshighlydamagedwithalocalizedpeeloff phenomenon.Thewhitenodulesarewell-knownCu-richdeposited particles(e.g.,Al2CuMgmetallicagglomerates...)asdetectedwith
Augerspectroscopyandreportedelsewhere[30–32].Theclose-up SEMmicrographofa“zoomin”corrosionpit(Fig.8)allowsone tobetterappreciatedetailsofthecoatingpeeloffstateandareal changeofcolornuanceinthedamagedareasuggestingachangein
thechemicalcomposition(differentsecondaryelectronsemission recorded).
TheAugerchemicalmappingachievedonthecoatingsurface showscleardifferencesinthespatialdistributionofceriumbefore (fig.5)andafter(fig.8)thecorrosionattacksofthecoatedpanel. Astheceriumisprimarilywelloverlaidontothesubstrateafter theCeCCimmersion,itseemstomovetothepitsareasduringthe corrosionevent.Actuallyarelativehighcontentisobservedinthe hollowringofthecorrosionpitandinitscenterpart.Color inten-sitygradient(inagreementwiththerelativeconcentrationofthe element)attestsofamigrationofceriumfromsurroundedzones tothecorrosionpit.Inthedamagedzone,therelative concentra-tionofceriumincreasesrelatetothesurroundingzones.Thisinflux iscertainlyexplainedfromthemigrationofceriumwhichensures theactivesubstrateprotectionagainstcorrosion.Intheattacked haloofthecoating,ceriumbecomesmoreconcentratedwhen rela-tivealuminumandoxygenamountsfalldown.Thisresultisoriginal andsignificantforthedirectvisualizationofthecorrosioninhibitor behaviorandtheprotectiveactionofCeCC.
Thechemicalmappingofoxygenrelativecontentshowsless drasticchangefromtheundamagedcoatingtothecorrosionpit. Thiscouldcorrespondtotheexistenceofceriumoxide/hydroxide inbothareaswithslightly stoichiometrydifferences.Inanother hand,aluminumisclearlywithdrawnfromtheattackedzone.Some additionalmeasurementswereachievedwithAugerspectroscopy indifferentpointsatthepanelsurface(figureESI.3)beforeand after1hand5hofimmersioninthecorrosiveNaClbath. Quantifi-cationdata,extractedfromtheconvolutionofAugerlinesOKLL (510.0eV),AlKLL(1387.0eV)andCeMNN(625.0eV),clearly con-firmthedisplacementofceriuminthecorrodedpartsofthepanel withanenhancementofitsrelativecontentduringthecorrosion process:18.6at.%intheaspreparedcoatingandupto27.3at.% (1hof immersion)and 39.4at.%(5hofimmersion)inthe cen-terdamagedzone.TheAugerspectroscopyandmappingisgreat complementtoSEMsurveysasthesoleSEMimagesinterpretation cannotexplainthewholerealityofthecoatingsurfaceevolution. Forinstanceinthecaseofthepanelimmersedduring1hinthe corrosivebath,itiseasytomonitortheCemigrationwiththeCe contentfrompoint1towardspoints2and3.Thisobservationgives arealsensetothe“active”protectionoftheinhibitor.
