HAL Id: hal-01264384
https://hal.archives-ouvertes.fr/hal-01264384
Submitted on 4 Mar 2016
HAL is a multi-disciplinary open access
archive for the deposit and dissemination of
sci-entific research documents, whether they are
pub-lished or not. The documents may come from
teaching and research institutions in France or
abroad, or from public or private research centers.
L’archive ouverte pluridisciplinaire HAL, est
destinée au dépôt et à la diffusion de documents
scientifiques de niveau recherche, publiés ou non,
émanant des établissements d’enseignement et de
recherche français ou étrangers, des laboratoires
publics ou privés.
Identification of the metallurgical parameters explaining
the corrosion susceptibility in a 2050 aluminium alloy
Mathilde Guérin, Joël Alexis, Eric Andrieu, Lydia Laffont-Dantras, Williams
Lefebvre, Grégory Odemer, Christine Blanc
To cite this version:
Mathilde Guérin, Joël Alexis, Eric Andrieu, Lydia Laffont-Dantras, Williams Lefebvre, et al..
Identifi-cation of the metallurgical parameters explaining the corrosion susceptibility in a 2050 aluminium alloy.
Corrosion Science, Elsevier, 2016, 102, pp.291-300. �10.1016/j.corsci.2015.10.020�. �hal-01264384�
O
pen
A
rchive
T
OULOUSE
A
rchive
O
uverte (
OATAO
)
OATAO is an open access repository that collects the work of Toulouse researchers and
makes it freely available over the web where possible.
This is an author-deposited version published in :
http://oatao.univ-toulouse.fr/
Eprints ID : 14698
To link to this article : DOI :
10.1016/j.corsci.2015.10.020
URL :
http://dx.doi.org/10.1016/j.corsci.2015.10.020
To cite this version : Guérin, Mathilde and Alexis, Joël and Andrieu,
Eric and Laffont-Dantras, Lydia and Lefebvre, Williams and Odemer,
Grégory and Blanc, Christine Identification of the metallurgical
parameters explaining the corrosion susceptibility in a 2050 aluminium
alloy. Corrosion Science, Vol.102. pp.291-300. ISSN 0010-938X
Any correspondance concerning this service should be sent to the repository
administrator:
staff-oatao@listes-diff.inp-toulouse.fr
Identification
of
the
metallurgical
parameters
explaining
the
corrosion
susceptibility
in
a
2050
aluminium
alloy
Mathilde
Guérin
a,b,
Joël
Alexis
b,
Eric
Andrieu
a,
Lydia
Laffont
a,
Williams
Lefebvre
c,
Grégory
Odemer
a,
Christine
Blanc
a,∗aUniversitédeToulouse,InstitutCARNOTCIRIMAT,UPS/CNRS/INPT,ENSIACET,4alléeEmileMonso,31030Toulouse,France bUniversitédeToulouse,LGP,ENIT/INPT,47Avenued’Azereix,65016,Tarbes,France
cUniversitédeRouen,GPM—UFRSciencesetTechniques,Avenuedel’Université—BP12,76801,SaintEtienneduRouvray,France
Keywords: Alloy Aluminium STEM Intergranularcorrosion Interfaces
a
b
s
t
r
a
c
t
Thecorrosionbehaviourofa2050aluminiumalloywasstudiedinaNaClsolution.Thestructureof precipitationdidnotfullyexplainthesusceptibilitytointergranular(inthe-T34state)and intragran-ularcorrosionfortheagedstate(the-T8state).Arelationshipbetweenthenatureofinterfaces,the grainscharacteristics(size,internalmisorientationandorientationaccordingtotheplaneexposedto theelectrolyte)ononehandandthecorrosionsusceptibilityofthealloyontheotherhandwasclearly established.Galvaniccouplingbetweengrainswithdifferentinternalmisorientationshelpedtoexplain theintergranularcorrosionsusceptibilityofthe-T34state.
1. Introduction
NewgenerationofAl–Cu–Li–Xalloysshowsremarkable combi-nationofdensity,mechanicalpropertiesandcorrosionresistance. Althoughotherphasescanbeobservedinthesealloys,the pre-cipitationofT1–Al2CuLiphaseiscommonlyconsideredasamajor
parameter to explain the corrosion behaviour of these alloys [1–9].Thisintermetallicphasepossessesamorenegative corro-sionpotentialthanthematrix[2,6].Whenthematerialisexposed toanaggressivemedium,agalvaniccouplingbetweentheT1phase
andthematrixoccursinfavourofthematrix.Inthe-T34 metallur-gicalstate,becauseofthepresenceofT1particlesonlyatthegrain
boundaries,galvaniccouplingleadstothedissolutionofthegrain boundarieswithamorenegativecorrosionpotentialthanforthe grains.Todesensitisethealloytointergranularcorrosion,anaging treatmentisappliedtothematerial.Thisleadstothe precipita-tionofT1particlesbothinthegrainsandatthegrainboundaries,
leavingtheagedstate(-T8).Fora-T8sample,becauseofthis struc-tureofprecipitation,thecorrosionpotentialsofthematrixandthe
∗ Correspondingauthorat:UniversitédeToulouse,CIRIMAT,UPS/CNRS/INPT,4 alléeEmileMonso,BP44362,31030ToulouseCedex4,France.
Fax:+330534323498.
E-mailaddress:christine.blanc@ensiacet.fr(C.Blanc).
grainboundariesarequitesimilar.Consequently,galvaniccoupling doesnotoccurbetweenthegrainandthegrainboundariesandthe materialissusceptibletointragranularcorrosion[2–4].
