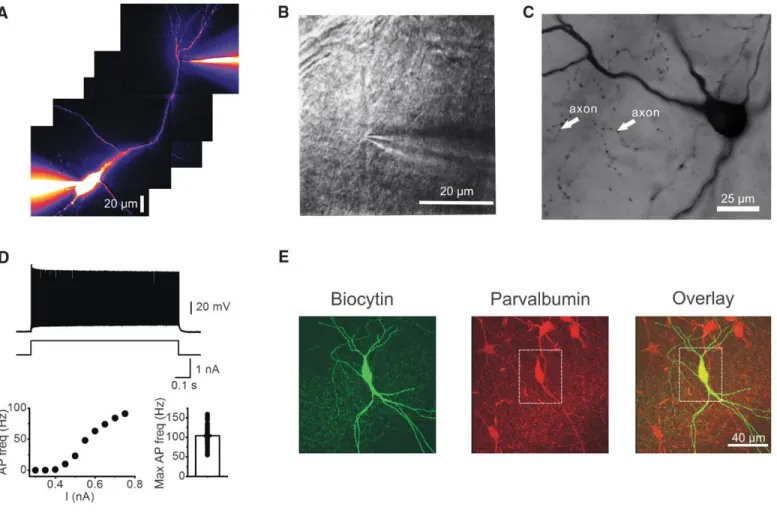
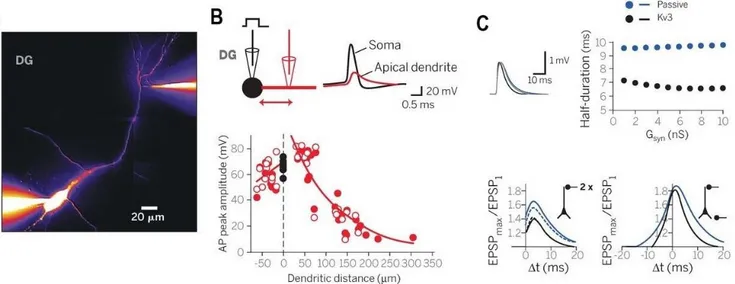
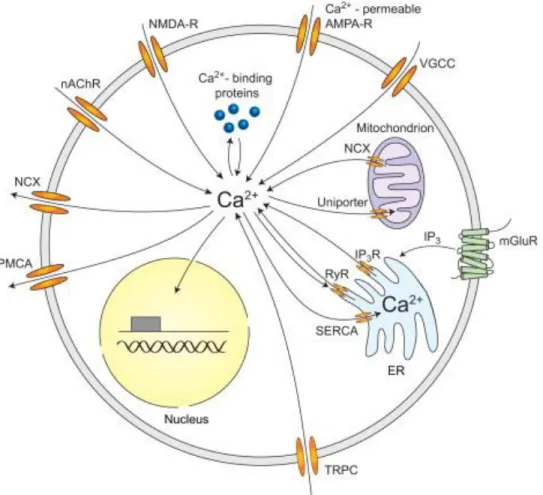
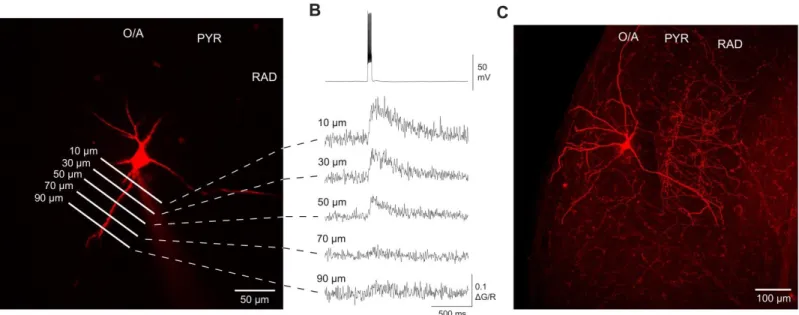
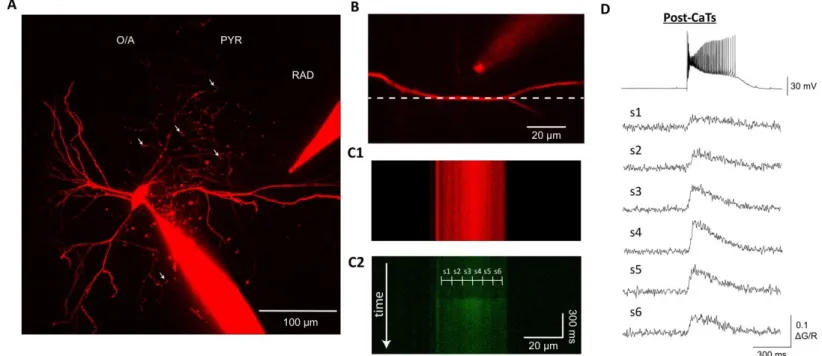
Ca²+ mechanisms of synaptic integration and plasticity in inhibitory interneurons
360
0
0
Texte intégral
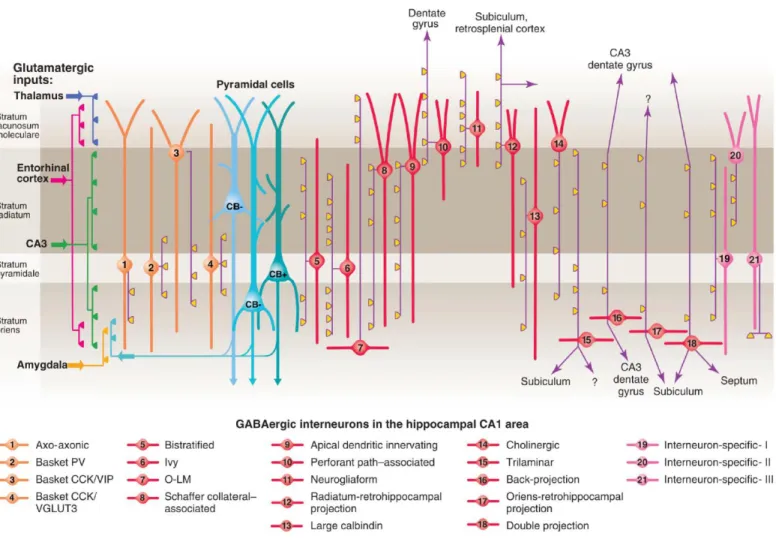
Figure
+7
Documents relatifs