Togofurtherintheunderstandingofchemicaleventsduring thecorrosionprocess,aXPSsurveyhasbeenrunintwospecific pointsofthecorrodedpaneldisplayedinFig.7(labelled1and2). Thisspecificzonewaschosenbecauseoftheimportantsizeofthe observedpit(around60mm×130mm)which fitswiththeused ofthefocusedX-raybeam(30mmdiameter).TheaimoftheXPS analysiswastoprecisethepossiblechemicalchangesduringthe ceriummigration.Forzone1,theCe3dspectrum(Fig.9A)fitsinto twodoubletsassignedtoaCe+III(hydro)oxidizedenvironment withpreviouslyreportedbindingenergies(U:880.8eV–901.3eV) and(V:885.9eV-904.8eV)forsuchoxidationstateofcerium.As thesurfacehasbeenexposedtothetreatmentsolution,thenear surfacemorphologyhasnecessarychangedtomoreroughnessand thedissolutionofthethinCe+IVlayer.Thephysicaland chemi-calperturbationofthesurfacealsoinfluencedtheCe3delectron emissionwhichdisplaysanoisysignal.Lossofcarbonatomic per-centagecombinedwiththeenhancementofcopperandaluminum elementssignalsofthesubstrateputsintoevidencetheNaClattack onthecoating.Thisphenomenonismorepronouncedinthevisible corrosioncrack(zone2)forwhichtheCecontentstronglyincreased (Ceat.%2timesmoreimportantthaninzone1,seeTable3)and substrateismorevisibleasthecoatingpeeledoffinthiszone.In thecorrosionpitzone(2),asignificantevolutionoftheCe3d spec-trumisnoticedwiththeappearanceCe+IVfeaturesandthespecific satellitepeakaround917eV(Fig.9B).
Fig.8. Ce(0.1M)CCcoating(immersiontime=300sintheconversionbath)aftercorrosionattack:SAMimagesforCe,AlandOelements(RVBscale)andSEMimageofthe samearea(topright).
U
0V
1V
0A
B
870 880 890 900 910 920 930Binding Energy (eV)
+III +III +III +III +III +III +III +III +IV +IV +IV +IV +IV +IV
V
2U
1Fig.9. XPSCe3dpeakoftreatedCe=0.1McoatingappliedonAA2024-T3:(A)zone 1and(B)zone2ofFig.7.
Then, it seems the cerium played its inhibitor role in the woundedzonewithagreatkineticanditsmigrationis accompa-niedwithachemicalstatetransformation(Ce+III→Ce+IV).The observationoftheO1sspectrum(Fig.10)givesthesametrend withthe top heights ofthe experimentalprofile which shifted
Table3
XPSSurfacecompositionofzones1and2ofthetreatedCe=0.1Mcoating(300s immersiontime)appliedontoAA2024-T3.
Element(at.%) Cu Ce C O Al N Na Cl Zone1 0.1 7.7 29.8 50.6 6.4 3.8 0.8 0.8 Zone2 0.4 15.0 25.3 43.1 14.5 1.2 0.3 0.2 H2O/NO3 -hydroxides Ce2O3 CeO2 526 528 530 532 534 536 538
Binding Energy (eV)
526 528 530 532 534 536 538 Binding Energy (eV)
A
B
Fig.10.XPSO1speakoftreatedCe=0.1McoatingappliedonAA2024-T3:(A)zone 1and(B)zone2ofFig.7.
asthe damageincrease attesting ofnewelectronicdistribution aroundoxygenatoms.Forzone1(fig.10A),anintensecomponent (BE=533.3eV)associatedwithresidualtracesofnitratesfrom ini-tialceriumsaltandwaterfromthetreatmentbath,isobserved.
Fig.11.CrosssectionofCerium-basedconversioncoating(with[Ce]=0.1M,immersiontime:300s)overAA2024T3alloyafter5hcorrosioninto3.5wt.%NaClsolution:(A) SEMviewand(B)Augerspectroscopyanalysisinthecoatingsection(point1)andwithinthecrevicecorrosionflaw(point2).
Thecomponentat532.1eVcorrespondstothehydroxidespecies (hydroxilizedcarbon,Al(OH)3....)whilethenarrowcomponentat
530.2eVcaneasilybeassociatedtoceriumsesquioxide[33].Inthe areaofthecorrosionpit(zone2),theoxygenpeakonlowenergy side(fig.10B),shiftsto529.6eVwhichitissignificantofaCe+IV stateasfoundinCeO2[32].Ashoulderaround530.2eVisstill
vis-ibleasexpectedintheCe+III/Ce+IVmixturereportedintheCe3d spectrum(fig.9B).Inviewoftheseresults,adirectoverviewofthe chemicalchangesoccurringunderthecorrosiveNaCleffectcanbe screened.Ceriumactsasaself-healingagentintheCeCCcoating withappropriate migrationtotheboundedzonesandthe initi-ationofredoxprocesstoblockthecathodiczones.Actually,the releaseofhydroxylsinthecathodiczonesleadstothe precipita-tionofceriumspeciesmainlyintointermediatehydroxidesthen intoceriumdioxide.