Ourresultsfrompreviousworkconfirmedthesusceptibilityof anAA2050alloytointragranularcorrosionafteranageing treat-mentat155◦Cfor30hand correlatedthistothehomogeneous
distributionofT1 phaseparticles inthegrainsand atthegrain
boundaries[9].For theAA2050-T34 alloy,susceptible to inter-granularcorrosion,amajorpartofcorrodedgrainboundariesdid notevidencethepresenceofT1precipitates.Thisresultsuggested
thatthepresenceoftheT1 phasewasnotnecessarytoinducea
susceptibilitytointergranularcorrosioninanAA2050-T34alloy. Furthermore,forboth-T8and-T34alloys,preferentialdissolution ofsomegrains andgrainboundarieswasrespectivelyobserved while,for-T8alloyforexample,homogeneousdistributionofT1
precipitateswasobservedbothinthegrainsandatthegrain bound-aries.Theseresultssuggestedthatparametersdifferentthanthe structureofprecipitationshouldcontributetoexplainthe corro-sionbehaviouroftheAA2050alloy.Thisshouldbeinagreement withresultsfoundinliterature.Forexample,Luoetal.showedthat arelationshipexistsbetweenthedislocationdensityinagrainand theabilityofthegrainboundarytobecorroded[10].Kimetal. showedthatthegrainboundarycharacterdistribution(GBCD)has agreateffectonintergranularcorrosionsusceptibilityofaluminum inHCl[11].Ralstonetal.workedontheinfluenceofthegrainsizeon
Fig.1. MethodologyusedforstudyingtheimpactofdifferentmetallurgicalparametersatthepolycrystalscaleonthecorrosionbehaviourofAA2050.
Fig.2. Opticalmicroscopeobservationsof(a)intergranularcorrosioninthe-T34sampleand(c)intragranularcorrosioninthe-T8sample.Bright-fieldTEMimagesof(b) the-T34sampleand(d)the-T8sample.
thecorrosionsusceptibilityofamaterialandnoticedthatthe‘grain size—corrosionresistance’relationshipiscomplex.Theysuggested thatthiscomplexitycouldbeincreasedduetotheheterogeneityof thegrainsizeinasample[12].
Theaimofthisstudywastodeterminetheoriginofthe suscepti-bilitytointergranularandintragranularcorrosionforAA2050-T34
andAA2050-T8alloysrespectively.Thestructureofprecipitation wasconsidered.However,attentionwasalsopaidto microstruc-turalparametersatthepolycrystalscale:thismeansthatthenature oftheinterfaces(eithergrainboundariesorsubgrainboundaries correspondingtothelevelofmisorientationattheinterfaces),the internalmisorientationandsizeofthegrains,andtheirorientation
Fig.3.HAADFSTEMobservationofGPzonesalonga<110>axisoftheAlmatrixintheAA2050-T34alloy.
withrespecttotheplaneexposedtotheelectrolytewere consid-eredaspotentialcriticalmetallurgicalparametersinfluencingthe corrosionsusceptibilityoftheAA2050alloy.Tostudytheimpactof thesemicrostructuralparameters,electronbackscatterdiffraction (EBSD)analyseswereperformedonhealthyandcorrodedsamples. 2. Experimentalprocedure
2.1. Material
The material used for this study was an aluminium–copper–lithiumAA2050-T34alloy(Albase,3.86%Cu, 0.86%Li,wt.%)providedbyConstellium(France).Thematerialwas receivedasa50mmthickplateformedbyhotrollingandfollowed by solutionheat treatment, water quenching and stretchingto achieve the final -T34 metallurgical state.A piece of theplate wasaged and corresponds to a -T8 metallurgicalstate. Dueto therollingprocess,themicrostructureofboththe-T34and-T8 samplesexhibitedahighdegreeofanisotropy.Observationsusing opticalmicroscopy(OM)ofthethickplateshowedthatthegrains werehighlyelongatedintherollingdirection.Theaveragesizesin thetransverse(LT)andshorttransverse(ST)directionswere350 and60mm,respectively,withalargediscrepancyinsizefromone graintoanother.Inthelongitudinal(L)direction,thediscrepancy issuchthatthecalculationofameanvalueismeaningless.Further EBSDanalysisrevealedtwopopulationsofgrains:onepopulation oflargepolygonisedgrains(withsizesvarying from350mmto 5mmintheLdirection)andanotherpopulationofrecrystallised grainswithsmallerdimensions(between5mmand20mminthe Ldirection).
2.2. StudyofthestructureofprecipitationoftheAA2050alloy Inordertotakeintoaccountthestructureofprecipitationin thecorrosionsusceptibilityoftheAA2050alloy,atransmission electronmicroscope(TEM)inthescanningtransmissionelectron microscopy(STEM)mode,whichhasahighdegreeoflocal pre-cision,wasused.STEMobservationswererecordedusingaJEOL ARM200FequippedwithaSchottkyFEG(fieldemissiongun)and aprobeCs-correctoroperatingat200kV.TheHAADF-STEM(High
AngleAnnularDarkField)andLAADF-STEM(LowAngleAnnular DarkField)imageswererecordedwitha0.1nmFWHMelectron
probesizeof30mradsemi-convergenceangle.Thedetectorranges weresetto54–220mradforHAADFand24–54mradforLAADF.The HAADFandLAADFsignalsaredominatedbyZ-contrastand diffrac-tioncontrast,respectively.Forthesetechniques,thesampleswere preparedusingmechanicalpolishingtoobtaina100mm-thickslice followedbyelectrolyticpolishingtomakeaportionofthesample transparenttoelectrons.