Inthisrespect,Aldykiewiczetal.[34]proposedthatthecathodic depositionofCeO2filmswasduetotheoxidationinthesolution
ofCe3+ionstotetravalentCe(OH)
22+ionsinsolution(whichcan
diffusereachinglocaldefects)followingthePourbaixdiagram[35]: 4Ce3++6H2O+O2→4Ce(OH)22++4H+ (2)
andprecipitateaccordingtothereaction:
Ce(OH)22+→CeO2+2H+ (3)
Tocorroboratethemigration ofceriumspeciesfromthe Ce-basedcoatingtothedamagedzonesoftheAA2024alloy,anoriginal surveywasfocuseddeeperinthesubstrateafter5hofcorrosion attack(Fig.11A).Substratewascross-cutintheareaofawell iden-tifiedcrevicecorrosionflaw.TheSEMmicrographrevealstheinner morphologyinthedirectionofthesection,theunstructured Ce-basedcoatingoverthesubstrateinaccordancewiththegeneral degradationundercorrosiveconditions.Inthecenterpartofthe micrograph,thesubstrateisconsumedandalargecrevice (under-cutpit)extendsoverfewmicrons.AlocalAugerchemicalanalysis inbothzones(labeled1and2onFig.11A)wasachievedtoprecise thechemicalentitiesbehaviorduringthecorrosionmechanism.We focusedtheexperimentona300eV−1500eVenergyscaletoget betterresolutionofthedetectedtransitions.TheAugerspectrum ofthedamagedCe-basedcoating(Fig.11B,point1)mainlyexhibits theAugertransitionsofaluminumandoxygen.Theabsenceofany
ceriumsignalissignificantofthecompletedissolutionofthe Ce-basedlayerorthemigrationofceriumentitiesdeepinthematerial. Thethicknessoftheobservedtop-layerovertheAA2024-T3 sub-strategotthinner(0.9mm±0.2mm)andwouldonlybemadeof nativealuminum(hydr)oxidephasesandhydrocarbon(forcarbon element,CKVVtransitionat263eVcan’tbedetectedintheenergy scaleofthestudy).Withinthecrevicecorrosionflaw,sameanalysis wasrun(Fig.11B,point2)andanewsetoftransitionsisdetected: CeMNN(largecomponentwithtop-heightat630eV)andCuLMM (seriesoftransitionsbetween710eVand902eV).Theco-presence ofcerium,copperandaluminumwasfoundindifferentother ana-lyzedzoneswithinthecrevice.Thismainobservationsuggeststhe precisebehaviorofthecorrosioninhibitorwhichwouldblockthe cathodiczones.
4. Conclusion
Onthewayoftheknowledgeandunderstandingofthecorrosion protectionofAA2024alloy,thepresentpapergivesa complemen-taryviewofthechemicalevolutionsatthesurfaceofthesubstrate. Ceriumconversioncoatingonprepared2024aluminumalloy con-sistedofAloxide,andmainlyofCe+IIIoxideandhydroxide.TheCe stateexhibitedamixtureofCe+IIIandCe+IVdependingonthesalt concentrationintheconversionbath.Theouterlayerofthecoating isCe+IVrichanditprobablyindicatestheCe+IIIoxidizedtoCe+IV duringcoatingformationintheinitialsolution.Thecomparison ofCeconcentrationsoftheconversionhasshownbettercharges transferresistance(thenbetterrelativecorrosionprotection)for apanelimmersedduring300sina[Ce]=0.1Mbath.Forhigher Ceconcentrationsorimmersiontime,thedepositbecomesmore powderedandunsuitabletoaircraftapplications.ThedetailedXPS studyhasallowedonetoconsideraclose-upinteractionbetween ceriumandtheAloxidelayerontopalloywiththeexistenceof Al/O/Cemixedchemicalenvironment. Microscopicand spectro-scopictoolswereevenpreciousindicatorstoappreciatethecerium inhibitorbehaviorintermofdisplacementandchemical modifica-tion.Thelocationofceriumhasbeenmonitoredatanytimewitha preciseSAMsurveywhichiscomplextosetupbutessentialforan elementalscreeningwithinconfinedorverysmallzonesof inter-est.Actually,undercorrosiveconditions,ceriumtendstomove,
in a shorttime, towardswoundedzones leavingthe surround-ingcoatingareaalmostfreeofinhibitorcontent.Thisdirectaction involvesachemicalchangewiththeformationofCe+IVoxideand hydroxide.Sameobservationsweredoneatthesubstratesurface (corrosionpits)andsub-surface(crevices).Theeffectivenessofthe CeCCcoatingbeforethedepositofaprimerand/orafinaltopcoat isdeterminedbythesynergyofmanyexperimentalparameters. Evenifceriumalonecannotguaranteethewholeprotectionofa metalpanelagainstcorrosion,itactsasareallyactiveagentwitha quickkineticforhealingwoundedzones.
Acknowledgments
Thanks to the french aeronautics consortium (AESE pole SOLGREEN©)andtheDGA(DirectionGénéraledel’Armement)for theirtechnicalandfinancialsupport.
AppendixA. Supplementarydata
Supplementarydataassociatedwiththisarticlecanbefound,in theonlineversion,athttp://dx.doi.org/10.1016/j.apsusc.2016.08. 170.
References
[1]K.A.Yasakau,M.L.Zheludkevich,O.V.Karavai,M.G.S.Ferreir,Prog.Org.Coat.
63(2007)352–361.
[2]S.M.Tamborim,A.P.Z.Maisonnave,D.S.Azambuja,G.E.Englert,Surf.Coat.
Technol.202(2008)5591–6001.
[3]S.S.Pathak,A.S.Khanna,T.J.M.Sinha,Prog.Org.Coat.60(2007)211–218.
[4]V.Palanivel,Y.Huang,W.J.V.Ooij,Prog.Org.Coat.53(2005)153–168.
[5]R.T.Foley,Corros.Sci.42(1986)277–288.
[6]M.W.Kendig,A.J.Davenport,H.S.Issacs,Corros.Sci.34(1993)41–49.
[7]B.R.W.Hinton,D.R.Arnott,N.E.Ryan,Mater.Forum7(4)(1984)211–217.
[8]D.Zhao,J.Sun,L.Zhang,Y.T.J.Li,J.RareEarths28(1)(2010)371–374.
[9]A.KumarMishra,R.Balasubramaniam,Corros.Sci.49(3)(2007)1027–1044.
[10]L.E.M.Palomino,P.H.Suegama,I.V.Aoki,Z.Pászti,H.G.deMelo,Electrochim.
Acta52(27)(2007)7496–7505.
[11]W.Pinc,P.Yu,M.O’Keefe,W.Fahrenholtz,Surf.Coat.Technol.203(23–25)
(2009)3533–3540.
[12]K.Brunelli,ManueleDabalà,IreneCalliari,MaurizioMagrini,Corros.Sci.47
(4)(2005)989–1000.
[13]A.Decroly,J.-P.Petitjean,Surf.Coat.Technol.194(2005)1–9.
[14]G.P.Bierwagen,D.E.Tallman,Prog.Org.Coat.41(2001)201–216.