2.3. Studyofthecriticalmicrostructuralparametersatthe polycrystalscale
EBSDanalyseswereperformedforthe-T34 and-T8samples usingaJEOL7000Ffieldemissionscanningelectronmicroscope equippedwithaNordlysIIF+cameraatanacceleratingvoltage of15kV.TheEBSDanalyseswerehelpfulinboth analysingthe microstructureofthealloyandcharacterisingthecorrosion dam-ageafterthetests.Allofthecorrosiontestswereperformedover 72handwereconductedinacontinuousimmersionatopencircuit potentialina0.7MsolutionofNaCl,whichwaspreparedby dis-solvingNormapurchemicalsaltsindistilledwater.Duringthetests, thesolutionwasstirredandopenedtotheair.Thetemperaturewas maintainedat25◦CusingaJulaborefrigeratedcirculator.Foreach
sample,EBSDmapswereperformedonthesame1.5mm×3mm areawithastepsizeof1–2mminthe(LT-ST)planebothbeforeand afterthecorrosiontests.Duringthecorrosiontests,onlythiszone wasexposedtotheelectrolyte(avarnishwaspaintedontoprotect therestofthesurface).Thesurfaceexposedtotheelectrolytewas largeenoughtoobtainrepresentativeresults.Overall,foreach sam-ple,morethan250grainswereanalysed.TheEBSDdatawerethen post-processedusingthecommercialorientationimagingsoftware packageOxfordChannel5.Tominimisemeasurementerrors,all grainscomprisinglessthan3pixelswereautomaticallyremoved fromthemapspriortodataanalysis.Inaddition,toeliminate spuri-ousboundariescausedbyorientationnoise,alowerlimitboundary misorientationcut-offof2◦wasused.Theanalysespermittedthe
determinationoftheorientationofthegrains(seeFig.1,map1) andthenatureoftheinterfaces(seeFig.1,map2).Inthiswork, theword‘interfaces’referstothegrainboundariesindependentof theirlevelofmisorientation.A10◦criterionwasusedto
differenti-atelowanglegrainboundaries(i.e.subgrainboundaries)andhigh anglegrainboundaries(i.e.grainboundaries).Thismisorientation waschosenbasedondataavailableforpurealuminium[13].Two
Fig.4. HAADFSTEMimagesofsubgrainboundariesintheAA2050-T34alloy(Z-contrast).
Fig.5.(a)HAADFSTEM(i.e.Z-contrast)and(b)LAADFSTEM(i.e.diffractioncontrast)imagesofthesameregionintheAA2050-T34alloy.Theregiontotherightisinperfect <110>orientationwhereasthecrystalorientationslightlychangesacrossthesubgrainboundaryidentifiedbythealignmentofdislocationsin(a).Thisslightchangeofcrystal orientationisclearlyvisibleonthehighermagnificationHAADFSTEMimagesdisplayedin(c)and(d),whichshowprecipitatesindicatedbyawhitedottedsquarein(a).
categories of grainboundaries were distinguished: coincidence sitelatticegrainboundaries(CSL)andrandomgrainboundaries. Incoincidentsite latticegrainboundaries,thedegreeoffit (6) betweenthestructuresofthetwograinsisdescribedbythe recip-rocaloftheratioofcoincidencesitestothetotalnumberofsites orientedinoppositiontotherandomgrainboundarieswherethe structurewasobservedasbeingdisorganised.Thegrainsizewas quantifiedbymeasuringthegrainareaandcalculatingthe equiv-alentgraindiameter assumingeach grain asa circle. Afterthe corrosiontests,allthecorrodedinterfacesandgrainsinthesame
zonewereanalysed.Thisrepresentedapproximately50interfaces (forthe-T34metallurgicalstate)and15grains(forthe-T8 metal-lurgicalstate)ineachsample.Theanalysiswasreproducedtwice foreachmetallurgicalstateleadingtosimilarconclusions.Afterthe testing,thecorrosionproductswereremovedusinggentle mechan-icalpolishing.ThecorrodedzonewasanalysedusingSEM with theelectronbackscatterdetectortoeasilyidentifyanycorrosion defects(seeFig.1,map3a).Thecorrosiondefectswereisolated usingimageanalysis(ImageJsoftware,seeFig.1,map3b).Thislast mapwassuperimposedonmap1ormap2toobtainacorrosion
map(seeFig.1,map4).Themisorientationlevelforeachcorroded interfaceandthecharacteristicsoftheadjacentgrains(forthe-T34 metallurgicalstate)orofthecorrodedgrains(forthe-T8 metallur-gicalstate)wereextractedfrommap4.Thegraincharacteristics studiedoverthisareaincludedthesize,theinternalmisorientation andtheorientationrelativetotheplaneexposedtotheelectrolyte. 3. Experimentalresultsanddiscussion
Asalreadydiscussed,a previousstudyonthesamematerial revealedasusceptibilitytointergranularcorrosionformaterialin the-T34state(Fig.2a)andtointragranularcorrosioninthe-T8 state(Fig.2c)[9].TEMobservationsfor-T8samplesagreedwith theliterature;theT1phaseprecipitateswereobservedbothinthe
grainsandatthegrainboundaries,explainingthesusceptibilityto intragranularcorrosion(Fig.2d).Forthe-T34samples,noT1
pre-cipitatewasobservedinthemajorpartsofthegrainboundaries (Fig.2b).
3.1. CorrosionbehaviouroftheAA2050-T34alloy
The corrosion behaviour of the system investigated being stronglydependentonthestructureofprecipitationassuggested bytheliterature[1–9],it isnecessarytodeterminewhetherT1
and/oru′precipitates,thatarealsoexpectedinsuchanalloy,can
beobservedintheT34state.BecausepreviousTEMobservations didnotallowthepresenceofT1precipitatestobeevidencedinthe
majorpartofthegrainboundaries,HAADFSTEMobservationswere performed.TheyrevealedfirstthatonlyCu-richGPzonesare iden-tifiedinthematrix(Fig.3)awayfromsubgrainorgrainboundaries, asexpected.ThelengthoftheseGPzonesmeasuredbyprojection alonga<110>axisisrangingbetween1and3nm.
Specialattentionwaspaidtothecharacterizationofsubgrain boundariesandHAADFSTEMimages(Fig.4)indicatedthat precip-itateswerealreadypresentalongtheseinterfaces.However,the densityofprecipitationwasfoundtostronglyvaryfromone sub-grainboundarytoanotheroralongthesameinterface.Inorder torevealelasticdistorsionsinducedbythedislocationsassociated totheseinterfaces,LAADFSTEMmodewasused.Suchamethod hasrevealeditsrelevancefortheimagingofdislocationsinSTEM [14].In theAA 2050-T34alloy,thesameregionswere simulta-neouslyimagedinLAADF(Fig.5b)andHAADFSTEM(Fig.5a)to unambiguouslydissociatethecontrastassociatedwithasubgrain boundary(i.e.thealignmentofdislocationsacrosswhichthecrystal orientationisslightlytitled)totheZ-contrastrevealingthe even-tualpresenceofprecipitates.Fig.5showsasubgrainboundaryina nearly<110>orientation.Precipitateswereclearlyvisibleinsome regionsofthisinterfaceandweredisplayedinlargermagnification inFig.5candd.Acrossthesetwoimages,fromrighttoleft,the lossoflatticeresolutionintheAl-matrixwasduetoasmall dis-orientationofthelatticeacrossthesubgrainboundary.InFig.5c, theprecipitateinthelowerpartoftheimageexhibitedatypical contrastofaT1precipitatealong<110>zoneaxisofaluminium,as
reportedintheliterature[15,16].TheCu-richplanardefectslying along{111}planesofaluminiuminFig.5dcouldnotbeclearly identified.TherecentworkbyGaoetal.[17]hasreportedGP-T1
zones,whichstructuredidnotcorrespondtotheCu-richplanar defectsinFig.5.TheGP-T1zonesreportedbyGaoetal.actually
dis-playtwoCu-richplanesparallelto{111}Al,separatedbyaregion withacompositionthatcouldnotbedeterminedunambiguously thoughitscontrastseemstoindicatethatit consistsofasingle planeofaluminium.Ourobservationsweremoreconsistentwith thosemadebyAraullo-Petersetal.byatomprobetomography[18]. TheseauthorshavedemonstratedacomplexcouplingbetweenMg andCusegregationsalongdislocationswiththeformationofplanar
defectsenrichedinCu,identifiedasT1precursors.Nevertheless,the
mechanismofformationofT1precipitatesfallsoutsidethescope
ofthispaper,whichpurposeistorelatemicrostructural character-izationtothecorrosionbehaviouroftheAA2050-T34alloy.
Themainresultherewasthat,intheT34state,thealloyalready containedT1precipitatesandotherplanardefectsenrichedin
cop-peralongsubgrainboundaries.Lithiumcouldalsobepresentin theinterfaces but,due toits lowamount,it wasimpossible to detectthiselement.Despitethefactthatnou’precipitatecould beobserved,wecannotexcludethatsomeoftheseprecipitates werepresentintheAA2050-T34alloyalonginterfaces.The den-sityofprecipitatesand planardefectsstronglyvariedfromone subgraintoanotherandwemadetheassumptionthatthese varia-tionswereduetothenatureofdislocationspresentinthesubgrain boundaries.Assuggestedbyseveralauthors,agrainboundarywith ahighdegreeofatomicdisarrangementshouldreactmorewith impuritiesoralloyingelementsthanagrainboundarywithhigh coincidenceleveloreventhanagrainboundaryoflowangle mis-orientation[19–21].Thissuggestedthatthelevelofmisorientation ofinterfacescouldbecorrelatedtotheircorrosionsusceptibility.A detailedinvestigationwashencerequiredtoestimatethe possibil-ityofsuchacorrelation.Thisinvestigation,whichwasperformed usingEBSDanalyses,ispresentedhereafter.
TheresultsoftheEBSDanalysesperformedpriortocorrosion testsfortheAA2050-T34samplearepresentedinFig.6.The dis-tributionofinterfacesaccordingtotheirlevelofmisorientationis showninFig.6a.Thelevelofmisorientationofeachinterfacewas determinedbymeasuringthemisorientationbetweenthegrains situated onboth sidesof theinterface.Theresultsshowedthat 85% ofinterfaceswere subgrainboundariesand only15% were grainboundaries.Amongthegrainboundaries,20%wereCSLgrain boundariesand80%wererandomgrainboundaries.63interfaces representedthemajorityoftheCSL(35%,Fig.6b).Therefore,during analysisofthecorrodedsamples,onlythe63CSLgrainboundaries wereconsidered.