[15]J.B.Ledeuil,A.Uhart,S.Soule,J.Allouche,J.C.Dupin,H.Martinez,Nanoscale (2014),http://dx.doi.org/10.1039/c4nr03211j.
[16]C.M.Rangel,T.I.Paiva,P.P.daLuz,Surf.Coat.Technol.202(2008)3396–3402.
[17]X.Yu,G.Li,J.AlloysComp.364(2004)193–198.
[18]L.S.Kasten,J.T.Grant,N.Grebasch,N.Voevodin,F.E.Arnold,M.S.Donley,Surf.
Coat.Technol.140(2001)11–15.
[19]R.G.Buchheit,S.B.Mamidipally,P.Schmutz,H.Guan,Corrosion58(2002)
3–14.
[20]J.P.Holgado,R.Alvarez,G.Munuera,Appl.Surf.Sci.161(2000)301–315.
[21]A.Pfau,K.D.Schierbaum,Surf.Sci.321(1994)71–80.
[22]M.F.Montemor,A.M.Simoes,M.G.S.Ferreira,M.J.Carmezim,Appl.Surf.Sci.
254(2008)1806–1814.
[23]Y.A.Teterin,A.Y.Teterin,A.M.Lebedev,I.O.Utkin,.Electron.Spectrosc.Relat.
Phenom.88–91(1998)275–279.
[24]J.Z.Shyu,K.Otto,W.L.H.Watkins,G.W.Graham,J.Catal.114(1988)22.
[25]A.E.Hughues,J.D.Gorman,P.J.K.Paterson,Corros.Sci.38(1996)1957–1976.
[26]-K.Bak,L.Hilaire,Appl.Surf.Sci.70–71(1993)191–195.
[27]A.Kotani,T.Jo,J.C.Parlebas,Adv.Phys.37(1988)37–85.
[28]J.Z.Shyu,K.Otto,W.L.H.Watkins,G.W.Graham,Catalysis114(1988)23–33.
[29]P.Campestrini,H.Terryn,A.Hovestab,J.H.W.deWit,Surf.Coat.Technol.176
(2004)365–381.
[30]L.E.M.Palomino,I.V.Aoki,H.G.deMelo,Electrochim.Acta51(2006)
5943–5953.
[31]Z.Liu,P.H.Chong,A.N.Butt,P.Skeldon,G.E.Thompson,Appl.Surf.Sci.247
(1–4)(2005)294–299.
[32]J.A.DeRose,T.Suter,A.Bałkowiec,J.Michalski,K.J.Kurzydlowski,P.Schmutz,
Corros.Sci.55(2012)313–325.
[33]M.Dabala,L.Armaelao,A.Buchberger,I.Calliari,Appl.Surf.Sci.172(2001)
312–322.
[34]A.J.Aldykiewiczs,A.J.Davenport,H.S.Isaacs,J.Electrochem.Soc.143(1)
(1996)147.
[35]S.A.Hayes,PuYu,T.J.O’Keefe,M.J.O’Keefe,J.O.Stoffer,J.Electrochem.Soc.

![Fig. 2. Cerium-based conversion coating (with [Ce] = 0.1 M; 300 s of immersion in the conversion bath) over AA2024-T3 alloy: (A) large scale XPS spectrum and (B) SEM micrograph of the cross section.](https://thumb-eu.123doks.com/thumbv2/123doknet/14403511.510399/6.892.204.674.121.457/cerium-conversion-coating-immersion-conversion-spectrum-micrograph-section.webp)

![Fig. 11. Cross section of Cerium-based conversion coating (with [Ce] = 0.1 M, immersion time: 300s) over AA2024T3 alloy after 5 h corrosion into 3.5 wt.% NaCl solution: (A) SEM view and (B) Auger spectroscopy analysis in the coating section (point 1) and w](https://thumb-eu.123doks.com/thumbv2/123doknet/14403511.510399/10.892.208.674.120.467/section-cerium-conversion-immersion-corrosion-solution-spectroscopy-analysis.webp)