AftercompletingthecorrosiontestsintheAA2050-T34sample, thecorrodedinterfaceswerelocated,andthelevelsof misorien-tationwererecorded(Fig.1).Theanalysiswasperformedon50 corrosiondefects,andledtotheestablishmentofarelationship betweenthelevelofmisorientationofaninterfaceandits suscep-tibilitytocorrosion.Fig.6ashowsthecorrodedinterfacefrequency accordingtotheirmisorientationlevel.Thedistributionwas differ-entfromthedatatakenpriortothecorrosiontest.Atotalof70%of thecorrodedinterfacesweregrainboundarieswhereasonly30% ofthecorrodedinterfacesweresubgrainboundaries.Considering thehighproportionofsubgrainboundaries,theseresultssuggesta higherresistancetocorrosionofthesubgrainboundaries. More-over, amongthecorrodedgrainboundaries,theresultsshowed thatthemostmisorientedinterfacestendedtobemore suscepti-bletocorrosion.Thestrongreactivityofthegrainboundarieswith ahighlevelofmisorientationmaybelinkedtotheirdisarranged structureandcorrelatedwiththeirhighenergy[19].Onthe con-trary,theresistancetocorrosionofthesubgrainboundariesshould beassociatedwiththeirlowenergy.Theseresultswereconsistent withotherworkonvariousmetals[20,22–24].Itshouldbenoted thattheinterfacesusceptibilitytocorrosion,andinparticularof somesubgrainboundariescompared totheothers,mayalsobe explainedbytheirchemicalcompositionandstructureof precipi-tation,ashighlightedintheSTEM-HAADFobservations.Itwillbe interestingtostudytherelationshipbetweenthechemical com-position/structureofprecipitationofaninterfaceanditslevelof misorientationbutthisshouldbethesubjectoffuturestudy. Con-cerningthesusceptibilitytocorrosionoftheCSLgrainboundaries, onlyfourcorrosiondefectsat63grainboundarieswereidentified. Anintergranularcorrosionsusceptibilityindexwasdeterminedby theratiooftheproportionofcorrosiondefectscorrespondingto
Fig.6. AA2050-T34alloy—(a)Distributionofthelevelofmisorientationoftheinterfacesbeforeandafterthecorrosiontests;(b)distributionoftheCSLgrainboundaries beforecorrosiontests.
Fig.7. AA2050-T34alloy—(a)Distributionofthegrainsurfaceareasbeforeandafterthecorrosiontestsforcorrosionatsubgrainboundariesandatgrainboundaries;(b) distributionofthegrainsurfacearearatioofthegrainssituatedonbothsidesofthecorrosiondefectsatthegrainboundaries.
Table1
StatisticalanalysisoftheintergranularcorrosionintheAA2050-T34alloybasedonthenatureoftheinterfacesatthepolycrystalscale.
Natureoftheinterfaces Subgrainboundaries Randomgrainboundaries CSLgrainboundaries
%Innon-corrodedspecimen(a) 85 12 3
%Incorrodedspecimen(b) 30 62 8
Fig.8.AA2050-T34alloy—(a)Relationshipbetweenthegrainsurfaceareaandtheinternalgrainmisorientation;(b)distributionoftheinternalgrainmisorientationbefore andafterthecorrosiontestsforcorrosionatthesubgrainboundaries.
acertaintype ofinterface(subgrain boundaries,randomorCSL grainboundaries)dividedbytheproportionofinterfacesofthis sametypeinthesample.TheresultsshowedthattheCSLgrain boundariesweretwo timesless sensitivetocorrosionthanthe randomgrainboundaries,whichwasasexpectedfromprevious observations(Table1).Moreover,thecalculatedindicesshowed thatsubgrainboundariesweretheleastsensitiveinterfaces.This resultwaspartiallyconsistentwiththeliteratureevenifCSLgrain boundarieswereoftenconsideredmoreresistantthansubgrain boundaries[20].Thistrendmustbefurtherconfirmedduetothe lownumberof63studied.
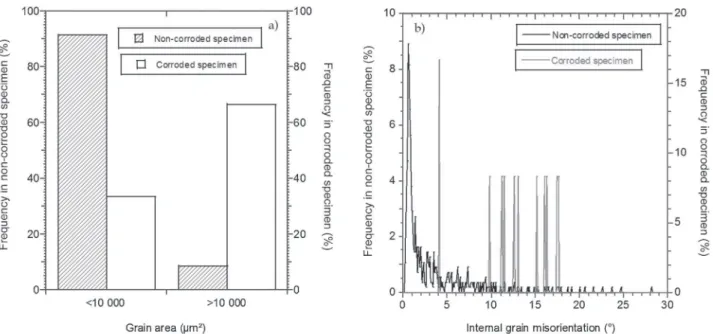
Inlightofthepreviousresults,itwasimportanttodistinguish corrosionatgrainboundariesandatsubgrainboundariesforthe AA2050-T34alloy.Inbothcases,thecharacteristicsofthegrains situatedoneachsideofacorrosiondefect(inthecaseof corro-sionatthegrainboundaries)orcontainingcorrosiondefects(in thecase of corrosion atthe subgrainboundaries)should influ-encethereactivityoftheinterface.Therefore, thepropertiesof thegrainswerealsoinvestigated.Theanalysesperformedprior tothecorrosiontestsshowedthatmostofthegrainswereless than10,000mm2inarea(Fig.7a).Moreprecisely,50%ofthegrains
measuredlessthan100mm2.Afterthecorrosiontests,thegrains
containingcorrodedsubgrainboundariesintheAA2050-T34 sam-pleswereidentifiedandtheirareaswererecorded.Fig.7ashows thatthecorrosionatsubgrainboundariesoccurredingrainswith alargearea(>10,000mm2,andmoreprecisely>80,000mm2).This
wasbestexplainedbythepresenceofmanysubgrainboundaries and dislocations insidethe largepolygonizedgrains, leadingto asignificantamountof storedenergyandconsequently,astate ofnon-thermodynamicequilibrium inthesegrains.Theanalysis of corrosion at thegrain boundaries of theAA 2050-T34 sam-pleincludedexaminingthegrainssituated onboth sidesofthe corrosion defects and recording theirareas. Fig. 7a shows the distributionof thegrains experiencing corrosion attheir inter-faces. Results highlighted that intergranular corrosion at grain boundariesoccurredattheinterfacebetweengrainsofvariable surfaceareas.Butbydrawingthedistributionofthearearatio, which representstheareaof thesmallestgraindivided bythe area of the largest grain (for the grains situated on each side of a corrosion defect), a dominant group of corrosion defects for a low area ratio (0–0.2, Fig. 7b) was observed. This result
meansthatintheAA 2050-T34sample,intergranularcorrosion developedmainlybetweengrainswithdisparateareas:agalvanic couplingbetweenasmallgrainandalargegrainshouldexplainthis resultbecausethereisastrongerreactivityattheirshared inter-face.Indeed,severalworkshaveshownthatthecorrosionpotential ofametalvariedwiththedislocationdensity[24,25]andthegrain size[26,27].Itwaspossibletotransposethisresultonthe poly-crystalscalebyconsideringthegalvaniccouplingbetweengrains ofdifferentsizesandpossessingdifferentdislocationdensities.
Therelationshipbetweenthegrainsizeanddislocationdensity isshowninFig.8aforanon-corrodedAA2050-T34samplewhere thedislocationdensityofagrainwasmeasuredthroughits inter-nalmisorientation.Fig.8ashowsthatthesmallestgrainshavethe smallestinternalgrainmisorientationinrelationtothelow dislo-cationdensity.Moreover,the-T34metallurgicalstateexhibiteda highproportionofgrainswithalowinternalmisorientation(<2◦,
Fig.8b).Afterthecorrosiontests,thegrainscontainingcorroded subgrainboundariesfortheAA2050-T34samplewereidentified andtheirinternalmisorientationsrecorded.Asexpected, corro-sionatthesubgrainboundariesoccurredingrainscontainingahigh internalmisorientation(Fig.8b).Whenconsideringthecorrosionat thegrainboundariesfortheAA2050-T34sample,theratiobetween theinternal misorientation of each grainsituated oneach side ofthecorrodedgrainboundariesmustbeexamined.Theresults showedthatthecorrodedgrainboundariescorrelatedtoahigh graininternalmisorientationratio,suggestingagainthatagalvanic couplingbetweenthegrains withdifferentinternal misorienta-tionsledtocorrosionattheirsharedinterface.Fig.9illustrates thisphenomenonofgalvaniccoupling.Intergranularcorrosionwas observedattheinterfacebetweenagrainwithlowinternal misori-entation(ontherightofthecorrosiondefectn◦1)andagrainwith
manysubgrainboundaries,whichassumedahighdegreeof inter-nalmisorientation(totheleftofthecorrosiondefectmarkedn◦1).
ItnotedthatdespiteoftheaveragebehaviourshowninFig.8a,the lowinternalmisorientationwithinagrainisnotnecessarily asso-ciatedwiththesmallsizeofthisgrain(identifiedasthegraintothe leftofcorrosiondefectmarkedn◦2).Therefore,toagreaterextent
thanthegrainsize,theinternalgrainmisorientationseemstobethe mostrelevantparametercapableofexplainingtheintergranular corrosionsusceptibilityoftheAA2050-T34alloy.
Fig.9. EBSD(IPF-Z0)micrographofthecorrosiondefectssituatedbetweengrainsshowinglargediscrepanciesofinternalmisorientationintheAA2050-T34alloyandthe correspondingSEMmicrograph.
3.2. CorrosionbehaviouroftheAA2050-T8alloy
ThesusceptibilitytointragranularcorrosionofAA2050-T8alloy waswell-correlatedtothehomogeneousdistributionofT1
pre-cipitatesbothinthegrainsandatthegrainboundaries.However, aspreviously noticed, sucha structure of precipitation didnot explainthepreferential dissolutionofsomegrainscomparedto theothers.ResultsobtainedfortheAA2050-T34alloysuggested thatmetallurgicalparametersatthepolycrystalscaleshould con-tributetoexplainthecorrosionsusceptibilityofAA2050-T8alloy also.First,attentionwaspaidtotheinfluenceofthegrainsizeon thecorrosionsusceptibility.AsfortheAA2050-T34alloy,astrong reactivityoflargegrainswasalsoobservedfortheAA2050-T8 sam-ple.Thedistributionofgrainsaccordingtotheirareaispresented inFig.10aforthis metallurgicalstate.Thedistributionobtained fortheAA 2050-T8samplewassimilartothatoftheAA 2050-T34samplebecausetheageingtreatmentdidnotmodifythesize ofthegrains.Forthe-T8state,whichissusceptibleto intragran-ularcorrosion, theanalysis performedafter thecorrosion tests showedthepreferentialdissolutionofgrainswithanareagreater than10,000mm2(Fig.10a).Thisresultwasinagreementwiththe
dataobtainedfortheAA2050-T34sampleforcorrosionat sub-grainboundariesandwasbetterexplainedbythepresenceofmany subgrainboundariesanddislocationsinsidethelargepolygonized grains,leadingtoasignificantamountofstoredenergyand con-sequently,astrongreactivity.Theliteraturereportsanevolution ofthecorrosionpotentialwiththedislocationdensitybutwithout anyidentifiedtrends[25].Itwasassumedinthepresentworkthat thegrainswiththehighestdislocationdensityhavethemost nega-tivecorrosionpotential.Incontrast,thesmallrecrystallisedgrains didnotexhibitsubgrainboundariesandtheircorrosionresistance wasconsequentlyhigher.
Then,theinfluenceoftheinternalmisorientationofagrainon itscorrosionsusceptibilitywasconsidered.Concerningthe rela-tionshipbetweenthegrainsizeanddislocationdensity,thesame resultwasobtainedforanAA2050-T8sampleasforanon-corroded AA 2050-T34 sample(Fig.8a), i.e. thesmallestgrains havethe smallestinternalgrainmisorientation.Moreover,asforthe-T34, thenon-corroded-T8metallurgicalstatesexhibitedahigh propor-tionofgrainswithalowinternalmisorientation(<2◦,Fig.10b).
Afterthecorrosiontests,resultsshowedthatintragranular corro-sionoccurredingrainscontainingahighinternalmisorientation (Fig.10b)showingthat themostrelevantparametercapableof explaining the intragranular corrosion susceptibility of the AA 2050-T8alloywastheinternalmisorientationofthegrains,asfor theintergranularcorrosionsusceptibilityoftheAA2050-T34alloy. AnotherparameterwasinvestigatedintheAA2050-T8sample: thegrain orientationrelated to theplaneexposed tothe elec-trolyte.Theanalysisperformedafterthecorrosiontestsonthe-T8 statesamplesshowedthatthemajorityofthecorrodedgrainsare orientedaccordingto(111)planesrelativetotheexposedplane (Fig.11).Itisworthnotingthatthe(111)planesarethedenser planesandtheyarethehabitplanesoftheT1phaseintheface
cen-trecubicstructureofaluminium.Theliteraturereportedthatthe dissolutionbehaviourofthegrainsdependedontheorientation ofthecrystalrelativetotheplaneexposedtotheelectrolyte[28]. Consequently,thepreferentialcorrosionofgrainsorientedwith the(111)planesexposedtotheelectrolytemaybeexplainedby amorenegativecorrosionpotentialthanforgrainsorientedtothe othercrystalplanes.Itissuggestedthatgalvaniccouplingbetween grainswithdifferentorientationsledtothepreferentialcorrosion ofsomegrains.Thishypothesisshouldbeadvancedtoexplainthe susceptibilityofthe-T34statetointergranularcorrosion.Inthis case,galvaniccouplingbetweengrainswithdifferentorientations
Fig.10.AA2050-T8alloy—(a)Distributionofthegrainsurfaceareasand(b)distributionoftheinternalgrainmisorientationbeforeandafterthecorrosiontestsfor intragranularcorrosion.
mayincreasethecorrosionsusceptibilityoftheirsharedinterface. FurthermeasurementsoftheVoltapotentialofthegrains accord-ingtotheircrystallineorientationusingKelvinForceMicroscopy shouldconfirmthishypothesis.
4. Conclusions
ThecorrosionbehaviourofAA2050wasstudiedforboth nat-urallyaged(-T34)andartificiallyaged(-T8)metallurgicalstates. Theimpactofthemicrostructuralparametersonbothintergranular (-T34)andintragranular(-T8)corrosionmechanismswas investi-gated.Theconclusionsareasfollows:
1Thestructureofprecipitationcontributedtoexplainthe suscepti-bilitytocorrosionoftheAA2050alloy.However,thepresenceof T1phaseprecipitatesatthegrainboundariesofa-T34alloywas
notnecessarytosensitisethealloytointergranularcorrosion. Theintragranularcorrosionofa-T8alloycanbeexplained par-tiallybyT1precipitateshomogeneouslydistributedinthegrains
andatthegrainboundariesbutsuchahomogeneousdistribution didnotexplainthestrongreactivityofsomegrainscomparedto others.
2Othermetallurgicalparametersatthepolycrystalscalewereto betakenintoaccounttoexplainthecorrosionmechanismsinthe AA2050alloy.
3Thenatureoftheinterfaceswasfoundtoplayadominantroleon theintergranularcorrosionsusceptibilityofthe-T34alloywitha highercorrosionresistanceforinterfacescharacterisedbyalow levelofmisorientation.
4Forboththe-T34and-T8samples,theinternalmisorientationof thegrainsisadrivingfactorintheintergranularand intragran-ularcorrosion mechanisms.Galvanic couplingbetweengrains withstronglydifferentinternalmisorientationshouldleadtothe corrosionoftheirsharedinterface.Grainswithhighinternal mis-orientationwerefoundtobethemostsusceptibletocorrosion. 5For-T8samples,grainsorientedinthe(111)planes,inwhichthe
T1phaseprecipitated,werethemostsusceptibletointragranular
corrosion. Acknowledgments
Thisworkwasfinanciallysupportedbythe“PRES/Région Midi-Pyrénées”.TheauthorswouldliketothanktheAirbusGroupfor manyfruitfuldiscussions,Constelliumforsupplyingthematerial. TheTEManalyseswereperformedwiththehelpofMarie-Christine Lafont.
References
[1]J.G.Rinker,M.Marek,Microstructure,toughnessandstresscorrosioncracking behaviorofaluminumalloy2020,Mater.Sci.Eng.64(1984)203–221.
[2]R.G.Buchheit,J.P.Moran,G.E.Stoner,Localizedcorrosionbehaviorofalloy 2090-theroleofmicrostructureheterogeneity,Corrosion46(1990)610–617.
[3]P.Niskanen,T.H.Sanders,J.G.Rinker,M.Marek,Corrosionofaluminumalloys containinglithium,Corros.Sci.22(1982)283–304.
[4]V.Proton,J.Alexis,E.Andrieu,J.Delfosse,A.Deschamps,F.DeGeuser,M.C. Lafont,C.Blanc,Theinfluenceofartificialageingonthecorrosionbehaviour ofa2050aluminium–copper–lithiumalloy,Corros.Sci.80(2014)494–502.
[5]C.Kumai,J.Kusinski,G.Thomas,T.M.Devine,Influenceofagingat200◦Con
thecorrosionresistanceofAl–LiandAl–Li–Cualloys,Corros.Sci.45(1989) 294–302.
[6]R.G.Buchheit,J.P.Moran,G.E.Stoner,ElectrochemicalbehaviorofT1(Al2CuLi)
intermetalliccompoundanditsroleinlocalizedcorrosionofAl–2%Li–3%Cu alloys,Corrosion50(1994)120–130.
[7]J.E.Kertz,P.I.Gouma,R.G.Buchheit,Localizedcorrosionsusceptibilityof Al–Li–Cu–Mg–ZnalloyAF/C458duetointerruptedquenchingfrom solutionizingtemperatures,Metall.Mater.Trans.A33(2001)2561–2573.
[8]H.Y.Li,Y.Tang,Z.D.Zeng,F.Zheng,ExfoliationcorrosionofT6-andT3-aged AlxCuyLizalloy,Trans.NonFerrousMater.Soc.China18(2008)778–783.
[9]M.Guérin,E.Andrieu,G.Odemer,J.Alexis,C.Blanc,Effectofvarying conditionsofexposuretoanaggressivemediumonthecorrosionbehaviorof the2050Al–Cu–Lialloy,Corros.Sci.85(2014)455–470.
[10]C.Luo,X.Zhou,G.E.Thompson,A.E.Hughes,Observationsofintergranular corrosioninAA2024-T351:theinfluenceofgrainstoredenergy,Corros.Sci. 61(2012)35–44.
[11]S.H.Kim,U.Erb,K.T.Aust,G.Palumbo,Grainboundarycharacterdistribution andintergranularcorrosionbehaviourinhighpurityaluminium,Scr.Mater. 44(2001)835–839.
[12]J.G.Brunner,N.Birbilis,K.D.Ralston,S.Virtanen,Impactofultrafine-grained microstructureonthecorrosionofaluminiumalloyAA2024,Corros.Sci.57 (2012)209–214.
[13]M.Winning,A.D.Rollett,Transitionbetweenlowandhighanglegrain boundaries,ActaMater.53(2005)2901–2907.
[14]M.Tanaka,K.Higashida,K.Kaneko,S.Hata,M.Mitsuhara,Cracktip dislocationsrevealedbyelectrontomographyinsiliconsinglecrystal,Scr. Mater.59(2008)901–904.
[15]P.Donnadieu,Y.Shao,F.DeGeuser,G.A.Botton,S.Lazar,M.Cheynet,M.De Boissieu,A.Deschamps,AtomicstructureofT1precipitatesinAl–Li–Cualloys
revisitedwithHAADF-STEMimagingandsmall-angleX-rayscattering,Acta Mater.59(2011)462–472.
[16]C.Dwyer,M.Weyland,L.Y.Chang,B.C.Muddle,Combinedelectronbeam imagingandab-initiomodelingofT1precipitatesinAl–Li–Cualloys,Appl.
Phys.Lett.98(2011)201909.
[17]Z.Gao,J.Z.Liu,J.H.Chen,S.Y.Duan,Z.R.Liu,W.Q.Ming,C.L.Wu,Formation mechanismofprecipitateT1inAlCuLialloys,J.AlloyCompd.624(2015)
22–26.
[18]V.Araullo-Peters,B.Gault,F.deGeuser,A.Deschamps,J.M.Cairney, MicrostructuralevolutionduringageingofAl–Cu–Li–xalloys,ActaMater.66 (2014)199–208.
[19]M.Froment,Surlemécanismedelacorrosionintergranulairedesmatériaux métalliques,J.Phys.Colloq.36(1975)371–385.
[20]S.H.Kim,U.Erb,K.Aust,G.Palumbo,Grainboundarycharacterdistribution andintergranularcorrosionbehaviorinhighpurityaluminum,Scr.Mater.44 (2001)835–839.
[21]V.Keast,D.Williams,Grainboundarychemistry,Curr.Opin.SolidStateMater. Sci.5(2001)23–30.
[22]B.W.Bennett,H.W.Pickering,Effectofgrainboundarystructureon sensitizationandcorrosionofstainlesssteel,Metall.Trans.A18(1987) 1117–1124.
[23]S.R.Ortner,V.Randle,Astudyoftherelationbetweengrainboundarytype andsensitisationinpartially-sensitisedAISI304stainlessstellusingelectron back-scatteringpatterns,Scr.Metall.23(1989)1903–1908.
[24]C.Luo,X.Zhou,G.E.Thompson,A.E.Hughes,Observationsofintergranular corrosioninAA2024-T351:theinfluenceofgrainstoredenergy,Corros.Sci. 61(2012)35–44.
[25]S.R.Salimon,A.I.Salimon,A.M.Korsunsky,Theevolutionofelectrochemical, microstructural,andmechanicalpropertiesofaluminiumalloy2024-T4 (D16AT)duringfatiguecycling,Proc.Inst.Mech.Eng.PartG(2010)339–353.
[26]K.D.Ralston,D.Fabijanic,N.Birbilis,Effectofgrainsizeoncorrosionofhigh purityaluminium,Electrochim.Acta56(2011)1729–1736.
[27]V.Proton,J.Alexis,E.Andrieu,J.Delfosse,M.-C.Lafont,C.Blanc,
Characterisationandunderstandingofthecorrosionbehaviourofthenugget ina2050aluminiumalloyfrictionstirweldingjoint,Corros.Sci.73(2013) 130–142.
[28]L.Lapeire,E.MartinezLombardia,K.Verbeken,I.DeGraeve,L.A.I.Kestens,H. Terryn,Effectofneighboringgrainsonthemicroscopiccorrosionbehaviorof agraininpolycrystallinecopper,Corros.Sci.67(2013)179–183.